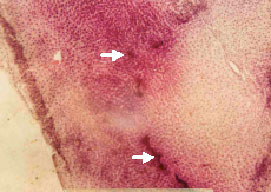
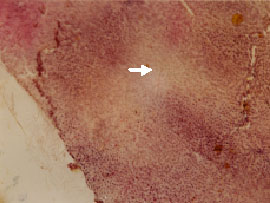
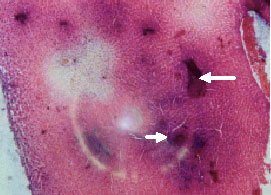
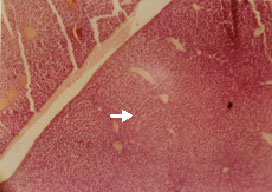
Research Article
Hepatoprotective Potential of Calcium and Magnesium Against Cadmium and Lead Induced Hepatotoxicity in Wistar Rats
Department of Biochemistry, Faculty of Medical Sciences, University of Jos, P.M.B. 2084, Nigeria
S.Y. Gazuwa
Department of Biochemistry, Faculty of Medical Sciences, University of Jos, P.M.B. 2084, Nigeria
G.A. Ubom
Department of Biochemistry, Faculty of Medical Sciences, University of Jos, P.M.B. 2084, Nigeria