ABSTRACT
The present study appraised the hepatoprotective effects of aqueous leaf extract of Camellia sinensis against carbon tetrachloride (CCl4)-induced liver injury in rats. The 100 and 200 mg kg-1 b.wt. of Camellia sinensis and 100 mg kg-1 b.wt. Silymarin was administered orally to separate groups of rats for 7 days prior to CCl4 administration. A significant decrease (p<0.05) was observed in both the groups treated with 100 and 200 mg kg-1 b.wt. of the leaf extract and Silymarin on the levels of the enzymes and non-enzyme markers of tissue damage and lipid peroxidation, with no significant changes in the relative organ weight of the treated groups. This result shows that the leaf extract of Camellia sinensis contains some phytochemical(s) that can protect the liver against CCl4-induced injury in rats.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajb.2015.86.92
URL: https://scialert.net/abstract/?doi=ajb.2015.86.92
INTRODUCTION
Camellia sinensis (Green tea) from the genus Camellia of flowering plant in the family Theaceae is a native to mainland of China, South and Southeast Asia but it is today cultivated across the world in tropical and subtropical regions. It is an evergreen shrub that is usually trimmed and the leaves and leaf buds are used to produce tea that is processed differently to attain a different level of oxidation (Mahmood et al., 2010) and it contains certain minerals and vitamins which increase the antioxidant potentials of this type of tea (Cabrera et al., 2003). Camellia sinensis extract is fast becoming ubiquitous supplement in consumer products such as shampoos, creams soaps, cosmetics, vitamins, drinks, lollipops and ice creams (Mukhtar and Ahmad, 2000).
In recent times more attention has been paid to the protective effects of natural antioxidants against drug-induced toxicities (such as CCl4) especially whenever free radical generation is involved (Wakawa and Hauwa, 2013) and several plants and other animal products have been shown to protect the liver against such damages (Dahiru et al., 2005, 2010; Obidah et al., 2010; Hamad et al., 2011; Khan and Alzohairy, 2011; Al-Fartosi et al., 2012). Also medicinal plants are used in preparations of herbal remedies and these herbal remedies which are perceived to be a cheaper means of treatment have often attained popularity for historic and cultural purposes (WHO., 1999), thus a large number of the world’s population depends on plants to treat many common ailments (Shri, 2003). The search for new and effective antioxidants in plants has given a new dimension to the antioxidant research (Guzdek and Nizankowska, 1996).
Fresh tea leaves are rich in flavanol monomers known as catechins, the major component of green tea extract which have various physiological effects and have been found to play important roles in the non-enzymatic protection against oxidative stress and body fat reduction (Nagao et al., 2005). Several studies suggest that Camellia sinensis may contribute to a reduction in the risk of cardiovascular disease and some forms of cancer (Sakanaka, 1997; Kao et al., 2000; Dulloo el al., 2000; Levites et al., 2003; Cabrera et al., 2006) and traditional uses include treating flatulence, regulating body temperature and blood sugar, promoting digestion and improving mental processes (Choi et al., 2004; Tsuneki et al., 2004; Wu and Yu, 2006).
This study is undertaken to appraised the hepatoprotective effect of aqueous leaf extract of Camellia sinensis on liver injury, a highly specialized organ that regulates a wide variety of high-volume biochemical reactions, including the synthesis and breakdown of small and complex molecules, many of which are necessary for normal vital functions in humans, against carbon tetrachloride (CCl4), a chemical model commonly used for animal studies to induce reactive oxygen formation and depletion of glutathione.
MATERIALS AND METHODS
Chemicals: Diagnostic kits for serum alanine aminotransferase (ALT) and aspartate amino transferase (AST), alkaline phosphatase (ALP) and bilirubin were purchased from Randox Laboratories Ltd. and other chemicals and solvents were of highest grade commercially available.
Camellia sinensis leaves: Freshly harvested leaves of the Camellia sinensis were used for the preparation of the crude extract. It was collected from highland tea farm land in Sarduana Local Government area of Taraba State-Nigeria (in the month of June). It was authenticated in the plant science department of Modibbo Adama University of Technology Yola and then it was then dried under room temperature.
Preparation of aqueous extract of Camellia sinensis: The freshly dried leaves of Camellia sinensis were grounded into fine powdered form using laboratory mortar and pestle. Twenty gram of the powder was weight and mixed with 250-300 mL of distilled water and boil for 5-7 min. The extract was filter through Whatman filter paper (No. 4) and the filtrate obtain was concentrated using rotary evaporator at 40°C. It was then stored under frozen condition until use.
Animals: A total number of twenty five male albino rats weighing between 110-150 kg were purchased from the animal farm, National Veterinary Research Institute Vom, Jos Plateau state-Nigeria. They were all housed in standard cages at room temperature under 12/12 night/dark and were fed with pelleted standard laboratory feed (Vital Feeds, Grand cereals and oil mills Jos) and water ad libitum. They were allowed to stay for 7 days to acclimatize before the commencement of the work.
