Research Article
Mineralogical Composition of Limestone Rock and Soil from Jubaila Formation
College of Food and Agricultur, King Saud University, P.O. Box 2460, Riyadh, 11451, Saudi Arabia
LiveDNA: 966.9021
Weathering significantly alters the chemical, mineralogical and physical properties of the rocks and results in different properties. However, the effect of weathering on the geological and geomechanical properties of limestone rock is not well known (Tugrul and Zarif, 2000). Limestone composition is dominantly that CaO in CaCO3 (CO2 content usually not given). The weathering of sedimentary rocks could proceed as predicted by their mineralogy. The Molar SiO2/Al2O3 ratio of limestone is around 9 (Birkeland, 1999). The alterations that would have to occur during soil formation would produce clay minerals from rocks.
Magnesium ion, comparing with other ions, has the greatest ability to prevent the growth of calcite. Moreover, the presence of Mg++ favors to formation of aragonite, a mg-calcite and hydrated carbonate from aqueous solutions at surface conditions (Heakal et al., 2000). The hydrates tend to convert to the anhydrous forms (calcite or aragonite) with time even at low temperatures. In marine environments and some soils, the amount of Mg substitutions can reach 10-15% mole Mg (Mackenzie et al., 1983). The solubility of magnesian calcites generally increases as the percent of Mg substitution increases (Walter and Morse, 1984). The dry transformation of aragonite to calcite under 100°C, needs tens of millions of years (Fyfe and Bischoff, 1965).
Three principal processes to account for genesis of clay minerals were distinguished: (1) inheritance from parent materials, (2) transformation of other clay minerals and (3) neoformation from soil solution (Millot, 1970). For example, there are three possible origins for smectite clay. It may be inherited from shale and hydrothermally altered rocks (Jackson and Sherman, 1953). Moreover, Reid-Soukup and Ulery (2002) noticed that, smectites may form as result of weathering transformations of other 2:1 phyllosilicates, particularly micas. Also, neoformed smectites are those that precipitate directly from soil or matrix solution. In arid and semi-arid regions, palygorskite, smectite, chlorite, illite, kaolinite and vermiculite are the dominant clay minerals (Bouza et al., 2007; Shadfan et al., 1985).
Many researchers believe that soils forming on or in association with limestone are derived from limestone rocks. The clay content which originated from limestone, might be considered as evidence for intensive weathering. Ross and Hendrick (1945) found that kaolinite is an end-product of clay mineral development in limestone soils. Murray and Keller (1993) noticed that most kaolinite results from weathering, by hydrothermal alteration and as an authigenic sedimentary mineral. Also, Keller and Frederiekson (1952) noticed that the little of montmorillonite in the Lenoir limestone may come from slight weathering of the limestone while release of Mg and Ca may allow these ions to take part in the crystal lattice of hydrous mica producing montmorillonite. Moreover, in Jordan, smectite/vermiculite interstratified mineral dominated the clay minerals for limestone-derived soils (Khresat and Taimeh, 1998).
Illite/smectite is a common mixed-layered mineral. It is a mineral formed of illite and smectite. Illite/smectite is the most abundant, varied and widespread of the mixed layered clay minerals in sedimentary rocks and soils (Moore and Reynolds, 1997). White (1950) concluded that the transformation of illite into montmorillonite could occur at both high and low temperatures. Also, it was asserted that this transformation can be produced by MgCl2. In addition, the conversion of illite to mixed-layered I/S is apparently continuous around 150°C and discontinuous above 200°C (Velde, 1985).
MATERIALS AND METHODS
Study area and sampling protocol: The study was undertaken in west of Riyadh city, Saudi Arabia. The study area is located on Jubaila formation at 24.641 N and 46.590 E. It is around 700 m above sea level. According to Al-Malik (1994), the rainy season normally starts in December and ends in May, with a peak that occurs in April. The main annual temperature is 24.7°C, while the main annual rainfall is 83.3 mm. Unweathered Jubaila limestone and soil were collected from the base of the roadcut (about 10-15 m below the surface) (Fig. 1). Samples were two limestone rocks (1 and 2), three unconsolidated rocks (3, 4, 5) and three soil samples (6, 7, 8).
