Research Article
Embryotoxicity and Teratogenicity of Enrofloxacin on Maternally Treated Chick
Department of Biology, Faculty of Science, Taif University, Taif, Kingdom Saudi Arabia
In commercial poultry farms the bacterial infection among laying chicken is common and usually needs rapid medication to avoid morbidity and mortality of the infected specimens. Therefore, huge quantities of antibiotics are used annually in poultry farming throughout the world, but the eventual fate of their residues and their potential damage to human health generally remains unknown. Enrofloxacin (1-Cyclopropyl-7-(4-ethyl-1-piperazinyl)-6-Fluoro-1, 4-Dihydro-4-Oxo-3-Quinolonecarboxylic Acid) is a synthetic, broad spectrum antimicrobial medication used in poultry medicine, belonging to the fluoroquinolone group of antibiotics (Wolfson and Hopper, 1989). Several studies showed that enrofloxacin (Baytril 10%) oral solution was indicated for use in ducks, turkeys, broiler chickens, broiler breeders and pullets being reared as layers for diseases of the respiratory and alimentary tract of bacterial or mycoplasmal origin (Hafez et al., 1990; Kempf et al., 1995; Glisson, 1996; Barrow et al., 1998; McDermott et al., 2002; Randall et al., 2005; Luke et al., 2006; Garmyn et al., 2009a, b). In many countries it is being used as the routine choice to avoid infection and to treat almost any bacterial disease in poultry. Enrofloxacin inhibits cell proliferation, induces apoptosis and DNA fragmentation of canine tendon cells and chondrocytes in a dose and time-dependent manner (Lim et al., 2008). It also causes disruption of spermatogenesis in the testes of mice in the form of reduction of sperms motility and count as well as morphological abnormalities (Aral et al., 2007). It is well documented that enrofloxacin and its metabolite ciprofloxacin as well as other fluoroquinolones have a tendency to accumulate as residues in avian eggs and other edible tissues (Gorla et al., 1997; McReynolds et al., 2000; Yorke and Froc, 2000; Chu et al., 2002; Shim et al., 2003; Christodoulou et al., 2007; Cho et al., 2008; Zhao et al., 2009; Frenich et al., 2010). Embryo lethality and teratogenicity of fluoroquinolone antibacterials in rats and rabbits were previously suggested (Guzman et al., 2003; Kim et al., 2000, 2003-5). Also, reduction of scavenger wildlife populations and fatal embryo chondral damage associated with enrofloxacin and its metabolite ciprofloxacin in eggs of threatened avian scavengers were investigated by Lemus et al. (2008, 2009). However, in the literatures scanned, no study was found concerning the developmental toxicity and teratogenicity of enrofloxacin on the developing chick embryo. Therefore, the present study was undertaken to investigate the possible embryotoxicity and teratogenicity of enrofloxacin on embryonic days (EDs) 6½ and 13 of the developing chick of fertile eggs collected at the first, seventh and tenth days after the last day of chicken treatment, respectively. Accordingly, the results may help to find out an explanation of the low rate (≈53%) of fertile chicken eggs hatchability (following chickens treatment, against infectious diseases, with enrofloxacin) in a new commercial poultry farm in Al-Taif, KSA.
The Enrofloxacin
Enrofloxacin (Baytril 10% oral solution) for dosing poultry was obtained from the National Veterinary Services Al-Taif, KSA. It is a clear aqueous oral solution containing as active ingredient 100 mg mL-1 enrofloxacin and 14 mg mL-1 benzyl alcohol as a preservative. In the poultry farm the enrofloxacin was added to the chicken’s drinking water (under the supervision of a licensed veterinarian) at a dose adjusted to give 10 mg kg-1 b.wt. of birds per day or equivalence, i.e., water at 50 ppm, continuously medicated water) for seven consecutive days.
Egg Incubation
The present study was carried out in the laboratory of Zoological Research, Biology Department, Faculty of Science, Taif University, KSA. This research was conducted from November-2009 to February-2010. A total of six hundred fertile white leghorn chicken eggs, each weighing about 70 g, were generously provided (in four successive groups, each 150 eggs) by the commercial poultry farm of Al-Taif, KSA. The first (control) group was obtained from chickens that did not receive any antibacterial medication; while the second, third and fourth ones were collected at the first, seventh and tenth days after the last day of chicken treatment with enrofloxacin, respectively.
