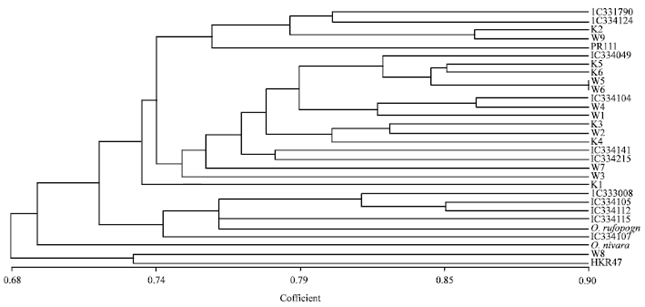
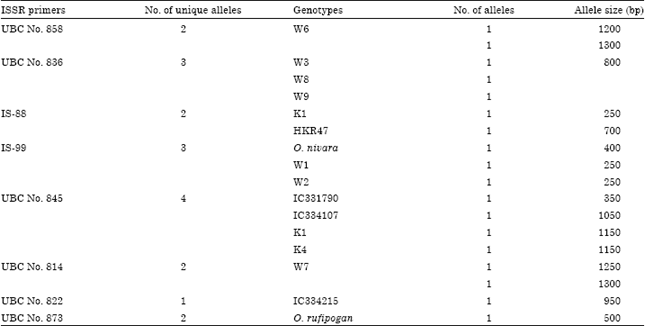
Research Article
Morphological and Molecular Variability in Weedy Rices of Haryana
Department of Genetics and Plant Breeding, CCS Haryana Agricultural University, Hisar, 125004, India
U. Ahuja
Department of Genetics and Plant Breeding, CCS Haryana Agricultural University, Hisar, 125004, India
V. Chawla
Department of Genetics and Plant Breeding, CCS Haryana Agricultural University, Hisar, 125004, India
R.K. Jain
Department of Genetics and Plant Breeding, CCS Haryana Agricultural University, Hisar, 125004, India
P. Kumari
Department of Genetics and Plant Breeding, CCS Haryana Agricultural University, Hisar, 125004, India
K.R. Batan
Department of Genetics and Plant Breeding, CCS Haryana Agricultural University, Hisar, 125004, India