 | |
| Fig. 1: | The location of studied samples showing Cross-section of limestone rocks with strip of soil (Jubaila Formation) |
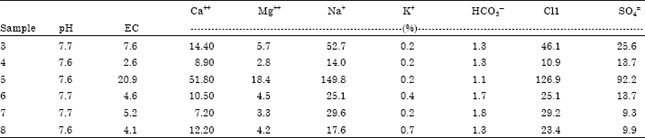
Physical and chemical analysis: Rock and soil samples were air-dried, powdered, homogenized under standard conditions using an agate mortar (<2 mm). Soil pH and EC were measured on 1:1 soil/water mixture according to Thomas (1996) and Rhoades (1996), respectively. Cations and inions (Ca2+, Mg2+, Na+, K+, HCO3¯, CO32¯, Cl¯, SO42¯) were analyzed in those extracted soil solutions (Gupta, 2007).
Mineralogical analyses: XRD was applied for whole sample of limestone rocks, soils and clay fraction of those samples. A part of each sample (Rock and soil) was treated chemically prior to particle size fractionation. Soluble salts and carbonates were removed by using the sodium acetate buffer method (Kunze and Dixon, 1994). Organic matter was removed by using H2O2 (Moore and Reynolds, 1997). Finally, free iron oxides were removed by using dithionite citrate-sodium bicarbonate (Kunze and Dixon, 1994). After chemical treatment, soil suspensions were dispersed by a combination of chemical and physical methods using Na-hexametaphosphate. Subsequently, a 5 min mixing with a standard electrical mixer was performed (Gee and Bauder, 1994).
All clay fractions of samples were oriented by using the glass slide method (Moore and Reynolds, 1997). Samples of clay fractions were saturated by Mg and K. Mg-saturated clay samples were solvated by ethylene glycol vapors (in a dessicator) over a period of 48 h prior. The K-saturated clay samples were studied both after air-drying and heating (for 2 h) at 550°C. Samples were subjected to XRD using CuKα (1.5406 A°) radiation (45 kV, 35 mA) on a Shimadzu (7000) vertical goniometer in a range of 2° 2θ to 40° 2θ. Moreover, powder samples of limestone rock and soil were subjected to XRD using same condition in a range of 2° 2θ to 60° 2θ (Whittig and Allardice, 1994). Mineral identification was determined using the standard parameters using the computer program ICDD for calcite and quartz. While, clay minerals were identified as explained by Moore and Reynolds (1997) and Dixon and Schulze (2002).
Differential Thermal Analysis (DTA) and Thermo Gravimetric Analysis (TGA) were carried out with whole sample fraction of limestone rocks and soils. Precalcined alumina was used as the inert material. Analyses were carried out by DTG 60H with a heating rate of 20°C min-1 from 25 to 1100°C in N. Weight of clay samples were around 25 mg.
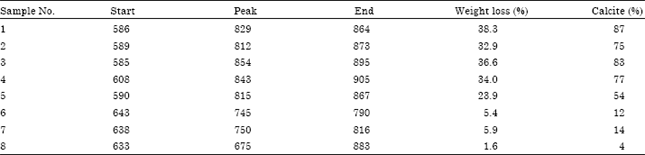
The basic chemical properties of studied soil samples are summarized in Table 1. Figure 2a-c shows the X-ray diffraction patterns of untreated rock and soil samples. Calcite was recognized by its strong peaks 012, 104, 006, 110, 113, 202, 024, 018, 116, 121 and 122. It was a major mineral of limestone rocks and other samples. X-ray diffractogram of the eighth sample (soil sample) indicates absent of most peaks of calcite. Table 2 shows the intensity of all peaks of minerals of samples. The existence of calcite and absence of other calcium carbonates (such as aragonite, Mg-calcite and nesquehonite, etc.) indicates how old is study area (Jubaila Formation). Because of, calcite and dolomite are described as stable phases; while, aragonite, Mg-calcite, nesquehonite, lansfordite and artinite are recognized as metastable (Doner and Grossl, 2002). The transformation of aragonite to calcite needs tens of millions of years (Fyfe and Bischoff, 1965). Previous studies specified age of Jubaila Formation to be of the Upper Jurassic which extends from about 161.2±4 to 145.5±4 Mya (million years ago) (Vaslet et al., 1991).
