Research Article
Changes in Different Blood Parameters during Mesobuthus tamulus gangeticus Pocock Envenomation
Department of Zoology, Mahatma Gandhi Post Graduate College, Gorakhpur-273 001, U.P., India
LiveDNA: 91.16671
zINTRODUCTION
Scorpion sting is a serious health problem of poor communities in tropical and subtropical countries. Out of 1500 scorpion species distributed throughout the world, 50 are lethal to humans (Keegan, 1980). The most toxic scorpion species such as Androctonus, Buthus, Centruroides, Leiurus and Tityus are distributed worldwide and belong to family Buthidae. In India, 45 scorpion species of family Buthidae have been reported (Murthy, 2000). The symptoms of this scorpion envenomation vary species to species, age of scorpion, venom composition and victim’s physiological response to the venom. Scorpion sting causes a wide range of conditions from severe local skin reactions to neurological, respiratory and cardiovascular collapse. Being a rich source of various polypeptides, scorpion venom exerts their action mainly by affecting specific functions of the target channels (Gordon et al., 1992). Buthoid venom causes severe consequences against a wide variety of organisms (Murthy and Haghanzari, 1999; Amaral and de Rezende, 1997). Its toxicity has been attributed to the presence of a large variety of basic polypeptides (Rochat et al., 1979).
All scorpion venoms cause initial transient hypertension followed by hypotension (Bawasker and Bawasker, 1991). Symptoms like severe pain, inflammation, hypersalivation, dysphagia and restlessness appear within a few minutes of sting and reach to a maximum severity within 5 h and persist for 24-72 h. Death of the victim occurs usually by anaphylaxis, bronchoconstriction, pharyngeal secretion, diaphragmatic paralysis and respiratory failure. Most of the symptoms of scorpion sting are due to release of catecholamines from adrenal glands and release of acetylcholine from postganglionic parasympathetic neurons. All lethal scorpion venoms cause acute myocarditis (Murthy and Yeolekar, 1986), intravascular coagulation (Murthy et al., 1988), acute pancreatitis (Murthy et al., 1989), pulmonary oedema (Murthy et al., 1991), increase in catecholamines (Amaral and de Rezende, 1997), angiotensin II (Murthy and Vakil, 1988), glucagon, cortisol (Murthy and Haghanzari, 1999), decrease in thyroxine (Murthy and Zare, 1998) and insulin secretion (Murthy and Haghanzari, 1999). Due to heterogeneous nature, scorpion venoms show variable reactions in the victim. However, closer the phylogenic relationship among the scorpions, more similar the symptoms and immunologic properties. Furthermore, the various constituents of in venom may act directly or indirectly and individually or synergistically to exert their effects.
In the present study, effect of scorpion M. tamulus gangeticus venom on Red Blood Cell (RBC) count, blood haemoglobin, Mean Corpuscular Haemoglobin (MCH), White Blood Cell (WBC) count, Packed Cell Volume (PCV) and plasma haemoglobin was determined in albino mice. In vitro effects of M. tamulus gangeticus venom on RBCs was also studied.
Isolation of Venom and Determination of Toxicity
Venom from living scorpions M. tamulus gangeticus was obtained by electric stimulation of scorpion telson, dissolved in phosphate buffer (50 mM, pH 7.2), centrifuged (MP01, Tarson Co., India) at 3,000 g at 4°C for 5 min and collected supernatant. Toxicity of M. tamulus gangeticus venom was determined in albino mice (NIH strain) weighing 25±5 g by Chaubey and Upadhyay (2008) method.
Experimental Protocol for Haematological Analysis
Three sets of albino mice weighing 25±5 g were taken to study the effect of scorpion M. tamulus gangeticus venom. Animals of first set consisting of twelve albino mice were injected with 40% of 24 h LD50 and that of second set consisting of twelve albino mice with 80% of 24 h LD50 of scorpion venom subcutaneously. Mice of first two sets were divided into two groups each of six animals. Blood samples were taken from mice of group A and B of first and second sets after 2 and 4 h of envenomation, respectively for different haematological analysis. Mice of third set consisting of six animals serving as control received only phosphate buffer (50 mM, pH 7.2). All the laboratory works of the present study were carried out in Post Graduate Department of Zoology, Mahatma Gandhi Post Graduate College, Gorakhpur, U.P., in the year 2010.
