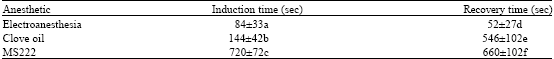Research Article
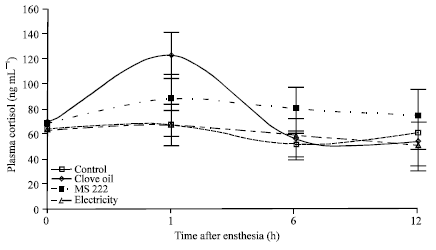
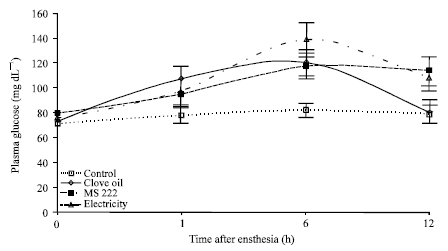
Comparison of Electroanesthesia with Chemical Anesthesia (MS222 and Clove Oil) in Rainbow Trout (Oncorhynchus mykiss) using Plasma Cortisol and Glucose Responses as Physiological Stress Indicators
Department of Aquatic Animal Health, Faculty of Veterinary Medicine, Uiversity of Tehran, P.O. Box 14155-6453, Tehran, Iran
S.S. Mirzargar
Department of Aquatic Animal Health, Faculty of Veterinary Medicine, Uiversity of Tehran, P.O. Box 14155-6453, Tehran, Iran
A. Abrishamifar
School of Electrical Engineering, Iran University of Science and Technology, P.O. Box 16846, Tehran, Iran
R. Lourakzadegan
Iranian Academic Center for Education, Culture and Research, P.O. Box 16845-311, Tehran, Iran
A. Bahonar
Department of Food Hygiene and Quality Control, Faculty of Veterinary Medicine, University of Tehran, P.O. Box 14155-6453, Tehran, Iran
H.E. Mousavi
Department of Aquatic Animal Health, Faculty of Veterinary Medicine, Uiversity of Tehran, P.O. Box 14155-6453, Tehran, Iran
A. Niasari
Department of Clinical Sciences, Faculty of Veterinary Medicine, University of Tehran, P.O. Box 14155-6453,Tehran, Iran