Experimental design and treatment: The rats were randomly divided into five groups of five rats per group and were given the extract as follows:
| Group 1: | Control |
| Group 2: | Rats were given single dose of CCl4+diet/water |
| Group 3: | (Silymarin) rats were given 100 mg kg-1 b.wt., Silymarin+CCl4+diet/water |
| Group 4: | (Treated) rats were given 100 mg kg-1 b.wt., leaf extract+CCl4+diet/water |
| Group 5: | (Treated) rats were given 200 mg kg-1 b.wt., leaf extract+CCl4+diet/water |
Groups 3 and 4 and 5 were pre-treated with Silymarin and the aqueous leaf extract of Camellia sinensis for 7 days, respectively prior to CCl4 administration.
The Ccl4 was dissolved in olive oil and administered intraperitoneally (1:1) 2 mL kg-1 b.wt., to induce liver injury.
Blood and tissue collection: All the rats from the various groups were sacrificed 48 h after the CCl4 administration using standard laboratory procedures and then blood samples were collected via ocular vein into clean containers and allow to stand for 10 min. It was then centrifuged at 3000 rpm for 15 min to obtain serum. The serum was then separated for the estimation of liver marker enzymes (transaminases and alkaline phosphatase) and total bilirubin.
The liver of the rats were quickly excised, weighed and then used for the determination of lipid peroxidation. The hepatic lipid peroxidation was determined as Thiobarbituric Acid Reactive Substance (TBARS) and expressed as the amount of malonaldehyde (MDA) (Uchiyama and Mihara, 1978).
Statistical analysis: Results were expressed as Means±Standard Error of Mean (SEM). The significance of differences was calculated by using student t-test, p<0.05 was considered statistically significant.
RESULTS
The result of the effect of pretreatment against CCl4-induced liver damage in rats with aqueous leaf extract of Camellia sinensis was examined on serum ALT, AST, ALP and TB levels as shown in Table 1.
The result in Table 1 showed a significant increase (p<0.05) in the serum levels of ALT, AST and ALP in group 2 (CCl4 group) as compared to group 1 (control). However there was an observed significant decrease (p<0.05) in the 100 and 200 mg kg-1 b.wt. extract treated groups prior to the CCl4 administration as compared to group 2. The result observed in treated groups is not statistically different from the group treated with Silymarin and it was observed to be dose dependent.
The result also show a significant increase (p<0.05) in the level of bilirubin concentration in group 2 (CCl4 group) as compare to group 1 rats. A pattern of statistical decrease was also observed in the pretreated groups in the level of the bilirubin concentration as compared to the Silymarin group and group 2.
The result in Table 2 shows the effect of pretreatment with the leaf extract on lipid peroxidation. It showed a significant increase in the MDA levels in group 2 as compared to group 1 (control) with a corresponding decrease in the pretreated groups (3 and 4) and the Silymarin treated group.
| Table 1: | Effects of pretreatment with aqueous leaf extract against CCl4 induced liver damage on enzymes and non enzyme markers of liver damage |
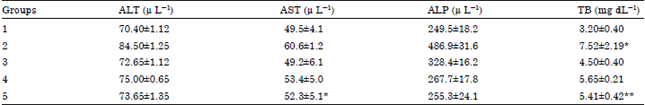 | |
Results are Mean±SEM (n = 5), *Significantly higher than control group (p<0.05), **Significantly lower than group induced with CCl4 | |
| Table 2: | Effects of pretreatment with aqueous leaf extract against CCl4 induced liver damage on liver lipid peroxidation |
 | |
| Results are Mean±SEM (n = 5), **Significantly lower than group induced with CCl4 | |
| Table 3: | Effects of pretreatment with aqueous leaf extract against CCl4 induced liver damage on relative organ weight |
 | |
| Results are Mean±SEM (n = 5) | |
The result in Table 3 showed a relative increase in relative organ weight and the mean final weigh in the CCl4 group as compared to group 1 and a relative decrease was observed in the relative organ weight of the pretreated groups as compared to the CCl4 group.
DISCUSSION
Hepatotoxicity resulting from exposure to environmental chemicals is now a major global public health problem and a number of these toxicants have been documented. CCl4 is one of the most commonly used hepatotoxins in the experimental study of liver diseases (Johnston and Kroening, 1998). The hepatotoxic effects of CCl4 are largely due to its active metabolite, trichloromethyl radical (Srivastava et al., 1990), these activated radicals bind covalently to the macromolecules and induce peroxidative degradation of membrane lipids of endoplasmic reticulum which are rich in polyunsaturated fatty acids. This lipid peroxidative degradation of biomembrane is one of the principal causes of hepatotoxicity of CCl4 (Cotran et al., 1994). Hepatotoxin can injure the hepatocytes via free radicals that initiate and propagate tissue damage.