Before incubation each egg group was stored in a refrigerator at 4°C for 24 h to allow egg contents to return to steady state after transport. The eggs were then transferred and maintained at 37.5°C in a full automatic egg incubator with full automatic control of humidity (relative 55-60%), egg turning, fan speed, ventilation and alarm until the desired stages of chick development (6½ and 13 day old embryos) were reached.
Experimental Protocol
To estimate the viability, fresh weights, crown-rump length, anterior-posterior head length, external malformations as well as skeletal anomalies in control and enrofloxacin-maternally treated chick embryos, each of the four egg groups was divided into six (25 eggs each) subgroups [3 replicates for each day of investigation (EDs 6½ and 13)]. In the replica the ratios of living and dead embryos were recorded, the living embryos were wet weighed and examined for the presence of external malformations (of the head, trunk, limbs and tail) under a dissecting microscope. On both EDs the crown-rump length and anterior-posterior head length were measured with a caliper.
Histological Procedures for Skeletal Staining
On ED 13, embryos were processed for staining with a whole mount double cartilage and bone staining technique previously described by Lamb et al. (2003) with some modifications. Affected and control embryos were removed from their extraembryonic membranes, washed in saline, injected under skin and within viscera with 95% ethanol, immersed for one hour in ethanol then they were skinned, eviscerated and fixed in pure ethanol for 24 h. The embryos were then stained overnight at room temperature in Alcian blue [0.01 g Alcian blue 8GX (SIGMA) was added to equal volumes of 75% ethanol and 25% glacial acetic acid] for 1 day. The skeletons were then stained with 0.015% alizarin red S in 0.5% KOH for 48 h, cleaned in the following ratios of 0.5% KOH in distilled water to glycerol: 80:20, 60:40, 40:60, 20:80 and finally were stored in pure glycerol.
Incidence of External and Skeletal Malformations
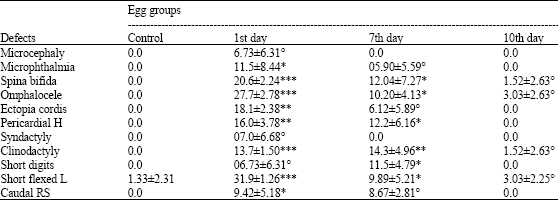
The abnormalities of the central nervous system were represented by neural tube defects in the form of spina bifida (a defect that involves the incomplete development of the spinal cord or its coverings), microcephaly (disproportionately small head) and delay of brain development. Microphthalmia was defined as a reduced size of the eye, ranging from its absence to about one half of its normal size. Oedema is an abnormal accumulation of fluid beneath the skin. Ectopia cordis (heart displaced outside the thoracic cavity). Omphalocele is characterized by the absence of abdominal muscles and skin; and the abdominal wall covering is replaced by a membrane. This body wall closure defect exists in the abdomen or in both thoracic and abdominal regions. Pericardial hernia is a malformation in which the viscera and the liver were seen above the heart or lungs. Limb deformities: syndactyly (partial or complete fusion or webbing between digits), clinodactyly (deflection of digits from the central axis), flexed limbs (ranging from simple flexion to excessive flexion or bending of a complete limb or a joint) and short limbs or digits. Caudal Regression Syndrome (CRS) was diagnosed when the caudal part of the embryo (including hind-limbs) or the tail was reduced in size or the whole tail was lacking. The stained axial and appendicular skeletons were examined for any changes (absence, irregular shapes, axis deviations, lack or reduction of cartilage and bone formation).
The embryotoxic effect (the proportion of living malformed and dead embryos) of enrofloxacin-maternally treated chick embryos was dependent on the time elapsed after the last treatment and day of embryonic investigation. The study did not exclude spontaneously dead or spontaneously malformed embryos from the evaluation, to obtain the total frequency of all affected embryos.
Statistical Analysis
Percentages of mortality, abnormal yolk, malformed embryos and skeletal element changes of the maternally treated groups were compared with the control by one-tailed student’s t-test. The fresh body weight, crow-rump and anterior-posterior head lengths data were expressed as group means±SE. Then a one-way parametric ANOVA was used to compare these parameters in each egg group with the control. Both tests were carried out using Microsoft Office Excel (Frye, 2003).
