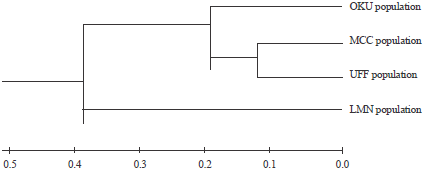
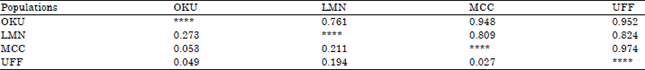
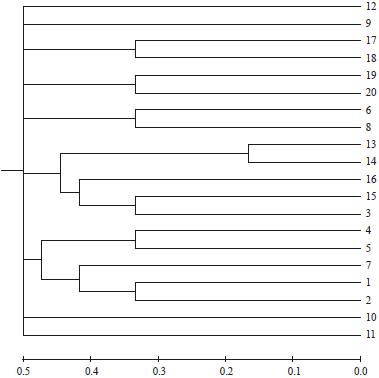
Research Article
Unveiling the Genetic Diversity in Clarias gariepinus (Burchell, 1822) Using Random Amplified Polymorphic DNA (RAPD) Fingerprinting Technique
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria
O.U. Udensi
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria
U.B. Ekaluo
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria
M.E. Kooffreh
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria
C.M. Okolo
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria
P.B. Ekpo
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria
N.C. Ogbonna
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria