Research Article
Physicochemical Analysis of Drinking Water in Kohdasht City Lorestan, Iran
Department of Environmental Health, Lorestan University of Medical Science, Khoramabad, Iran
H. Mirhossaini
Department of Environmental Health, Lorestan University of Medical Science, Khoramabad, Iran
B. Kamareii
Department of Environmental Health, Lorestan University of Medical Science, Khoramabad, Iran
S. Dehestani
Department of Environmental Health, Lorestan University of Medical Science, Khoramabad, Iran









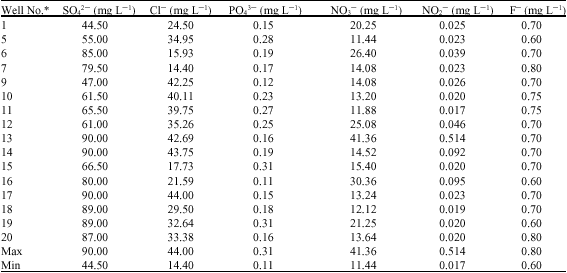
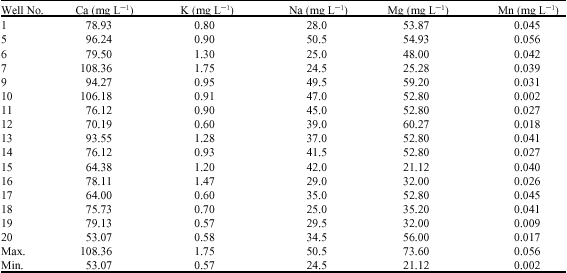
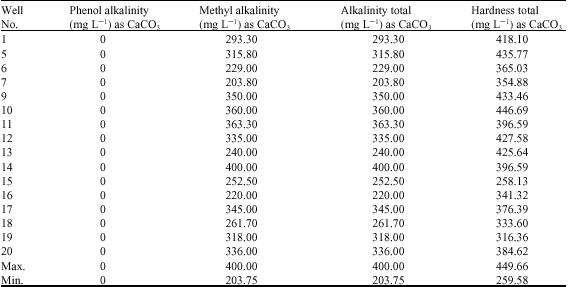
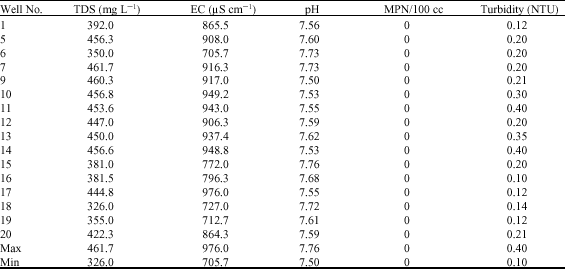
Alfred Adjei Sarkodie Reply
A very good work done.