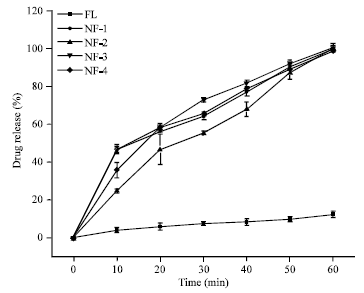
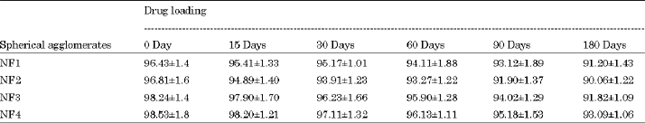
Research Article
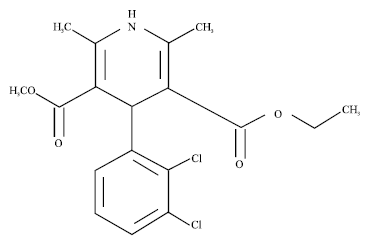
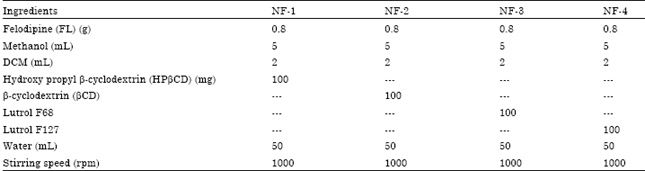
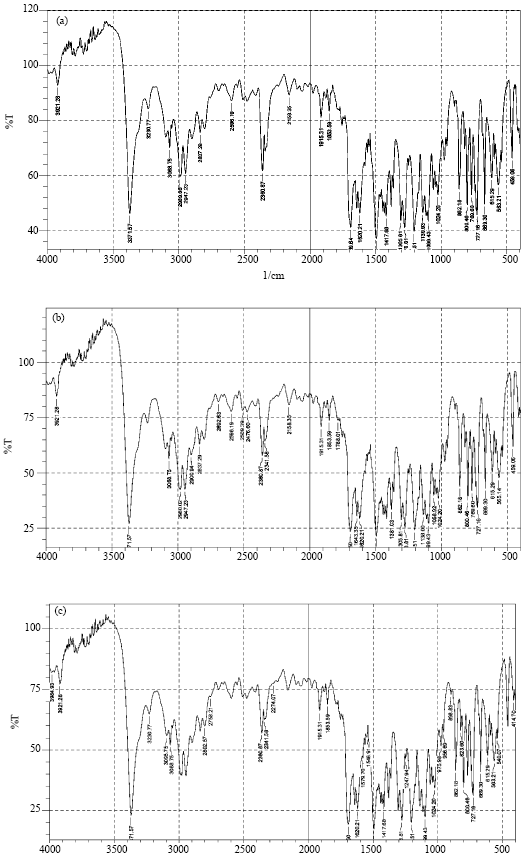
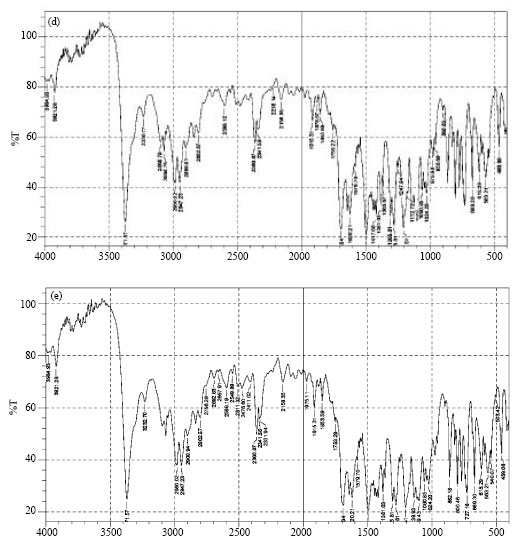
Modification of Felodipine Properties using Spherically Agglomerated Solid Dispersions
Department of Pharmaceutical Chemistry, Sudhakarrao Naik Institute of Pharmacy, Pusad-445204, Dist: Yavatmal, Maharashtra, India
LiveDNA: 91.6535
P.S. Kawtikwar
Shri Sureshdada Jain Institute of Pharmaceutical Education and Research, Jamner-424206, Dist: Jalgaon, Maharashtra, India
D.M. Sakarkar
Department of Pharmaceutical Chemistry, Sudhakarrao Naik Institute of Pharmacy, Pusad-445204, Dist: Yavatmal, Maharashtra, India