Research Article
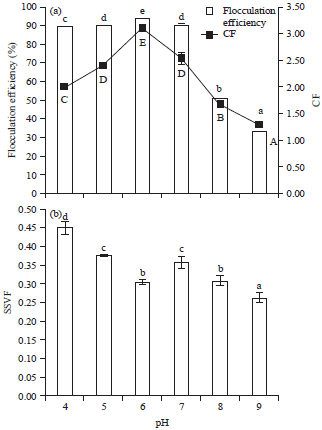
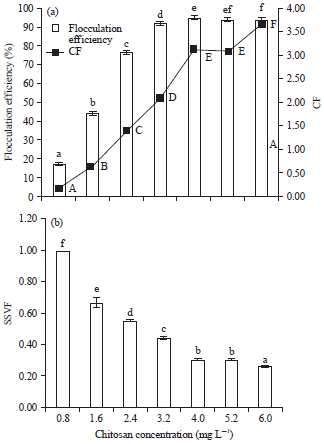
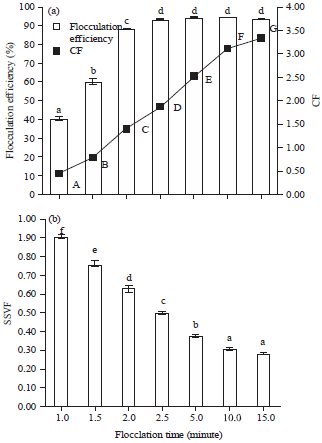
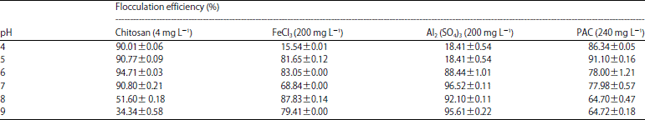
Optimization of Harvesting of Microalgal Thalassiosira pseudonana Biomass Using Chitosan Prepared from Shrimp Shell Waste
Faculty of Food Technology, Nha Trang University, 02 Nguyen Dinh Chieu, 57000 Nha Trang, Vietnam
Nguyen Thi Khanh Huyen
Faculty of Food Technology, Nha Trang University, 02 Nguyen Dinh Chieu, 57000 Nha Trang, Vietnam
Nguyen Cong Minh
Institute for Biotechnology and Environment, Nha Trang University, 02 Nguyen Dinh Chieu, 57000 Nha Trang, Vietnam
Tran Thi Le Trang
Institute of Aquaculture, Nha Trang University, 02 Nguyen Dinh Chieu, 57000 Nha Trang, Vietnam
Nguyen The Han
Faculty of Food Technology, Nha Trang University, 02 Nguyen Dinh Chieu, 57000 Nha Trang, Vietnam