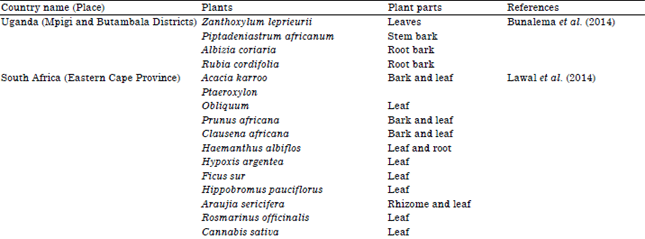
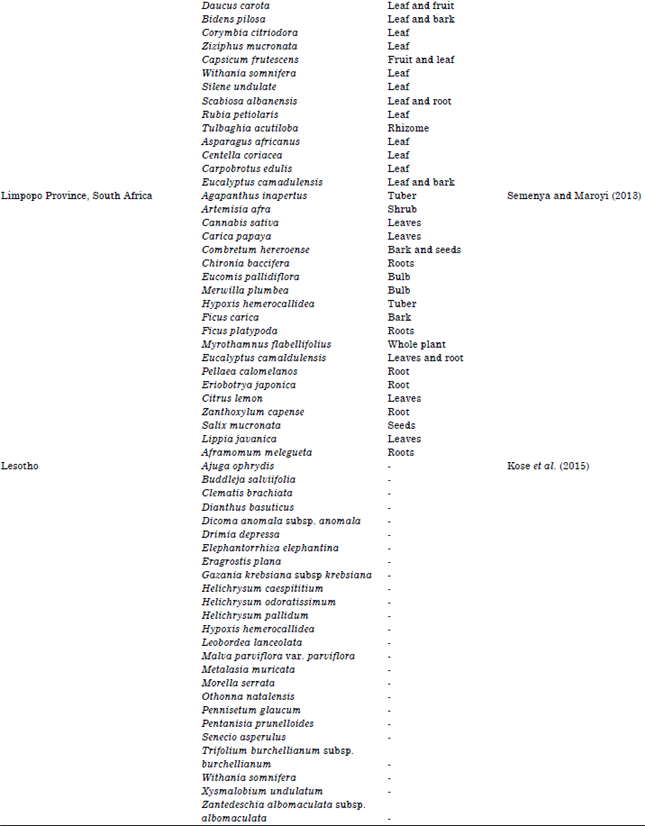
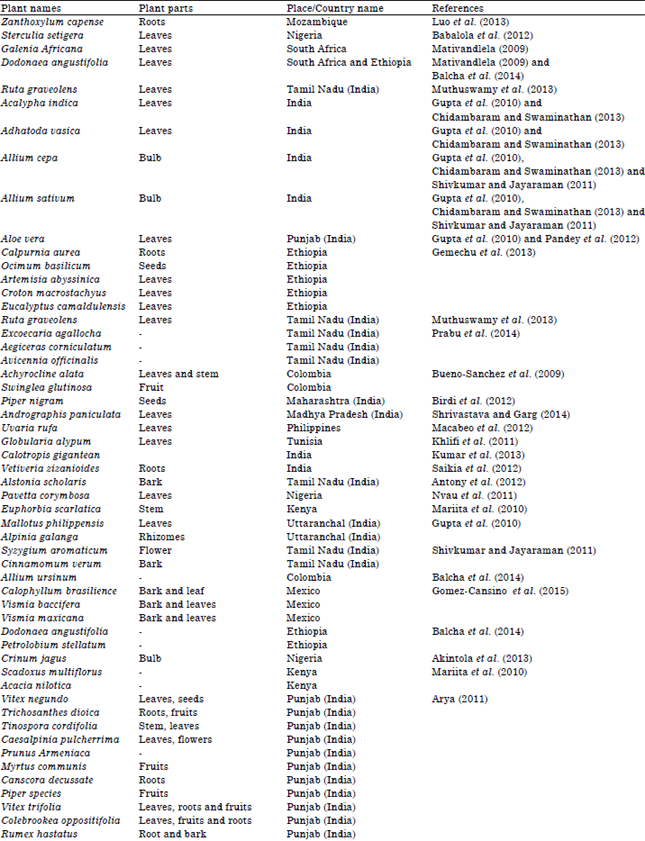
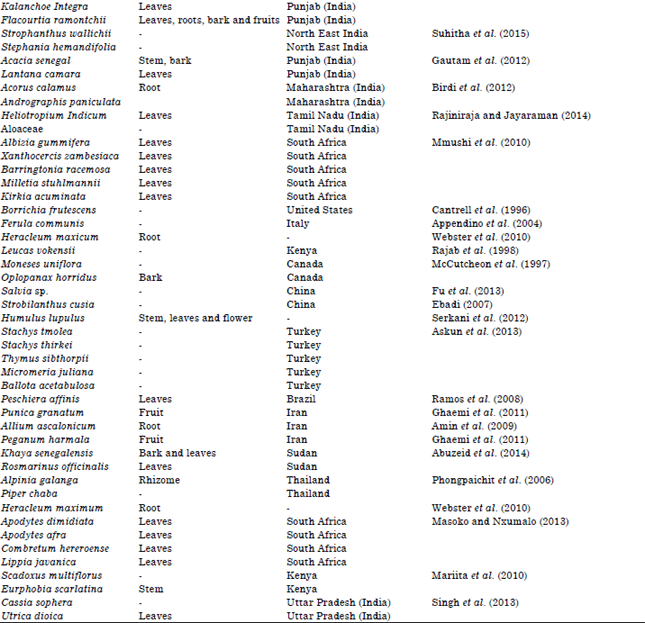
Review Article
Plants as Future Source of Anti-Mycobacterial Molecules and Armour for Fighting Drug Resistance
Amity Institute of Microbial Technology, Amity University Rajasthan, Kant Kalwar, NH-11C Delhi, Jaipur Highway, Jaipur, 303002, India
Mukta Jain
Amity Institute of Microbial Technology, Amity University Rajasthan, Kant Kalwar, NH-11C Delhi, Jaipur Highway, Jaipur, 303002, India
S.V. Singh
Animal Health Division, Central Institute for Research on Goats, Makhdoom, PO-Farah, Mathura, 281122, Uttar Pradesh, India
Kuldeep Dhama
Division of Pathology, Indian Veterinary Research Institute, Izatnagar, Bareilly, 243122, Uttar Pradesh, India
G.K. Aseri
Amity Institute of Microbial Technology, Amity University Rajasthan, Kant Kalwar, NH-11C Delhi, Jaipur Highway, Jaipur, 303002, India
Neelam Jain
Amity Institute of Biotechnology, Amity University Rajasthan, Kant Kalwar, NH-11C Delhi, Jaipur Highway, Jaipur, 303002, India
