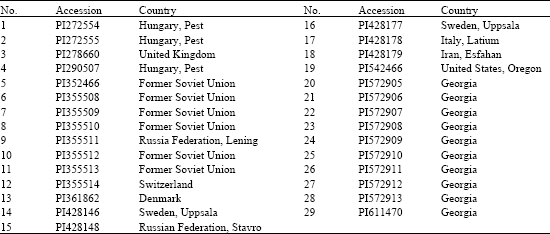
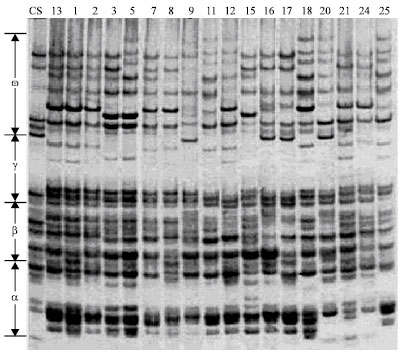
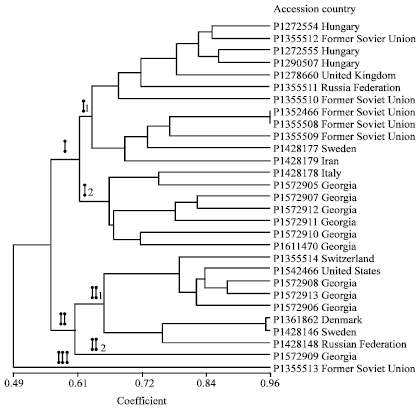
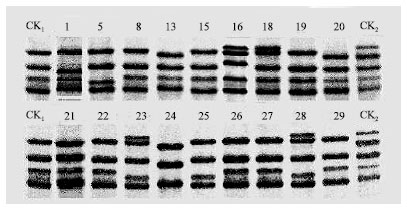
Research Article
Variation of High-Molecular-Weight Glutenin Subunits and Gliadin in T.aestivum ssp. macha
Triticeae Research Institute, Sichuan Agricultural University, Yaan, Sichuan 625014, China
Wei Li
Triticeae Research Institute, Sichuan Agricultural University, Yaan, Sichuan 625014, China
Yu-Ming Wei
Triticeae Research Institute, Sichuan Agricultural University, Yaan, Sichuan 625014, China
You-Liang Zheng
Triticeae Research Institute, Sichuan Agricultural University, Yaan, Sichuan 625014, China