Mortality Rate
The mortality percentages of embryos from eggs collected at the first, seventh and tenth days after the last day of chicken treatment with enrofloxacin, respectively, were listed in Table 1. On ED 6½ the mortality rates recorded for embryos of eggs collected at the first and seventh days were significantly increased (p<.001 and p<0.05, versus control), while on ED 13 relatively higher incidences (p<0.001 and p<0.0001, versus control) were estimated. However, the mortality rates recorded (on both EDs) for embryos of eggs collected at the tenth day were insignificantly (p>0.05, versus control) increased.
I- 6½-Day-Old Chick Embryos Control
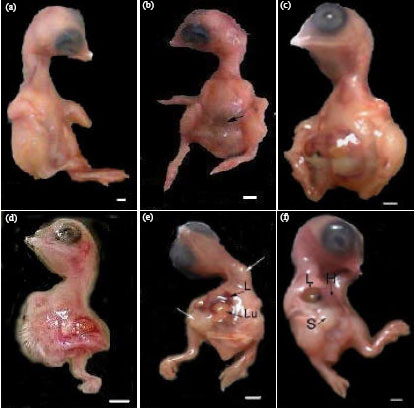
In eggs opened on ED 6½ the embryos were found lying within the amnion and above normally appeared yolk sac. They were provided with numerous normally branched blood vessels of the vitelline circulation that were extended from the embryonic heart and reached the outer extremities of the yolk, while other vessels were returned back (Fig. 1a). By this stage of development the embryonic eyes and the three brain regions [forebrain (prosencephalon), midbrain (mesencephalon) and hindbrain (rhombencephalon)] showed normal development, while the beak was slightly developed. The cervical region was a little curved, while the cranial region made an angle of about 90° on the body axis. The heart was completely enclosed within the thoracic cavity. The fore- and hind-limb parts were still in the original paddle-shaped appendage buds with no detectable digits (Fig. 2a-d). There was one (1.33%) spontaneously malformed embryo exhibited ectopia cordis and short flexed limbs (Table 2). The means of fresh body weights, crown-rump and anterior-posterior head lengths recorded for such 6½-day-old control chick embryos were 0.64±0.01 g, 1.99±0.03 and 1.08±0.03 cm, respectively (Table 3).
| Table 1: | Percentages of mortality, abnormal yolk and developmental defects in living 6 ½ and 13-day-old chick embryos |
 | |
| 1st, 7th and 10th days are embryos of eggs collected on the first, seventh and tenth days after the last day of chicken treatment with enrofloxacin, respectively. The data are Means±SD°p>0.05, *p<0.05, **p<0.001, ***p<0.0001 (one tailed student’s t-test) compared to controls | |
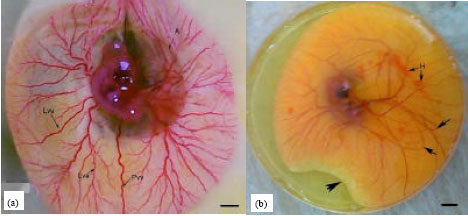 | |
| Fig. 1: | (a, b): Photographs of 6½-day-old chick embryos on yolk (a) A control embryo enriched with normal vitelline circulation and (b) An embryo of an egg obtained at the first day of following maternal treatment with enrofloxacin displaying inappropriate yolk formation (arrow head), poorly developed and abnormally oriented vitelline vessels (arrows). A, amnion; Pvv, posterior vitelline vein; Lva, lateral vitelline artery; Lvv: Lateral vitelline vein; H: Hemorrhage. Scale bar = 1 mm |
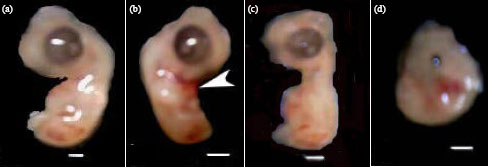 | |
| Fig. 2: | Photographs of 6½-day-old chick embryos, (a) A control embryo, (b) An embryo of an egg obtained at the 7th day of the last day of maternal treatment with enrofloxacin exhibited body wall closure defect (arrow head), (c) and (d) embryos of eggs collected at the 1st day of the last day of maternal treatment with enrofloxacin, (c) Embryo with abnormally oriented neck region and caudal regression syndrome and (d) An embryo with C-shaped body of reduced size and microphthalmia. Scale bar = 5 mm |
| Table 2: | Percentages of particular structural anomalies in living 6½ -day-old chick embryos |
 | |
| The data are Means±SD °p>0.05, *p<0.05, **p<0.001, ***p<0.0001 (one tailed student’s t-test) compared to controls. Abbreviations: H: Hernia, L: Limb, RS: Regression syndrome. Each embryo displayed 1-3 types of the structural anomalies | |
| Table 3: | Mean body weight (in grams), crown-rump and anterior-posterior head lengths (in centimetres) of 6½ and 13 day-old chick embryos |
 | |
| The data are Mean±SE °p>0.05; *p<0.05 (One-Way ANOVA) compared to controls | |
Enrofloxacin-Maternally Treated
