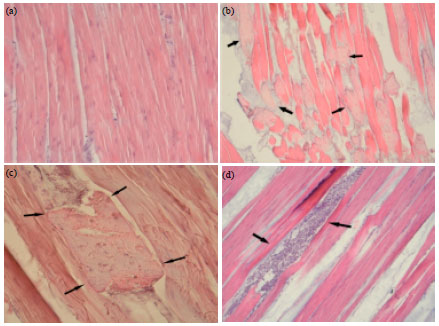
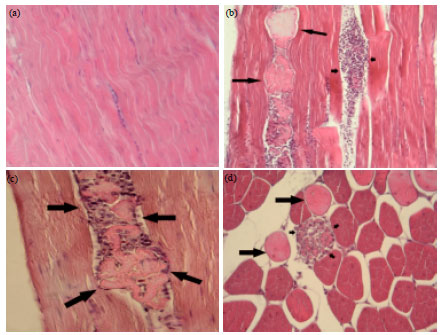
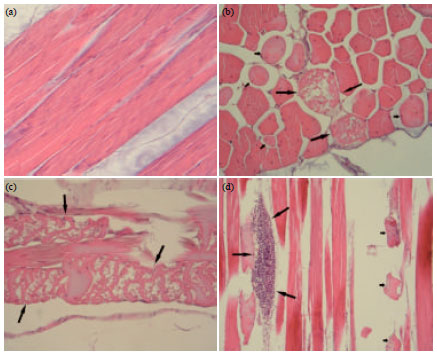
Research Article
The Influence of Dietary Boron Supplementation on Performance, Some Biochemical Parameters and Organs in Broilers
Department of Biochemistry, Faculty of Veterinary Medicine, University of Erciyes, 38039 Melikgazi, Kayseri, Turkey
Fatma Uyanik
Vocational Health College, University of Onsekiz Mart Canakkale, Canakkale 17100, Turkey
Berrin Kocaoglu Guclu
Department of Animal Nutrition and Nutritional Diseases, Faculty of Veterinary Medicine, University of Erciyes, 38039 Melikgazi, Kayseri, Turkey
Ayhan Atasever
Department of Pathology, Faculty of Veterinary Medicine, University of Erciyes, 38039 Melikgazi, Kayseri, Turkey