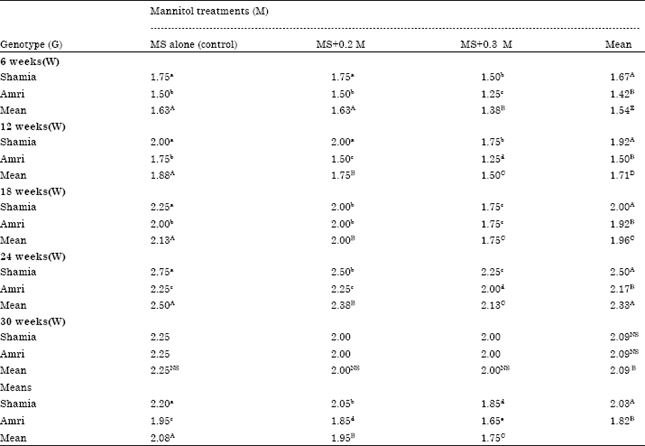
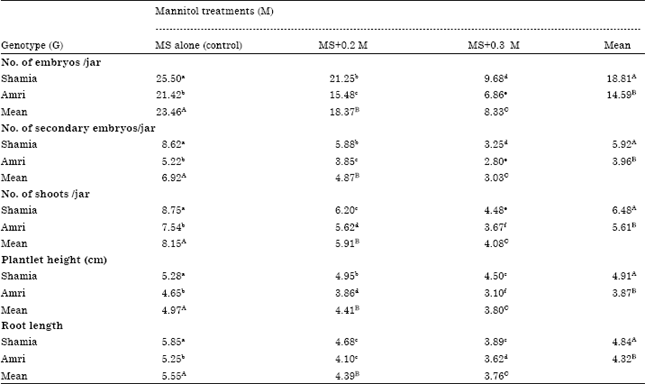
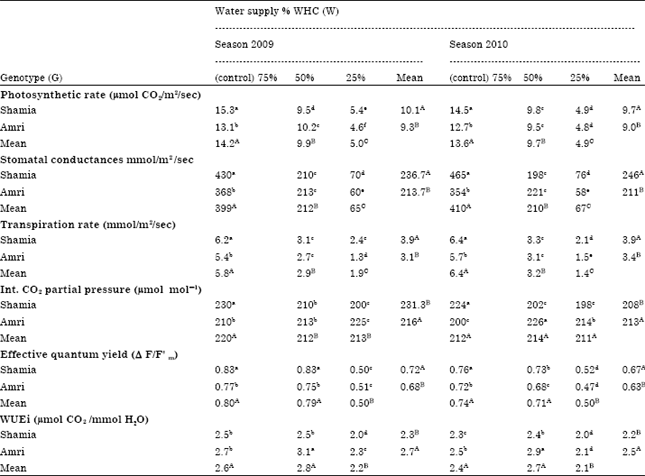
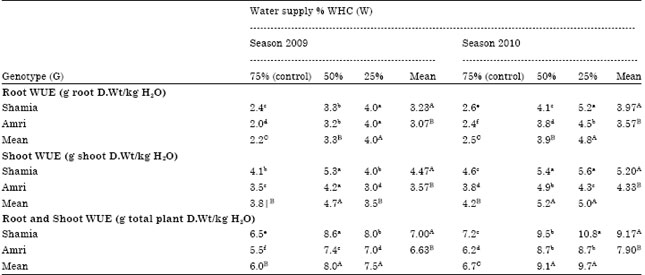
Research Article
In vitro Selection and Photosynthetic Characterization of Date Palm Regenerated Seedling as Affected by Water Stress
Department of Agriculture Bot, Faculty of Agriculture, Mansoura University, Egypt
A.M. Hanan El-Hosieny
Hort. Res. Inst. A.R.C. Egypt