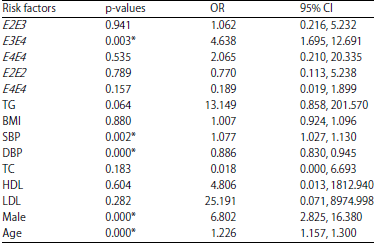
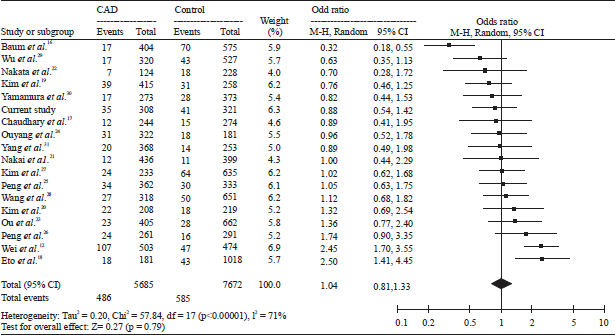
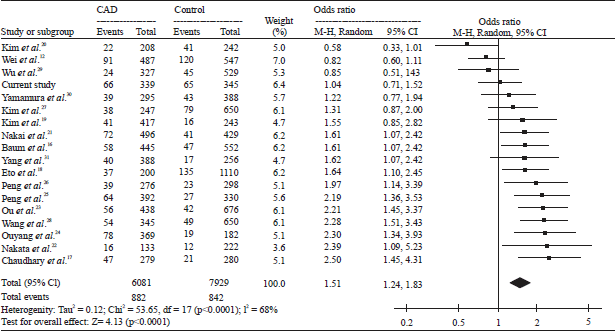
Research Article
Relation Between apoE Gene Polymorphism and Coronary Artery Disease in South East and East Asian Countries
Department of Basic Medical Sciences for Nursing, Kulliyyah of Nursing, International Islamic University Malaysia, Kuantan, Pahang, Malaysia
LiveDNA: 964.16203
Farrah Wahida aladzemi
Department of Basic Medical Sciences, Kulliyyah of Medicine, International Islamic University Malaysia, Kuantan, Pahang, Malaysia
Nurul Ashikin Muhammad Musa
Department of Basic Medical Sciences, Kulliyyah of Medicine, International Islamic University Malaysia, Kuantan, Pahang, Malaysia
Nik Nur Fatnoon Nik Ahmad
Department of Internal Medicine, Kulliyyah of Medicine, International Islamic University Malaysia, Kuantan, Pahang, Malaysia
Nor Zamzila Binti Abdullah
Department of Pathology and Laboratory Medicine, Kulliyyah of Medicine, International Islamic University Malaysia, Kuantan, Pahang, Malaysia
Norlelawati Binti Talib
Department of Pathology and Laboratory Medicine, Kulliyyah of Medicine, International Islamic University Malaysia, Kuantan, Pahang, Malaysia