Research Article
Analysis of the Textural Characteristics and Pore Size Distribution of a Commercial Zeolite using Various Adsorption Models
Faculty of Chemical Engineering, Universiti Teknologi, Shah Alam, 40450 Selangor, Malaysia
Chun Yang Yin
School of Chemical and Mathematical Sciences, Murdoch University, Murdoch, WA 6150, Australia
Robert Mikhail Savory
Faculty of Chemical Engineering, Universiti Teknologi, Shah Alam, 40450 Selangor, Malaysia









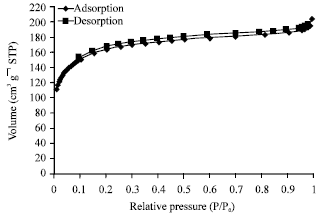
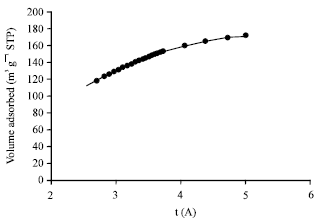
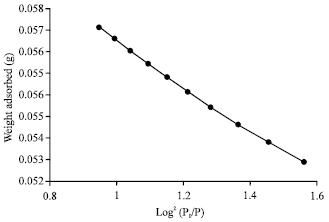
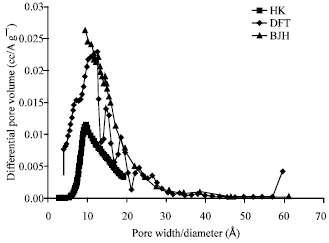
alien blanco flores Reply
this aspects is very important to investigations work