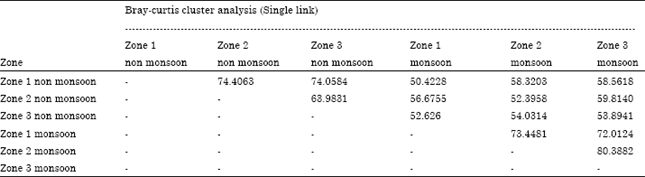
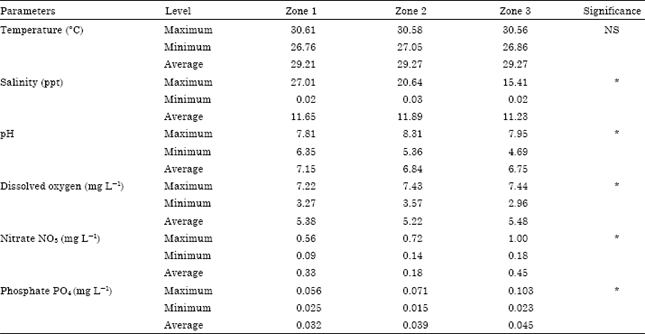
Research Article
Spatial Variation and Community Composition of Phytoplankton along the Pahang Estuary, Malaysia
Department of Biotechnology, Kulliyyah of Science, International Islamic University Malaysia, Bandar Indera Mahkota, 25200 Kuantan, Pahang, Malaysia
B.M. Ahmad Azfar
Department of Biotechnology, Kulliyyah of Science, International Islamic University Malaysia, Bandar Indera Mahkota, 25200 Kuantan, Pahang, Malaysia
B. Akbar John
Department of Biotechnology, Kulliyyah of Science, International Islamic University Malaysia, Bandar Indera Mahkota, 25200 Kuantan, Pahang, Malaysia
Y.B. Kamaruzzaman
Department of Biotechnology, Kulliyyah of Science, International Islamic University Malaysia, Bandar Indera Mahkota, 25200 Kuantan, Pahang, Malaysia