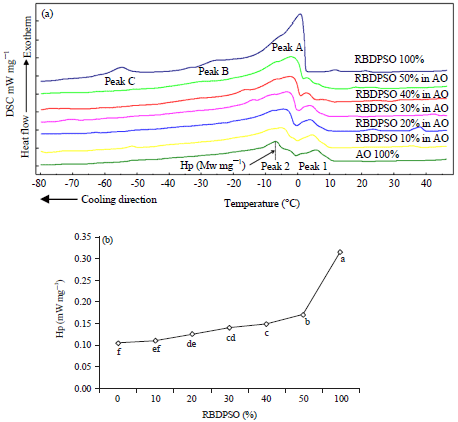
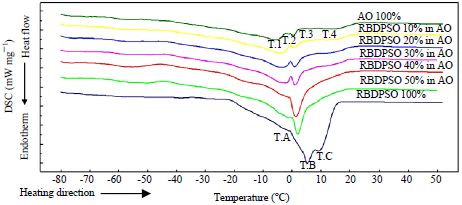
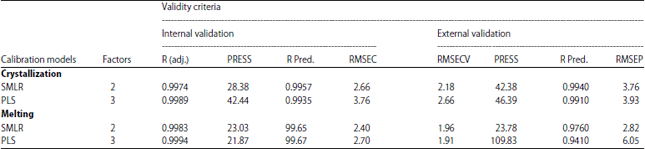
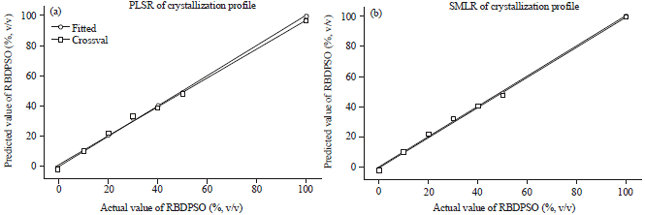
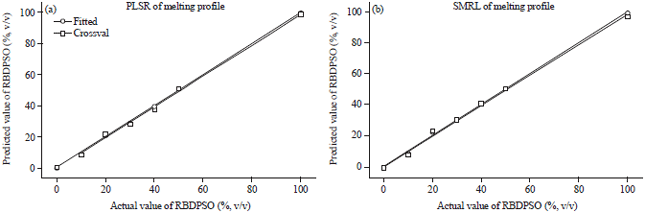
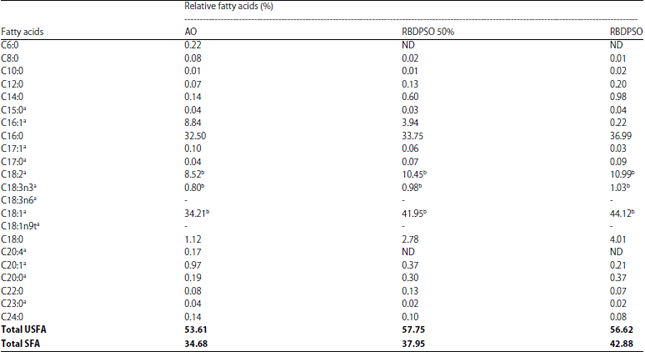Research Article
Authentication of Avocado Oil (Persea americana Mill.) Using Differential Scanning Calorimetry and Multivariate Regression
Faculty of Pharmacy, Gadjah Mada University, Yogyakarta, 55281, Indonesia
Abdul Rohman
Research Center of Halal Products, Gadjah Mada University, Yogyakarta, 55281, Indonesia
LiveDNA: 62.2819
Sugeng Riyanto
Faculty of Pharmacy, Gadjah Mada University, Yogyakarta, 55281, Indonesia