Several studies have provided a considerable support for evidencing the protective effects of camel milk on liver damage (Hamad et al., 2011; Khan and Alzohairy, 2011; Al-Fartosi et al., 2012). Also, these studies declared that the protective effect of camel milk against CCl4-induced oxidative stress in the rat is due to its antioxidant properties. Camel milk was found to contain high concentrations of vitamins A, B2, C and E and is very rich in magnesium and other trace elements, these vitamins act as antioxidants and have been found to be useful in preventing toxicant-induced tissue injury (Yousef, 2004). The efficacy of any hepatoprotective drug is dependent on its capacity of either reducing the harmful effect or restoring the normal hepatic physiology that has been distributed by a hepatotoxin. Camel milk decreased (p<0.05) CCl4 induced elevated enzyme levels in tested groups, indicating the protection of structural integrity of hepatocytic cell membrane or regeneration of damaged liver cells (Palanivel et al., 2008).
The magnitude of hepatic damage is usually assessed by the measure of the levels of the cytosolic transaminases (alanine amino transferase (ALT) and aspartate aminotransferase (AST)), alkaline phosphatase, lipid peroxide and bilirubin which are reliable markers of liver function, in circulation (Perez Gutierrez and Solis, 2009) and from the result obtained in this study, hepatotoxicity was observed in the rats treated with CCl4 as shown by the increase in the levels of the serum marker of hepatic injury, this could be attributed to the damage caused by CCl4 on the structural integrity of the liver (Dahiru et al., 2005; Galati et al., 2005; Perez Gutierrez and Solis, 2009; Dahiru et al., 2010).
The result obtained in this study showed that pretreatment with the extract of Camellia sinensis caused a relative difference in the concentration of the marker enzymes and bilirubin concentration which is an index of jaundice (a condition indicating liver injury), possibly due to either increase production or decrease uptake by the liver, decrease conjugation and secretion from the liver or blockage of bile duct which might be as a result of CCl4 administration, as compared to the CCl4 treated group.
The difference in the result was also observed in the levels of malonaldehyde (MDA) in the pretreated group as compared to the CCl4 group, this was shown to be elevated after CCl4 administration, a toxicant that is known to increase lipid peroxidation. There was also an observed difference in the relative organ weight, a condition that normally reflect the pathological state of the liver.
The findings in this study revealed that the leaves of Camellia sinensis provided a protection the liver against hepatotoxicity due to CCl4 administration, as demonstrated by the significant difference in the levels of the marker enzymes, the non enzyme marker, bilirubin as well as the decrease liver susceptibility to lipid peroxidation and invariably oxidative stress as seen in the marked levels of MDA in the pretreated group as compared to the CCl4 induced liver damage group.
Thus the leaves of Camellia sinensis can be said to possess phytochemicals that have the capability of protecting the liver against CCl4 induced hepatotoxicity as well as cellular degeneration and fatty liver development though the mechanism is not understood.
CONCLUSION
The result of this study showed that the leaves of Camellia sinensis could block or minimized adipogenesis to a greater extend thus having a potent hepatoprotective agent against CCl4 induced liver damage in rats.
REFERENCES
- Al-Fartosi, K.G., A. Majid, M.A. Auda and M.H. Hussein, 2012. The role of camel's milk against some oxidant-antioxidant markers of male rats treated with CCl4. Int. J. Res. Pharmaceut. Biomed. Sci., 3: 385-389.
Direct Link - Cabrera, C., R. Artacho and R. Gimenez, 2006. Beneficial effects of green tea-A review. J. Am. Coll. Nutr., 25: 79-99.
PubMedDirect Link - Cabrera, C., R.Gimenez and M.C. Lopez, 2003. Determination of tea components with antioxidant activity. J. Agric. Food Chem., 51: 4427-4435.
CrossRefDirect Link - Choi, Y.B., Y.I. Kim, K.S. Lee, B.S. Kim and D.J. Kim, 2004. Protective effect of Epigallocatechin gallate on brain damage after transient middle cerebral artery occlusion in rats. Brain Res., 1019: 47-54.
CrossRefPubMedDirect Link - Dahiru, D., D.N. Mamman and H.Y. Wakawa, 2010. Ziziphus mauritiana fruit extract inhibits carbon tetrachloride-induced hepatotoxicity in male rats. Pak. J. Nutr., 9: 990-993.
CrossRefDirect Link - Dahiru, D., E.T. William and M.S. Nadro, 2005. Protective effect of Ziziphus mauritiana leaf extract on carbon tetrachloride-induced liver injury. Afr. J. Biotechnol., 4: 1177-1179.
Direct Link - Dulloo, A.G., J. Seydoux, L. Girardier, P. Chantre and J. Vandermander, 2000. Green tea and thermogenesis: Interactions between catechin-polyphenols, caffeine and sympathetic activity. Int. J. Obesity, 24: 252-258.
Direct Link - Galati, E.M., M.R. Mondello, E.R. Lauriano, M.F. Taviano, M. Galluzzo and N. Miceli, 2005. Opuntia ficus indica (L.) Mill. fruit juice protects liver from carbon tetrachloride-induced liver injury. Phytother. Res., 19: 796-800.
CrossRefPubMedDirect Link - Hamad, E.M., E.A. Abdel-Rahim and E.A. Romeih, 2011. Beneficial effect of camel milk on liver and kidneys function in diabetic sprague-dawley rats. Int. J. Dairy Sci., 6: 190-197.
CrossRefDirect Link - Johnston, D.E. and C. Kroening, 1998. Mechanism of early carbon tetrachloride toxicity in cultured rat hepatocytes. Pharmacol. Toxicol., 83: 231-239.
PubMed - Khan, A.A. and M.A. Alzohairy, 2011. Hepatoprotective effects of camel milk against CCl4-induced hepatotoxicity in rats. Asian J. Biochem., 6: 171-180.
CrossRefDirect Link - Kao, Y.H., R.A. Hiipakka and S. Liao, 2000. Modulation of obesity by a green tea catechin. Am. J. Clin. Nutr., 72: 1232-1233.
PubMedDirect Link - Levites, Y., T. Amit, S. Mandel and M.B. Youdim, 2003. Neuroprotection and neurorescue against Aβ toxicity and PKC-dependent release of nonamyloidogenic soluble precursor protein by green tea polyphenol (-)-epigallocatechin-3-gallate. FASEB J., 17: 952-954.
CrossRefPubMedDirect Link - Mukhtar, H. and N. Ahmad, 2000. Tea polyphenols: Prevention of cancer and optimizing health. Am. J. Clin. Nutr., 71: 1698s-1702s.
Direct Link - Perez Gutierrez, R.M. and R.V. Solis, 2009. Hepatoprotective and inhibition of oxidative stress in liver of Prostechea michuacana. Rec. Nat. Prod., 3: 46-51.
Direct Link - Srivastava, S.P., N.O. Chen and J.L. Holtzman, 1990. The in vitro NADPH-dependent inhibition by CCl4 of the ATP-dependent calcium uptake of hepatic microsomes from male rats. Studies on the mechanism of the inactivation of the hepatic microsomal calcium pump by the CCl3.radical. J. Biol. Chem., 265: 8392-8399.
PubMedDirect Link - Mahmood, T., N. Akhtar and B.A. Khan, 2010. The morphology, characteristics and medicinal properties of Camellia sinensis' tea. J. Med. Plants Res., 4: 2028-2033.
Direct Link - Tsuneki, H., M. Ishizuka, M. Terasawa, J.B. Wu, T. Sasaoka and I. Kimura, 2004. Effect of green tea on blood glucose levels and serum proteomic patterns in diabetic (db/db) mice and on glucose metabolism in healthy humans. Biol. Med. Chem. Pharmacol., 4: 18-18.
CrossRef - Nagao, T., Y. Komine, S. Soga, S. Meguro, T. Hase, Y. Tanaka and I. Tokimitsu, 2005. Ingestion of a tea rich in catechins leads to a reduction in body fat and malondialdehyde-modified LDL in men. Am. J. Clin. Nutr., 81: 122-129.
PubMedDirect Link - Palanivel, M.G., B. Rajkapoor, R. Senthil Kumar, J.W. Einstein and E.P. Kumar et al., 2008. Hepatoprotective and antioxidant effect of Pisonia aculeata L. against CCl4-induced hepatic damage in rats. Scientia Pharmaceutica, 76: 203-215.
CrossRefDirect Link - Uchiyama, M. and M. Mihara, 1978. Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal. Biochem., 86: 271-278.
CrossRefDirect Link - Wakawa, H.Y. and M. Hauwa, 2013. Protective effect of Erythrina senegalensis (DC) leaf extract on carbon tetrachloride-induced liver injury in rats. Asian J. Biol. Sci., 6: 234-238.
CrossRefDirect Link - Wu, A.H. and M.C. Yu, 2006. Tea, hormone-related cancers and endogenous hormone levels. Mol. Nutr. Food Res., 50: 160-169.
CrossRefPubMedDirect Link - Yousef, M.I., 2004. Aluminum-induced changes in hemato-biochemical parameters, lipid peroxidation and enzyme activities of male rabbits: Protective role of ascorbic acid. Toxicology, 199: 47-57.
CrossRefDirect Link








