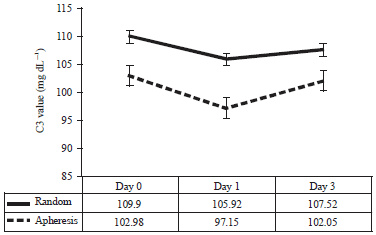
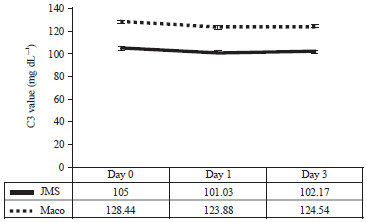
Research Article
Effect of Preparation and Storage Methods of Platelet Products on Complement Activity
Blood Transfusion Research Center, High Institute for Research and Education in Transfusion Medicine, Tehran, Iran
Javad Sajedianfard
Department of Physiology, School of Veterinary Medicine, Shiraz University, Shiraz, Iran
Mohammad Ali Jalali Far
Health Research Institute, Research Center of Thalassemia and Hemoglobinopathy, Ahvaz Jundishapur University of Medical Sciences, Khozestan, Iran