The 45.3 and 33.3% of eggs collected at the first and seventh days after chicken treatment with enrofloxacin, respectively and opened on ED6½ showed weakly ingested yolk; yolk within the yolk sac exhibited incomplete uneven appearance and poorly developed vitelline circulation as well as extravasations of blood and abnormally oriented blood vessels (Fig. 1b, Table 1). The prevalent types of structural anomalies observed in 6½-day-old embryos of eggs collected at the first and seventh days of chicken treatment with enrofloxacin were significantly increased and included spina bifida, body wall closure defects, ectopia cordis, caudal regression syndrome and short flexed limbs (Table 2). Conversely, the structural anomalies observed in embryos of eggs collected at the tenth day following chicken treatment were infrequent. The mean of body weights, crown-rump and anterior-posterior head lengths recorded for embryos obtained from eggs collected at the first and seventh days of chicken treatment were significantly decreased (p<0.05, versus controls). However, the mean of these parameters was insignificantly decreased (p>0.05, versus controls) when the embryos were of eggs collected at the tenth day following chicken treatment (Table 3).
II-13-Day-Old Chick Embryos Control
By this stage of chick development, the definitely avian features became very pronounced than in the previous stage. Feathers and feather tracts were visible on different body parts, the beaks enlarged and became hard with scales covered the tip of the upper ones. The nostril was a narrow slit. There was relatively large size of the eyes and midbrain and the auditory meatus was observed with no ear pinna. Both eyelids were well developed, where they met each others when the eye was closed. The limbs became easily distinguished into wings and legs with longer distal segments of limbs (metacarpus and metatarsus, respectively). The wing of the embryo showed the normal wing parts of the hen (humerus, radius and ulna, first digit, metacarpus, second and third digits). The leg was covered with distinct scales and the digits made of distinct phalanges ended by claws. Each leg consisted of femur, tibia and fibula, metatarsus and four digits (Fig. 3a, 4a). There was one (1.33±2.31%) spontaneously malformed embryo exhibited short flexed limbs (Table 4). The means of fresh body weights, crown-rump and anterior-posterior head lengths recorded for 13-day-old control chick embryos were 7.64±0.43 g, 6.96±0.25 cm, 1.81±0.2 cm, respectively (Table 3).
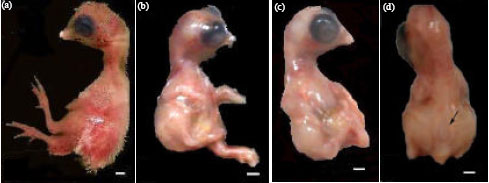 | |
| Fig. 3: | (a-d): Photographs of 13-day-old chick embryos. (a) A control embryo (b-d) Embryos of eggs obtained at the 7th day of the last day of maternal treatment with enrofloxacin, (b) with body edema, abnormally developed legs and reduced tail, (c) Clumped legs and caudal regression syndrome and (d) Delay of feather formation, body edema and spina bifida (arrow). Scale bar = 1 mm |
 | |
| Fig. 4: | (a-f): Photographs of 13-day-old chick embryos to show the ventral body wall (a) A control embryo (b-f) Embryos of eggs obtained at the 1st day of the last day of maternal treatment with enrofloxacin, (b) with poorly developed abdominal wall (arrow), (c) The abdominal contents, including liver, stomach and gut, extrude ventrally within enlarged membranous sac, clumped legs and caudal regression syndrome, (d) Omphalocele and shortened malformed left limb, (e) Omphalocele with pericardial hernia, edema (arrows) and malformed right limb and (f) Omphalocele with pericardial hernia and head, body edema. L: Liver, Lu: Lung, H: Heart, S: Stomach. Scale bar = 1 mm |
| Table 4: | Percentages of particular structural anomalies in living13-day-old chick embryos |
 | |
| The data are Mean±SD °p>0.05, *p<0.05, **p<0.001, ***p<0.0001 (one-tailed student’s t-test) compared to controls. Each embryo displayed 1-3 types of the structural anomalies | |
Enrofloxacin-Maternally Treated
