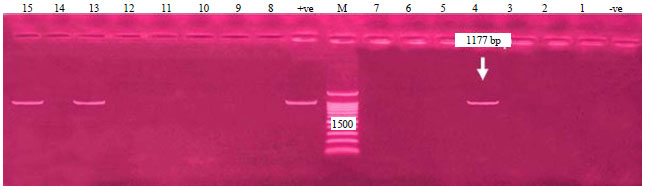
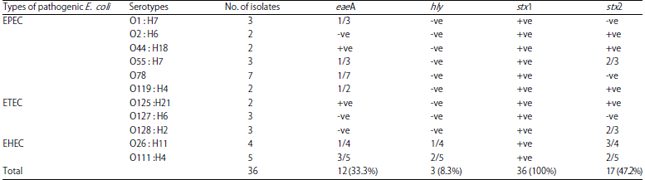
Research Article
Genetic Variation among Avian Pathogenic E. coli Strains Isolated from Broiler Chickens
Deptartment of Bacteriology, Mycology and Immunology, Faculty of Veterinary Medicine, Suez Canal University, 41522 Ismailia, Egypt
Abdelazeem M. Algammal
Deptartment of Bacteriology, Mycology and Immunology, Faculty of Veterinary Medicine, Suez Canal University, 41522 Ismailia, Egypt
LiveDNA: 20.14499
Soad A. Nasef
Reference Laboratory for Veterinary Quality Control on Poultry Production (Dokki), Animal Health Research Institute, 12618 Giza, Egypt
Wael K. Elfeil
Department of Avian and Rabbit Medicine, Faculty of Veterinary Medicine, Suez Canal University, 41522 Ismailia, Egypt
LiveDNA: 20.5311
Ghada H. Mansour
Reference Laboratory for Veterinary Quality Control on Poultry Production (Dokki), Animal Health Research Institute, 12618 Giza, Egypt