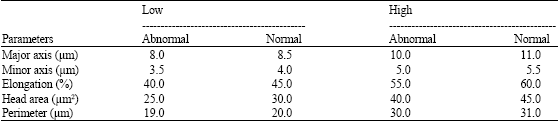
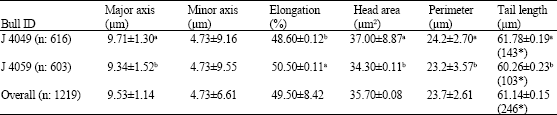
Research Article
Analyses of Morphological and Morphometrical Deviations of Bull Spermatozoa by Computer Assisted Semen Analysis Technique
Semen Bank, Department of Animal Genetics and Breeding, Madras Veterinary College, Tamilnadu Veterinary and Animal Sciences University, Chennai-600 007, India
J. Kalatharan
Semen Bank, Department of Animal Genetics and Breeding, Madras Veterinary College, Tamilnadu Veterinary and Animal Sciences University, Chennai-600 007, India
K. Thilak Pon Jawahar
Semen Bank, Department of Animal Genetics and Breeding, Madras Veterinary College, Tamilnadu Veterinary and Animal Sciences University, Chennai-600 007, India