Research Article
Gill Histopathological Lesions of the Sturgeons
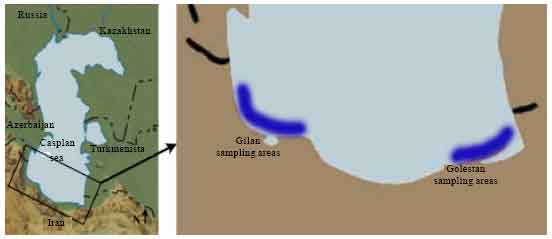
Department of Marine Biology, Faculty of Marine Sciences, Khorramshahr University of Marine Science and Technology, G.C., Khorramshahr, P.O. Box 669, Iran
Behrooz Abtahi
Department of Biology, School of Biological Sciences, Shahid Beheshti University, G.C., Tehran, Iran
Mahmood Bahmani
International Sturgeon Research Institute, Rasht, Iran