Short Communication
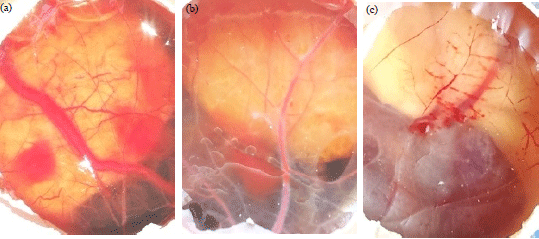
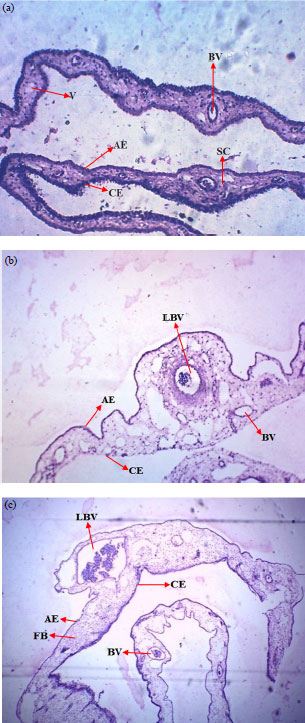
Molybdenum Salts Possess Potent Angiogenic Modulatory Properties: Validation on Chorioallantoic Membrane (CAM) of Chicken
Department of Biochemistry and Genetics, Barkatullah University, 462026 Bhopal, Madhya Pradesh, India
LiveDNA: 91.12456
Pratibha Vishwakarma
Department of Biochemistry and Genetics, Barkatullah University, 462026 Bhopal, Madhya Pradesh, India
Abhinav Dwivedi
Department of Biochemistry and Genetics, Barkatullah University, 462026 Bhopal, Madhya Pradesh, India
Anshumala Kujur
Department of Biochemistry and Genetics, Barkatullah University, 462026 Bhopal, Madhya Pradesh, India
Kuldeep Dhama
Division of Pathology, ICAR-Indian Veterinary Research Institute, Izatnagar, 243122 Bareilly, Uttar Pradesh, India
LiveDNA: 91.4710
Ashok Munjal
Department of Biochemistry and Genetics, Barkatullah University, 462026 Bhopal, Madhya Pradesh, India
LiveDNA: 91.12458