Embryos of eggs collected at the first and seventh days after maternal treatment with enrofloxacin and examined on ED 13 were found to exhibit a general delay of feather development (Fig, 3b, c), highly significant (p<0.0001, versus control) increase in the percentages (45.5, 37.8%) of structural anomalies, respectively; while viability along with insignificantly low percentage (2.78%) of structural anomalies were noted in embryos of eggs collected at the day ten of chicken treatment (Table 1). In the first case, the most prevalent types of anomalies were represented by microphthalmia, spina bifida, omphalocele, pericardial hernia, clinodactyly, short and flexed limbs (Fig. 3b, c). The omphalocele anomaly (the embryonic body wall closure defect) was of two types. In the first one the abdominal wall was completely absent and this condition was usually associated with spina bifida formation, in the form of incomplete covering of the spinal cord in the lumbar area (Fig. 3d and 4c, d). In the second type of omphalocele no sign of the embryonic thoracic and abdominal walls was detected and all viscera were enclosed within a covering membrane. Among these embryos, two exhibited displacement of the heart (pericardial hernia) where in one of them the liver was seen in the left side above the heart and lungs (Fig. 4e) and in the other one the liver was seen in the right side above the heart (Fig. 4f). In these embryos, limb anomalies ranging from mild to severe types were encountered. These included variable degrees of shortness and flexion of limbs, joints and digits (Table 4, Fig. 4c-f). Oedemas formation in the head and/or the whole body parts were also observed in some embryos (Fig. 3b-d, 4c, f). However, the incidence of microcephaly and CRS was infrequent.
III-Skeletal Elements and Ossification Events in 13-day old Control and Enrofloxacin-Maternally Treated Embryos
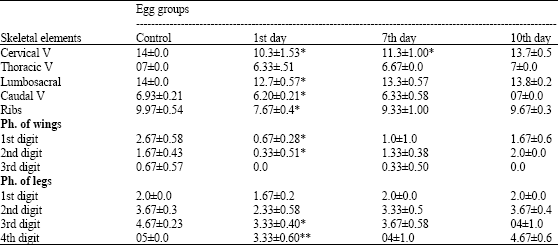
The skeletal elements that showed ossification in the skull and lower jaw of control 13-day-old chick embryo involved the external parts of premaxilla, maxilla, lacrymal, frontal, supraocciptal, exoccipitals, basioccipital, squamosal, quadrate, pterygoid, inferior temporal arch, dentary, angular and articular. However, intense blue staining was observed in the cartilaginous parts (nasal, the inner parts of premaxilla and maxilla, interorbital septum and parietal) of the cranium (Fig. 5a, b, 6a-f). The cervical vertebrae were 14 in number, bone formation was observed in the centra of the vertebrae 3-12, while the rest were fully cartilaginous. The distal parts of the transverse processes of the vertebrae 3-12 showed initial bone formation. The centra of the thoracic vertebrae were partially ossified together with parts of the skeleton of the thorax and lumbosacral, while the joints; free caudal vertebrae and pygostyle were fully cartilaginous.
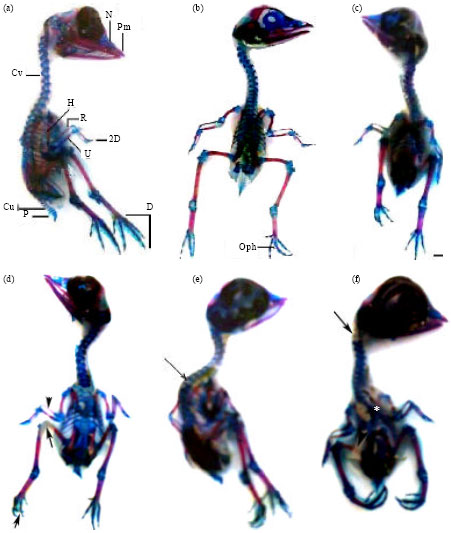 | |
| Fig. 5: | (a-f): Photographs of 13-day-old chick embryos showing skeletal system. (a, b) lateral, dorsal views of controls, c and d-f embryos of eggs collected at 10th and 1st days following chicken treatment with enrofloxacin, respectively, (c) Normally appeared skeleton, (d) Delay of cartilage formation of joints (long arrow), incomplete ossification of radius (arrow head) and digits (short arrow), (e) Abnormally curved thoracic region (arrow) and (f) Delay of cartilage formation of the first two cervical vertebrae (arrow), abnormally short legs and reduced bone of femur (arrow head), scapulae and lumbosacral region (*). Pm: Premaxilla, N: Nasal, Cv: Cervical vertebrae, H: Humerus, R: Radius, U: Ulna, 2D: second digit, Cu: Free caudal vertebrae, P: Pygostyle, D: Digits. Alizarin red S and Alcian blue staining. Scale bar = 1 mm |
Ossification was also observed in the vertebral portions of ribs, scapulae, humerii, medial parts of radii and ulnae, metacarpals of the fore limb; ilium, the medial parts of the femur, tibia and fibula, metatarsals and phalanges of the hind limb (Table 5, Fig. 5a, b).