Manali Datta
Amity Institute of Biotechnology, Amity University Rajasthan, Kant Kalwar, NH-11C Delhi, Jaipur Highway, Jaipur, 303002, India
Neeraj Kumar
Amity Institute of Microbial Technology, Amity University Rajasthan, Kant Kalwar, NH-11C Delhi, Jaipur Highway, Jaipur, 303002, India
Parul Yadav
Amity University Science and Instrumentation Centre, Amity University Rajasthan, Kant Kalwar, NH-11C Delhi Jaipur Highway, Jaipur, 303002, India
Sujata Jayaraman
Amity Institute of Microbial Technology, Amity University Rajasthan, Kant Kalwar, NH-11C Delhi, Jaipur Highway, Jaipur, 303002, India
Saurabh Gupta
Animal Health Division, Central Institute for Research on Goats, Makhdoom, PO-Farah, Mathura, 281122, Uttar Pradesh, India
Kundan Kumar Chaubey
Animal Health Division, Central Institute for Research on Goats, Makhdoom, PO-Farah, Mathura, 281122, Uttar Pradesh, India
Jagdip Singh Sohal
Amity Institute of Microbial Technology, Amity University Rajasthan, Kant Kalwar, NH-11C Delhi, Jaipur Highway, Jaipur, 303002, India