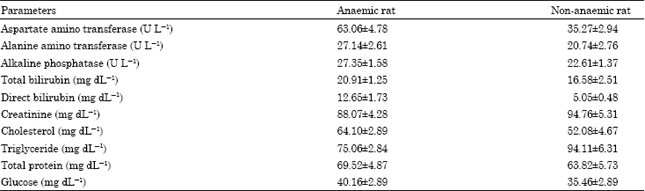
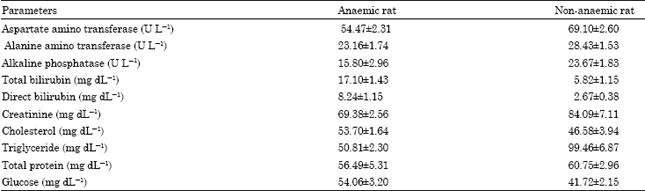
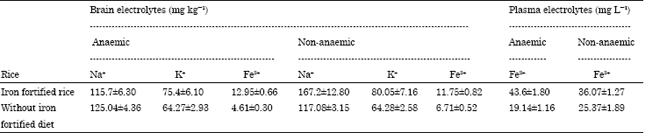
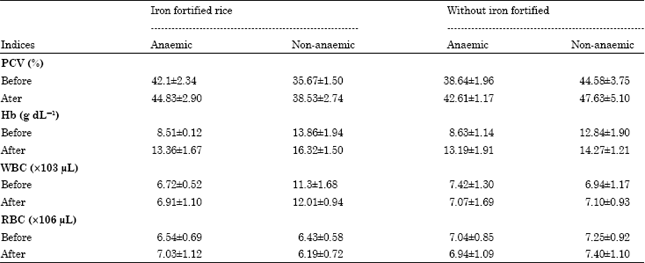
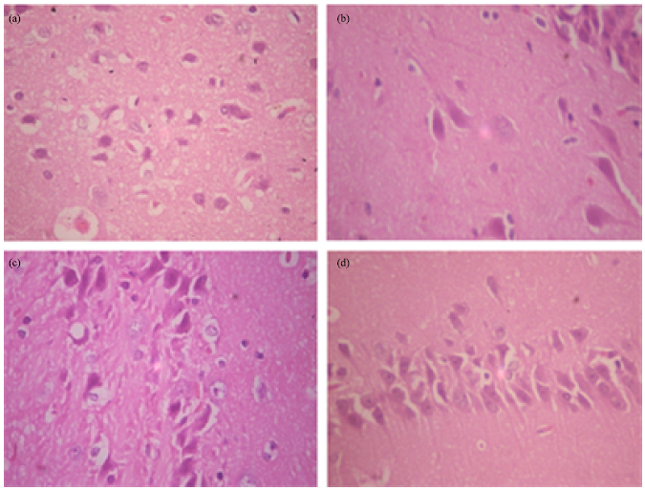
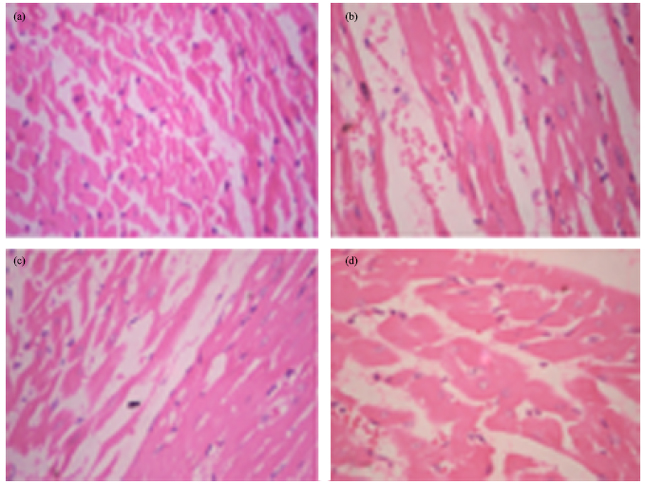
Research Article
Biochemical Studies of Iron Fortified Nigerian Rice Fed to Phenylhydrazine Induced Anaemic Rats
Department of Biochemistry, College of Medicine, University of Lagos, P.M.B. 12003, Lagos, Nigeria
LiveDNA: 234.23436
A.C. Oyewole
Department of Biochemistry, College of Medicine, University of Lagos, P.M.B. 12003, Lagos, Nigeria