Embryos of eggs collected at the first day after maternal treatment with enrofloxacin and examined on ED 13 were found to exhibit delay and wide variations in the processes of chondrogenesis and ossification in most of their skeletal parts. Skeletal abnormalities included lack of cartilage formation which appeared as reduced number of cervical, thoracic, lumbosacral and free caudal vertebrae; delayed chondrogenesis of the cervical transverse processes and joints; axis deviations of hind-limbs and their digits; absent/hemicentric body of thoracic or lumbosacral vertebrae; fused vertebrae; delayed ossification of skull bones (parietal, squamosal and inferior temporal arch); and shortness of ossified portions of wing and hind limb parts (Table 5, Fig. 5c-f and 6b-d).
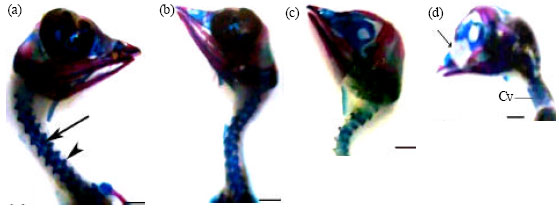 | |
| Fig. 6: | (a-d): Photographs of 13-day-old chick embryos showing skulls and cervical vertebrae. (a) A control skull with various ossified and cartilaginous elements; the cervical vertebrae with ossified centra (arrow) and transverse processes (arrow head), (b-d) skulls of embryos of eggs collected at 10th and 1st days following chicken treatment with enrofloxacin, respectively, (b) reduced in size but normally appeared skull and cervical vertebrae, (c) normally appeared skull with shortened cervical vertebrae and (d) Lack of skull cartilage and bone formation (arrow) and fused cervical vertebrae. Scale bar = 1 mm |
| Table 5: | Mean number of vertebrae, ribs and phalanges of 13-day-old chick embryos |
 | |
| The data are Mean±SD *p<0.05, **p<0.001 (one-tailed student’s t-test) compared to controls. V: Vertebrae, Ph: Phalanges | |
Alternatively, these skeletal abnormalities were of insignificant (p>0.05) incidence as compared to controls; when eggs of the embryos were collected at the seventh and tenth days of chicken treatment with enrofloxacin, except the lack of cartilage formation of the cervical vertebrae in embryos of the seventh day collected egg groups (Table 5).
Detection of enrofloxacin and its metabolite ciprofloxacin residues in avian eggs and edible tissues was proved (Gorla et al., 1997; McReynolds et al., 2000; Shim et al., 2003; Christodoulou et al., 2007; Cho et al., 2008; Lemus et al., 2008, 2009). This property led to the speculation that enrofloxacin residue in fertile eggs of treated chicken may result in abnormal development of their embryos. This speculation is confirmed by the results of the present study. These results revealed new findings (were not previously recorded following treatment with any fluoroquinolone antibacterials) represented by statistically significant inappropriate yolk sac formation on ED 6½ and omphalocele on both EDs 6 ½ and 13, in embryos of eggs collected at the first and seventh days following chicken treatment with enrofloxacin. Furthermore, enrofloxacin has caused highly significant incidence of mortality rates and reductions in the mean of fresh body weights, crown-rump and anterior-posterior head lengths of the maternally-treated embryos. These findings are in accordance with those of Lemus et al. (2009) who suggested that the enrofloxacin and its metabolite ciprofloxacin resulted in reduced breeding success of two threatened avian scavengers. Similarly, Kim et al. (2003) declared a concentration-dependant decrease of rat litter size, fetal weight and placental weight and severe increases in resorption rate and fetal morphological alterations following maternal treatment with the fluoroquinolone antibacterial DW-116. The reduction of embryonic body weight of the present study could be explained by the obtained higher incidences of inappropriate yolk formation that may cause a decrease in residual yolk composition which is the main energy source for the developing embryo. Speake et al. (1998) stated that yolk supplies more than 90% of the total energy requirements of the embryo by oxidation of yolk lipids.
