Research Article
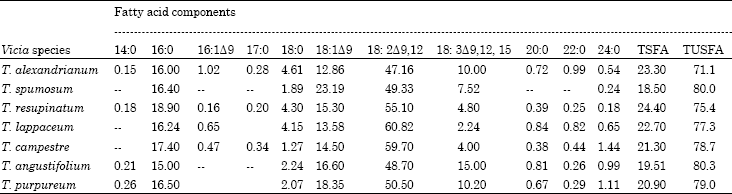
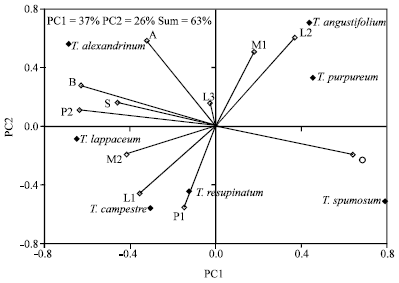
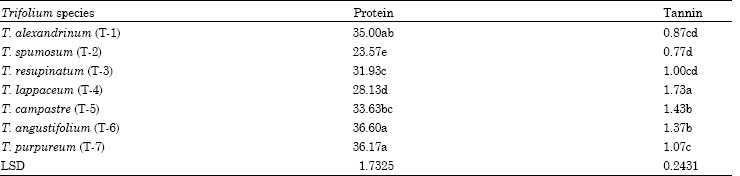
Tannin, Protein Contents and Fatty Acid Composition of the Seeds of Some Trifolium L. species from Turkey
Department of Field Crops, Faculty of Agriculture, Bingol University, Bingol, Turkey
Alpaslan Kocak
Department of Biology, Faculty of Science and Arts, Bingol University, Bingol, Turkey
Mahmut Kaplan
Department of Field Crops, Faculty of Agriculture, Erciyes University, Kayseri, Turkey
Mevlut Akcura
Department of Field Crops, Faculty of Agriculture, Bing�l University, Bing�l, Turkey
Adil Bakoglu
Program of Field Crops, Vocational School of Higher Education,Bingol University, Bingol, Turkey
Eyup Bagci
Department of Biology, Faculty of Science and Arts, Firat University, Elazig, Turkey