Determination of red blood cells count, blood haemoglobin, Mean Corpuscular Haemoglobin (MCH), white blood cells count, Packed Cell Volume (PCV) and plasma haemoglobin:
At the end of the experiment, blood samples were individually collected from each mouse in dry clean tubes using ethylenediaminetetraacetic acid (EDTA) as anticoagulant for haematological examination. Determination of red blood cells count, blood haemoglobin, mean corpuscular haemoglobin, white blood cells count, packed cell volume and plasma haemoglobin was done according to Dacie and Lewis (1984).
In vitro Study of Haemolysis
In vitro haemolytic assay was performed by pre-incubating red blood cells with different concentrations of M. tamulus gangeticus venom i.e., 10, 20, 30, 50, 70 and 90 μg/100 μL suspensions of red blood cells. Control contained no venom. The number of red blood cells was counted for each concentration of venom.
Statistical Analysis
Results have been expressed as Mean±SE of six replicates. Student t-test (Armitage et al., 2002) was used to test the significant deference with controls and between two sublethal doses and the exposure periods.
Median lethal dose (LD50) of scorpion M. tamulus gangeticus venom was 2.5 mg kg-1 b.wt. of albino mice. Mice were treated with 40 and 80% of 24 h LD50 of scorpion venom and red blood cell count, blood haemoglobin, mean corpuscular haemoglobin, white blood cell count, packed cell volume and plasma haemoglobin was measured after 2 and 4 h of experimental envenomation. The number of red blood cells was decreased to 75.80 and 61.13% of the control after 4 h of treatment with 40 and 80% of 24 h LD50 of M. tamulus gangeticus venom, respectively (Table 1). The hemoglobin level in blood was increased to maximum (135% of the control) after 4 h of treatment with 80% of 24 h LD50 of M. tamulus gangeticus venom (Table 1). The level of mean corpuscular haemoglobin was increased to 131.95 and 163.62% of the control after 4 h of treatment with 40 and 80% of 24 h LD50 of M. tamulus gangeticus venom, respectively (Table 1). The maximum increase in number of total white blood cells (132.76% of control) was found after 4 h of treatment with 80% of 24 h LD50 of M. tamulus gangeticus venom (Table 1). Packed cell volume was observed 119 and 134.78% of the control after 4 h of treatment with 40 and 80% of 24 h LD50 of M. tamulus gangeticus venom, respectively (Table 1). Haemoglobin level in plasma was increased to 0.7 and 1.1 g/100 mL of plasma after 4 h of treatment with 40 and 80% of 24 h LD50 of M. tamulus gangeticus venom, respectively (Table 1). The variation in red blood cells, blood haemoglobin, mean corpuscular hemoglobin, total white blood cells, packed cell volume and plasma haemoglobin was time and dose-dependent (p<0.05, Student t-test).
| Table 1: | Effect of 40 and 80% of 24 h LD50 of scorpion M. tamulus gangeticus venom on Red Blood Cell (RBC) count, blood haemoglobin (Hb), Mean Corpuscular Haemoglobin (MCH), White Blood Cell (WBC) count, Packed Cell Volume (PCV) and plasma haemoglobin (Hb) in albino mice |
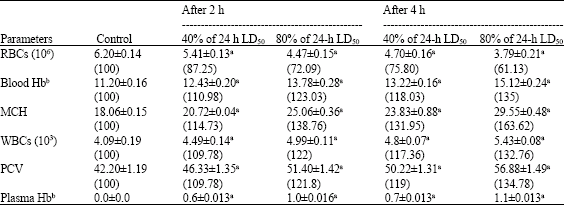 | |
| Values are Mean±SE of six replicates. Values in parentheses indicate percent level with control taken as 100%. aSignificant (p<0.05, Student t-test). bValues have been represented as g/100 mL | |
| Table 2: | In vitro effect of scorpion M. tamulus gangeticus venom on RBCs of albino mice |
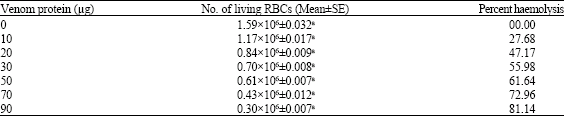 | |
| Values are Mean±SE of six replicates. aSignificant (p<0.05, Student t-test) | |
In vitro hemolytic assay included pre-incubation of red blood cells in different concentrations of M. tamulus gangeticus venom in suspension. Incubation of red blood cells with scorpion venom caused concentration-dependent lysis of red blood cells (p<0.05, Student t-test). The per cent hemolysis was found 27.68, 47.17, 55.98, 61.64, 72.96 and 81.14% at 10, 20, 30, 50, 70 and 90 μg of M. tamulus gangeticus venom, respectively (Table 2).
