Research Article
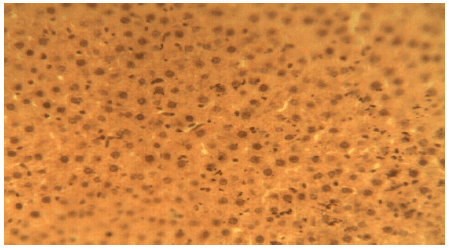
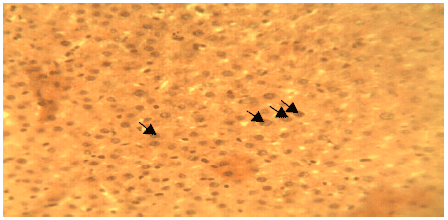
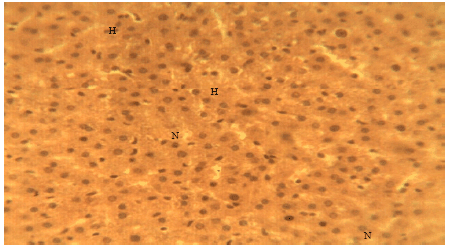
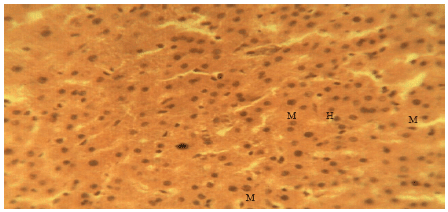
Liver Protective Activity of the Methanol Extract of Crinum jagus Bulb against Acetaminophen-induced Hepatic Damage in Wistar Rats
Department of Biochemistry, Faculty of Basic Medical Sciences, University of Calabar, Calabar, Nigeria
Florence C. Nwinyi
Department of Veterinary Pharmacology and Toxicology, Faculty of Veterinary Medicine, University of Abuja, P.M.B. 117 Abuja, Nigeria
Julius O. Ode
Department of Veterinary Pharmacology and Toxicology, Faculty of Veterinary Medicine, University of Abuja, P.M.B. 117 Abuja, Nigeria