The most prevalent and severe types of anomalies encountered on ED 13 (of eggs collected at the first and seventh days following chicken treatment with enrofloxacin) following maternal treatment with enrofloxacin were represented by spina bifida, omphalocele, pericardial hernia and limb defects. The association of omphalocele, observed in the present study, with spina bifida and skeletal defects are in accordance with Weber et al. (2002) and Ledbetter (2006) who indicated that omphalocele frequently occurs in conjunction with other abnormalities, including cardiac or genitourinary abnormalities, neural tube or skeletal defects, as well as chromosomal anomalies, such as trisomy 13 and 18. Besides, in the trunk region, the absence of AP-2 disrupts the ventral body wall formation, resulting in thoracoabdominoschisi as suggested by Ledbetter (2006). Likewise, mutation of the BMP-1 or hoxb genes also can cause abnormal body wall closure and associated sternal defects (Ramirez-Solis et al., 1993; Suzuki et al., 1996). Accordingly, the presently obtained spina bifida; omphalocele and pericardial hernia could be a consequence of gene mutation induced by enrofloxacin treatment due to the fact that it is a potent inhibitor of cell proliferation, induces apoptosis and DNA fragmentation (Yoon et al., 2004; Lim et al., 2008). The present predominance embryotoxic and teratogenic effects of enrofloxacin on embryos of eggs collected at the first and seventh days (and their lack on day 10) following chicken treatment suggests that enrofloxacin withdrawal is longer than one weak. This assumption is in agreement with San et al. (2007) who calculated a withdrawal time of 6-9 days of enrofloxacin plus ciprofloxacin in samples of edible tissues and feathers of white leghorn hens.
In the present study 13-day old embryos of eggs collected at the first day after maternal treatment with enrofloxacin were found to exhibit delay and wide variations in the processes of chondrogenesis and ossification in vertebrae, ribs, joints and other skeletal parts. The skeletal changes described here are quite similar to the observations described previously by some other investigators (Kim et al., 2000, 2003-2005) following administration of another fluoroquinolone antibacterial (DW-116) to pregnant rats and rabbits, respectively. This result is also in accordance with the findings of Lemus et al. (2009) that enrofloxacin and ciprofloxacin were clearly associated with severe alterations in the development of embryo cartilage and bones. Conversely, Maslanka and Jaroszewski (2009) indicated that treatment with a therapeutic dose of enrofloxacin for a period exceeding the recommended duration of therapy does not cause chondrotoxicity in growing chickens. Also, only very high dosage of enrofloxacin, significantly exceeding the therapeutically applied doses, can induce toxic effects in articular cartilage in 21-day-old male broiler chickens and intensity of chondrotoxicity was dose- and time-dependent (Maslanka et al., 2009).
Based on the results presented here, it may be concluded that enrofloxacin is a highly embryotoxic and teratogenic compound that resulted in inappropriate yolk formation, high rates of mortality and significant reductions in the mean of wet body weights, crown-rump and anterior-posterior head lengths and different types of structural anomalies, as well as delay of chondrogenesis and ossification of the maternally treated chick embryos. Nevertheless, it is reasonable to assume that these teratogenic effects were a consequence of chicken treatment with enrofluxacin as they were not seen in control embryos and probably due to the fact that quinolones tend to accumulate in chicken eggs and other edible tissues. It remains necessary to indicate that, continuous exposure to antibiotics could increase mortality rates, at least in newly hatching chick.
The author greatly appreciates the facilities provided by the Department of Biology, Faculty of Science, Taif University, KSA. Also, the author acknowledges the precious help of Prof. Dr. Bedir, M. A. Prof. of animal ecology, Zoology Dept., Faculty of Science, Suez Canal University, Egypt due to final revision of the manuscript.