Research Article
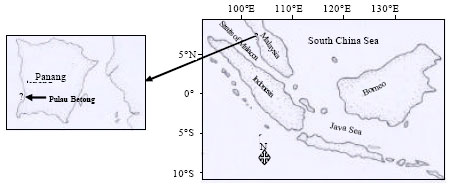
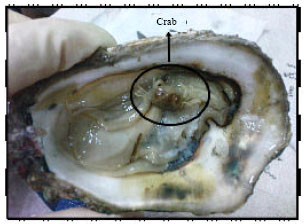
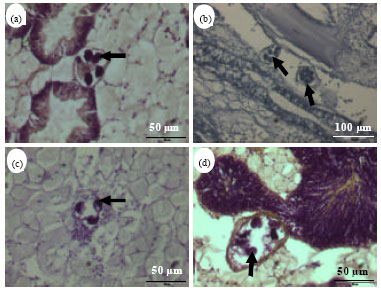
Parasites of Slipper-cupped Oyster Crassostrea iredalei from Pulau Betong, West Coast of Penang, Malaysia
Marine Science Laboratory, School of Biological Sciences, University, Sains Malaysia, 11800 Penang, Malaysia
N.B. Kamsol
Marine Science Laboratory, School of Biological Sciences, University, Sains Malaysia, 11800 Penang, Malaysia
Zulfigar Yasin
Marine Science Laboratory, School of Biological Sciences, University, Sains Malaysia, 11800 Penang, Malaysia
Aileen Tan Shau- Hwai
Marine Science Laboratory, School of Biological Sciences, University, Sains Malaysia, 11800 Penang, Malaysia