Moreover, quartz was identified by its characteristic peak of 0.425 nm (100) and 0.335 nm (101). Other peaks of quartz were appeared with soil samples (6-8). A small peak could be noticed at ~0.727 nm among all samples which could indicate kaolinite mineral. In rock limestone (sample 1, 2), the relative peak intensity of 0.727 nm was smaller than other samples; whereas, the highest intensity of that peak was with soil samples (6-8).
| Table 1: | Basic chemical properties of studied soil samples |
 | |
| Table 2: | X-ray powder diffraction data of studied samples |
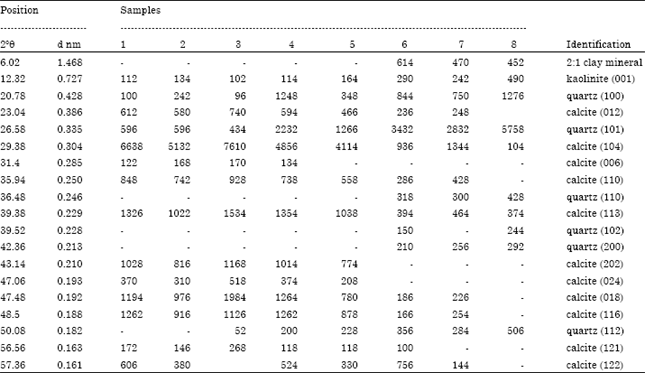 | |
Finally, soil samples have another peak around 1.468 nm, suggesting the presences of 2:1 clay minerals (Borchardt, 2002; Dixon and Schulze, 2002). The small intensity, at ~0.727 and 1.468 nm points to low content of clay minerals among studied samples.
With soil samples (6-8), the DTA signal exhibits an endothermic peak around 100°C which might be due to the removal of adsorbed water on external surfaces for clay minerals. These clay minerals could be smectite and/or illite (Borchardt, 2002) or which called expansive phyllosilicates (Reid-Soukup and Ulery, 2002). On the TGA curves (Fig. 3) a 2.74-4.68% (with average 3.96%) weight loss was determined associated with that endothermic peak. Samples (3-5) gave the smaller intensity of the first peak. Whereas, it was disappeared with limestone rock samples (1, 2) (Fig. 3). The low intensity or absence of the first peak confirms a lower presence of expansive phyllosilicates than in soil samples. The patterns of XRD support this result. Figure 2 shows the small intensity at (1.468 nm) of illite/smectite of soil samples (6-8), while that peak was absence with other samples.
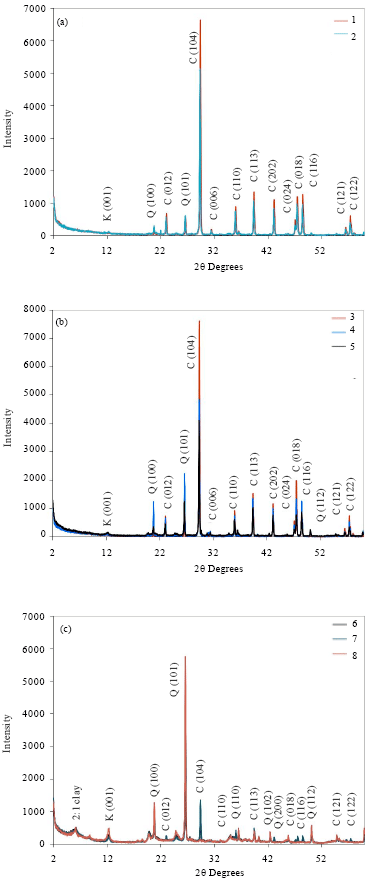 | |
| Fig. 2(a-c): | XRD patterns of whole samples fraction (K: Kaolinite; Q: Quartz; C: Calcite) |
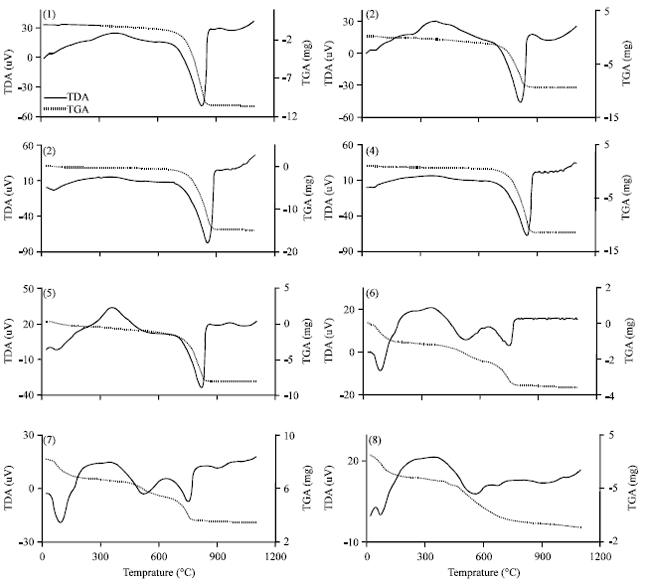 | |
| Fig. 3(1-8): | DTA and TGA curves of whole samples fraction |
An endothermic peak was observed at 520-560°C with soil samples (6-8). This peak is associated with dehydroxylation of kaolinite. This endothermic peak was disappeared with limestone rocks and other samples except for sample (6) which had a small endothermic at 526°C. The dehydroxylation temperatures were ≤560°C. These temperatures are in the upper range of the usually reported temperatures of dehydroxylation of soil kaolins. They are often reported to be around or even below 500°C (Hart et al., 2002, 2003) while those of reference kaolinites often are between 500 and 550°C (Hart et al., 2002). The size of the peak, as well as the peak temperature, is reduced slightly as the particle size decreases and as the crystallinity decreases. The difference seems to be greater for the crystallinity factor than for the particle size (Grim, 1968). The crystallinity of kaolinite has been found to be associated with pedo-environmental factors of soil. For example, the presence of interstratified 2:1 minerals and Fe in kaolinite is considered to be responsible for decreasing kaolinite crystallinity (Singh and Gilkes, 1992).
The influence of pH on structural of kaolinites has been reported by Sei et al. (2006). Thus acid media result in ordered and coarse-grained particles. While, alkaline media cause disordered and fine grained kaolinites.
| Table 3: | The changes in the peak's temperature and weight loss due to the CO3= decomposition of calcite mineral |
 | |
Smykatz-Kloss (1974) recognized the following classification to realize the degree of structural order of kaolinite: extremely disordered kaolinites have (Tendo<530°C), very disordered kaolinites (530°C<Tendo<555°C), less disordered kaolinites (555°C<Tendo<575°C) and well ordered kaolinites (Tendo>575°C). From these suggestions, kaolinite of the clays studied could be described as disordered. The structural disorder is expected to modify the chemical and physical properties of kaolinite. Moreover, the differential thermal analysis is sensitive to shape of kaolinite particles. Spherical particles have lower dehydroxylation temperatures than hexagonal particles (Huertas et al., 1997). The ideal form for the well crystallized kaolinite particles is hexagonal. The exothermic peak of kaolinite (≈1000°C) did not appear in this study. This could be explained by the presence of even a small amount of iron oxide or hydroxide which are suggested to modify the temperature exothermic.
Finally, other endothermic peak was observed between 668-854°C. This peak could be explained by the presence of calcite (Paterson and Swaffield, 1987). The DTA diagram shows systematic decreases in the peak area of the calcite decomposition with the increases of weathering of limestone rock (Fig. 3). This endothermic peak signified the thermal decomposition of the CO3= present in the Ca-bearing minerals (Calcite) of each sample. Table 3 illustrates the change in the peak temperature, the weight loss and the calculation of calcite content in samples. In general, the order of calcite content follows the same order of intensity of peak (104).
