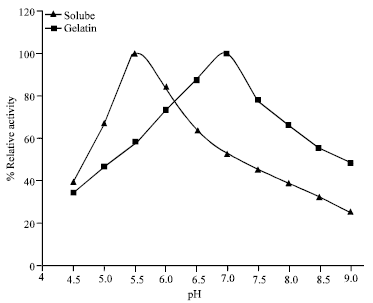
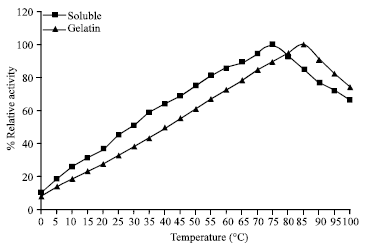
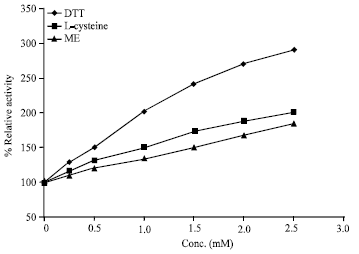
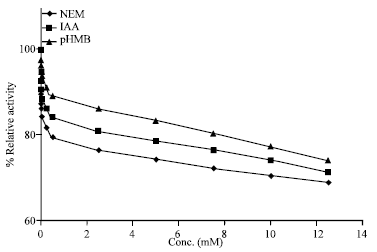
Research Article
Immobilization of Soybean α-amylase on Gelatin and its Application as a Detergent Additive
Department of Biochemistry, Faculty of Sciences, Banaras Hindu University, Varanasi-221005, U.P., India
Om Prakash
Department of Biochemistry, Faculty of Sciences, Banaras Hindu University, Varanasi-221005, U.P., India