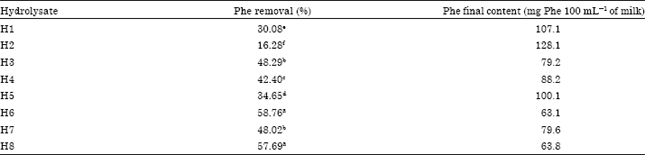
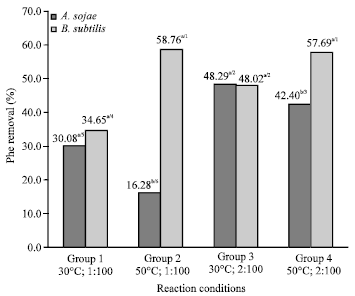
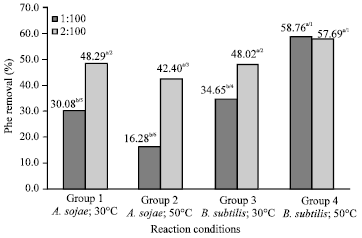
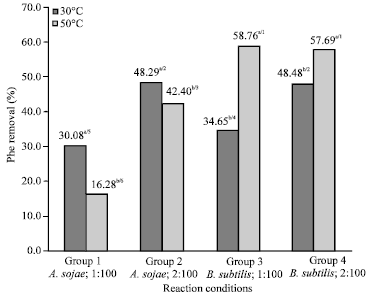
Research Article
Effect of Enzyme Type, Enzyme Substrate Ratio and Temperature on Phenylalanine Removal from Milk
Universidade Federal de Minas Gerais (UFMG), Belo Horizonte, Minas Gerais, Brazil
Mariana W.S. de Souza
Universidade Federal de Minas Gerais (UFMG), Belo Horizonte, Minas Gerais, Brazil
Carlos O. Lopes Junior
EDETEC Ind�stria Aliment�cia S/A, Belo Horizonte, Minas Gerais, Brazil
Viviane D.M. Silva
EDETEC Ind�stria Aliment�cia S/A, Belo Horizonte, Minas Gerais, Brazil
Marcos J.B. Aguiar
Universidade Federal de Minas Gerais (UFMG), Belo Horizonte, Minas Gerais, Brazil
Wendel O. Afonso
EDETEC Ind�stria Aliment�cia S/A, Belo Horizonte, Minas Gerais, Brazil
Mauro R. Silva
EDETEC Ind�stria Aliment�cia S/A, Belo Horizonte, Minas Gerais, Brazil