Research Article
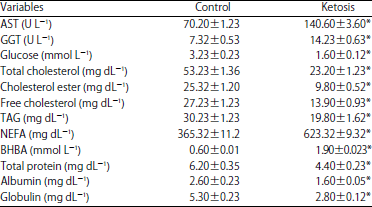
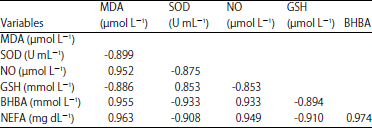
Biochemical Markers of Ketosis in Dairy Cows at Post-paturient Period: Oxidative Stress Biomarkers and Lipid Profile
Department of Clinical Studies, College of Veterinary Medicine, King Faisal University, Al-Ahsa, Saudi Arabia
LiveDNA: 20.3770
S.M. El-Bahr
Department of Biochemistry, Faculty of Veterinary Medicine, Alexandria University, Egypt
LiveDNA: 20.3674