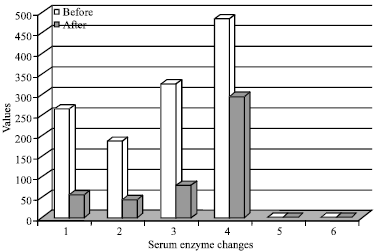
Case Study
Successful Treatment of Stanozolol Induced-hepatotoxicity with Silymarin in a Bitch
Department of Clinical Sciences, Faculty of Veterinary Medicine, Shahid Chamran University of Ahvaz, Ahvaz, Iran
R. Avizeh
Department of Clinical Sciences, Faculty of Veterinary Medicine, Shahid Chamran University of Ahvaz, Ahvaz, Iran
H. Najafzadeh Varzi
Department of Pharmacology and Toxicology, Faculty of Veterinary Medicine, Shahid Chamran University of Ahvaz, Ahvaz, Iran