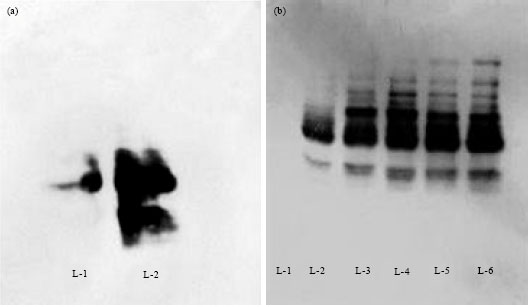
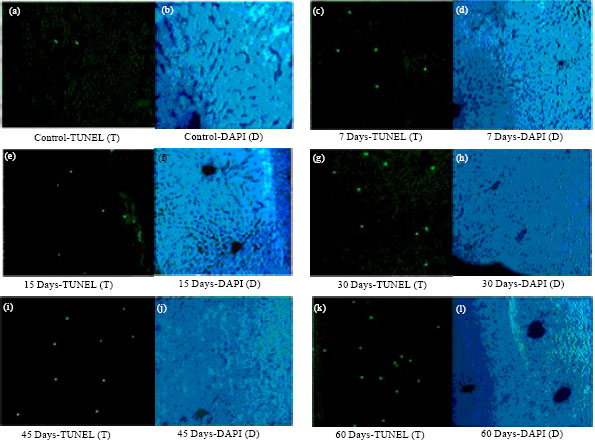
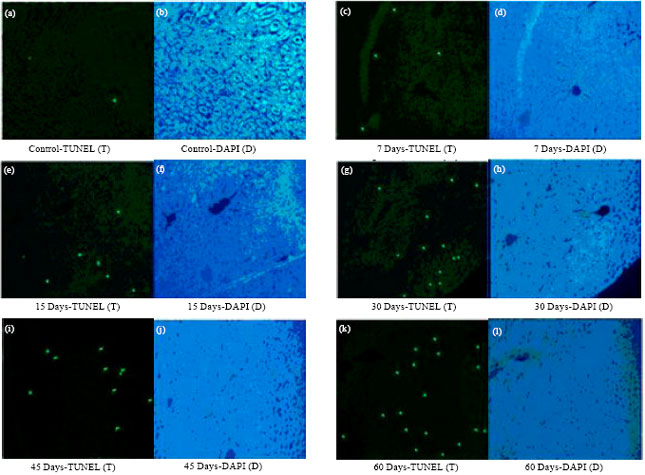
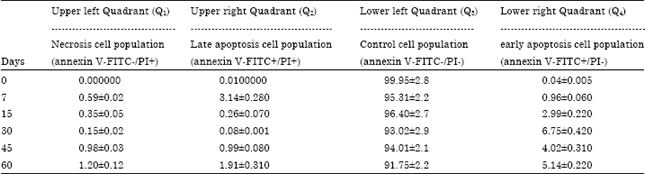
Research Article
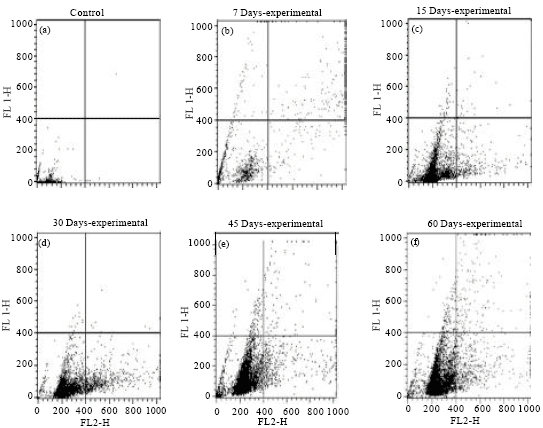
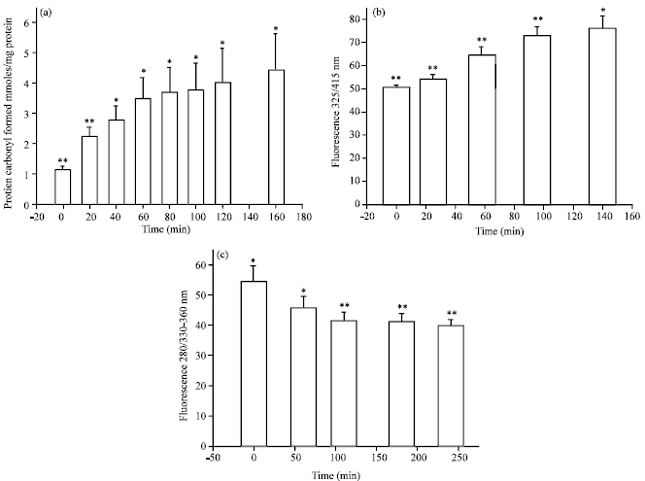
Oxidative Damage of Liver, Kidney and Serum Proteins with Apoptosis of above Tissues in Guinea Pigs Fed on Carbonated Soft Drink
Department of Biotechnology and Dr. B.C. Guha Centre for Genetic Engineering and Biotechnology, India
K. Panda
Department of Biotechnology and Dr. B.C. Guha Centre for Genetic Engineering and Biotechnology, India
P. Nandi
Department of Environmental Science, 3Department of Biochemistry, University of Calcutta, 35, Ballygunge Circular Road, Kolkata-700019, India
S.K. Banerjee
Department of Biotechnology and Dr. B.C. Guha Centre for Genetic Engineering and Biotechnology, India