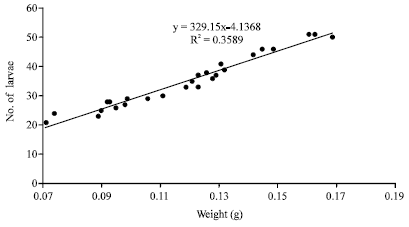
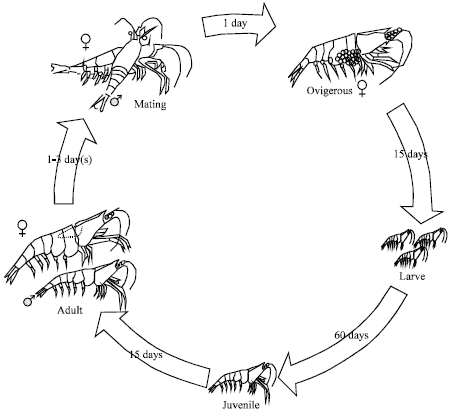
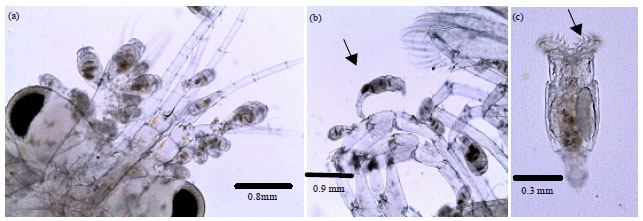
Research Article
Breeding and Life Cycle of Neocaridina denticulata sinensis (Kemp, 1918)
Department of Aquaculture, Faculty of Agriculture, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia
A. Christianus
Institute of Bioscience, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia