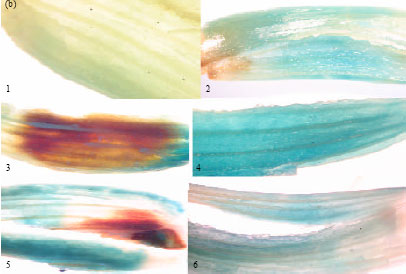
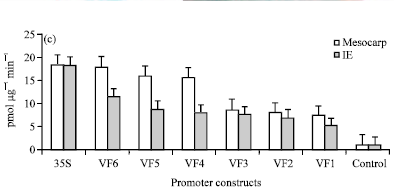
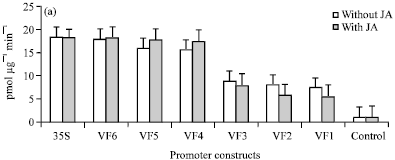
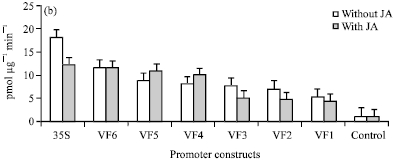
Research Article
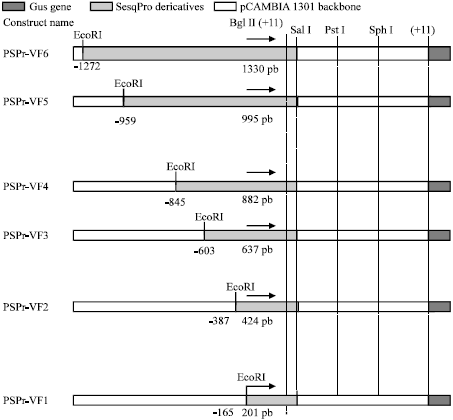
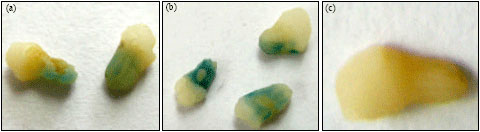
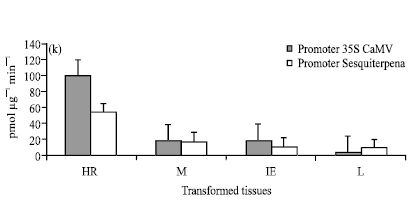
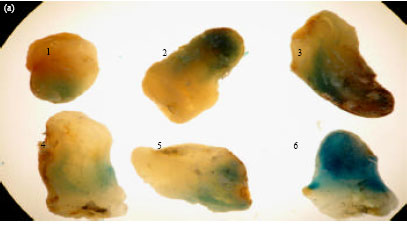
Functional Analysis of the Elaeis oleifera Sesquiterpene SynthasePromoter Reveals Non-Specific Tissue Expression and Activation under Stress Condition
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600, Bangi, Selangor, Malaysia
Norazreen Abdul Rahman
Rubber Research Institute, Malaysia Rubber Board, 4700, Sungai Buloh, Selangor Darul Ehsan, Malaysia
Chan Kok Fei
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600, Bangi, Selangor, Malaysia
Zamri Zainal
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600, Bangi, Selangor, Malaysia
Nik Marzuki Sidik
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600, Bangi, Selangor, Malaysia
Che Radziah Che Mohd Zain
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600, Bangi, Selangor, Malaysia