Mesobuthus tamulus gangeticus venom caused significant reduction in red blood cell count and increase in white blood cell count, blood haemoglobin, mean corpuscular haemoglobin, packed cell volume and plasma haemoglobin in experimentally envenomed albino mice. Decreased red blood cell count due to the haemolytic effect of scorpion venom is supported by increased hemoglobin level in plasma (Cronkite, 1973). This causes anemia and circulatory hypoxia (Dacie and Lewis, 1984). Maximum increase in blood haemoglobin (135%) observed after treatment with M. tamulus gangeticus venom may probably the result of haemoconcentration caused by a massive release of catecholamines and angiotensin II (Goyffon et al., 1982; Murthy and Vakil, 1988). An increase in plasma haemoglobin after scorpion M. tamulus gangeticus envenomation indicates intravascular haemolysis (Cronkite, 1973). When the haemolysis rate is excessive, the plasma extra corpuscular haemoglobin cannot be converted into bilirubin as quickly as it is released. When plasma haemoglobin concentration exceeds the haemoglobin binding capacity and kidney tubular capacity, the excess free plasma haemoglobin is filtered and excreted in the urine causing haemoglobinuria. Different scientific groups have given different reasons of intravascular haemolysis. Cobra venom releases an enzyme, phospholipase, which converts lecithin to lysolecithin, a powerful haemolytic and cytotoxic substance (Cronkite, 1973). Since lecithin is present in erythrocytes and all cells, the introduction of the venom into the body stimulates the production of haemolytic substance, lysolecithin. This could be the cause of haemolysis associated due to scorpion envenoming (Radmanesh, 1990). Haemolytic activity of scorpion venom peptides may also be associated with certain structural characteristics formed by the constituent peptides when come in the contact with biological membranes (Torres-Larios et al., 2000). An imbalance is created due to increased secretion of catabolic counter-regulatory hormones like catecholamines, epinephrine, norepinephrine, glucagon, cortisol, thyroxine, triiodothyronine and reduction in anabolic hormone, insulin which might have contributed the fragility of red blood cells resulting in haemolysis (Murthy and Zare, 2001). Increased mean corpuscular haemoglobin in experimental albino mice after M. tamulus gangeticus envenomation is an indicative of haemolysis (Dacie and Lewis, 1984). Increase in white blood cell count observed after treatment with scorpion M. tamulus gangeticus venom was found similar with envenomation of red scorpion Mesobuthus tamulus which may probably due to the myocardial infarction (Murthy and Zare, 2001). Packed cell volume was increased after scorpion M. tamulus gangeticus envenomation in experimental mice similar to other red scorpion envenomation (Murthy and Zare, 2001). The elevation of angiotensin II after scorpion envenomation produces a significant decrease in blood volume by shifting the fluid from intravascular to extravascular compartments and consequently increases packed cell volume (Douglas, 1985; Murthy and Vakil, 1988). All the variations in red blood cell count, white blood cell count, blood haemoglobin, mean corpuscular haemoglobin, packed cell volume and plasma haemoglobin was time and dose-dependent (p<0.05, Student t-test, Table 1). In vitro pre-incubation of red blood cells with scorpion venom caused concentration-dependent lysis of red blood cells (p<0.05, Student t-test, Table 2). This result of in vitro study and increased plasma haemoglobin in envenomed albino mice confirm that scorpion M. tamulus gangeticus envenomation caused lysis of red blood cells indicating intravascular haemolysis in experimentally envenomed albino mice. Earlier, the presence of haemolytic activity has been reported in other scorpions like Tityus serrulatus and Heterometrus fastigiousus (Correa et al., 1997; Chaubey, 2008). All these earlier studies support the findings of the present study.
Author is highly thankful to University Grants Commission, New Delhi for financial assistance (No. F. 6-2(57)/2008).