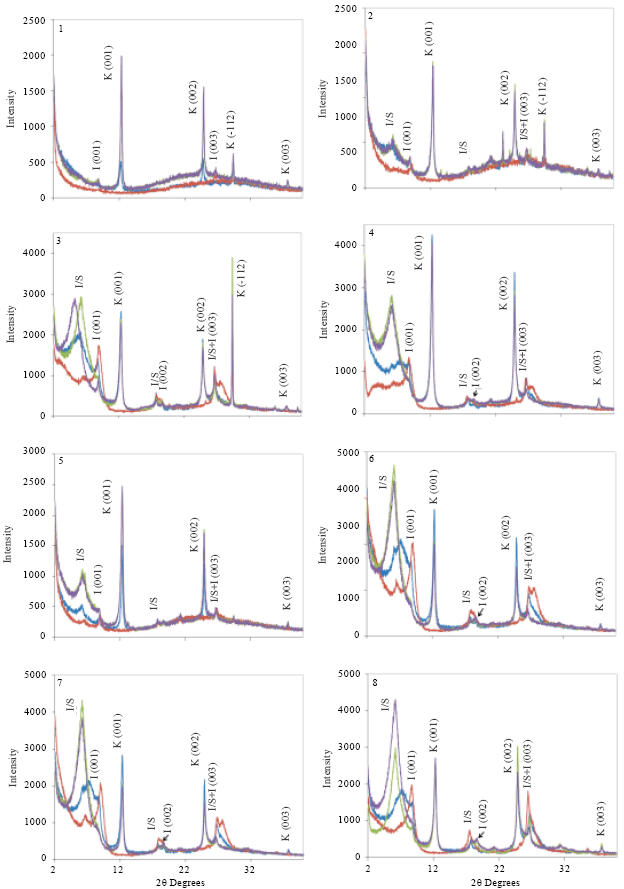
Clay fraction: As shown in Fig. 4, clay fraction of all samples had reflections at around 0.72 nm (001) and 0.358 nm (002) was not affected by solvation but vanished after heating. In view of these behaviors, the dominant clay mineral of the limestone rocks and soil samples is kaolinite. The d (001) values for kaolinite in the clay fraction varied from 0.718 to 0.728 nm with a median value of 0.721 nm. The range for the (002) d-values was narrow (0.358-0.359 nm) compared with (001) d-values. The intensity ratio of 001/002 ranged from 0.73-0.89, except with sample 1 which had a higher intensity of 002 comparing with 001 reflection. According to the intensity of first peak (001), the limestone rock samples (1 and 2) have the lowest content of kaolinite.
Moreover, there are reflections at around 1.024 nm (001), 0.502 nm (002) and 0.335 nm (003). Those reflections didn’t affected by other treatments which suggested presence of illite mineral. Sample 1 which is one of limestone rocks, has just the first reflection of illite. Moreover, the intensity of first peak (001) for both limestone rocks was small comparing with other samples. The low intensity indicates a low amount of illite minerals in limestone rocks. Thompson and Ukrainczyk (2002) reported formation of illite in pedogenic and sedimentary environments.
Figure 4 shows that, the glycol-treated gives a strong reflection at about 5.24° 2θ (1.687 nm) which, in the air-dried condition, shifts to about 6.14° 2θ (1.44 nm). Moreover, heating treatment at 550°C resulted in a diffraction pattern similar to that of illite. Because of EG salvation has caused significant changes in the diffraction pattern of sample 3, smectite component should be presented.
 | |
| Fig. 4(1-8): | XRD analysis of clay fraction of all samples |
Furthermore, a reflection is noted at 1.768° 2θ, therefore, the diagnosis is likely illite/smectite (Moore and Reynolds, 1997). This suggestion was supported by absence of the (003) and (005) peaks. With exception of the first, other samples have the same reflections around 1.43-1.47 nm with which don't shift with EG treatments (Fig. 4). Smectite lost its ability to expand in the presence of ethylene glycol, as it becomes more deeply buried by overlying sediments (Thompson and Ukrainczyk, 2002). From above, all samples, except the first, contain illite/smectite mineral. Some researchers found that a part of smectite has been generated in an Mg and Si rich basin by the transformation of the illite in a sedimentary environment (Birkeland, 1999). Moreover, Bernhard et al. (1999) suggested that in an Si rich environment with moderate or low leaching combined with low drainage, I/S may occur.
X-ray diffraction and DTA with DTG indicated that, calcite and quartz are the major minerals in all analyzed rock and soil samples. Moreover, low amount of kaolinite was found in all samples; while 2:1 was noticed with soil samples only. Kaolinite was the dominant mineral in clay fraction of all samples. Furthermore, illite and illite/smectite were found with clay fraction. The content of illite and illite/smectite was more with soil samples comparison with other samples. The existence of calcite and absence of other calcium carbonates (such as aragonite, Mg-calcite and nesquehonite, etc.) indicates how old is study area (Jubaila Formation). Transformation of Mg-calcite to calcite, supply Mg ion which is necessary of smectite generated by transformation of illite. Because of, soil’s strap was stuck between limestone rocks made low leaching and drainage environment which supported generation of illite/smectite mineral.
The author wishes to thank Prof. Abdulazeem Salam, Khalid Al-Enazi and Saaed Solih, Soil Science Department, King Saud Univ., Riyadh, Saudi Arabia, for their assistance and efforts during laboratory work.