Research Article
Isolation of Fungi and their Mycotoxin Extract from Stored Wheat and Other Grains Importer in Saudi Arabia
Princess Nora Bint Abdul Rahman University, P.O. Box 102275, Riyadh, 11675, Kingdom of Saudi Arabia
LiveDNA: 966.23136
Wheat is an ingredient used in many foods and is one of the most important foods in European an American culture. Bread, pasta, crackers and many cakes, among other foods and cooking recipes, are made using flour or including this as an ingredient. Wheat (Triticum spp.) (Donner et al., 2000) is a cereal grain, originally from the Levant region of the Near East and Ethiopian Highlands but now cultivated worldwide. In 2010, world production of wheat was 651 million tons, making it the third most-produced cereal after maize (844 million tons) and rice (672 million tons). However, pathogens that contaminate wheat may survive for extended periods (Berghofer et al., 2003; Cabanas et al., 2008; Gashgari et al., 2010). Wheat were also found to be contaminated in variable amounts by potentially toxigenic fungi including Aspergillus, Alternaria and Fusarium (Halt, 1998; Tournas and Katsoudas, 2008). These fungi are present in soil and plant material, cause the decay of stored grain and food (Herrman, 2002). The word mycotoxin was derived from mycotoxicosis which was a term first used in 1955 to describe diseases of animals caused by toxics metabolic by-products of certain fungi (Herrman, 2002) which includes mushrooms, molds and yeast. Mycotoxins can appear in the food chain as a result of fungal infection of crops, either by being eaten directly by human, or used as livestock feed. Mycotoxins greatly resist decomposition or being broken down in digestion, so they remain in the food chain even after heat treatment, such as cooking and freezing (Urraca et al., 2004). The most common mycotoxins are aflatoxins, ochratoxin A, fumonisins, deoxynivalenol, T-2 toxin and zearalenone. Of these, aflatoxins represents the main threat world-widely due to their occurrence and toxicity. Aflatoxins are produced by the fungi Aspergillus flavus and Aspergillus parasiticus (Aycicek et al., 2005). Aflatoxins occur naturally in most of the commodities, including wheat, corn, soybean and peanut and other grains which are consumed by human and animal. High moisture and temperature are two main factors that cause the occurrence of mycotosins at pre-harvest and post harvest stages (Aycicek et al., 2005). Aflatoxins may increase stress susceptibility and compromise growth efficiency. The clinical signs of aflatoxicosis are extremely varied. Signs of acute aflatoxicosis include depression, nervousness, abdominal pain, diarrhea and death (Herrman, 2002). Since these toxins have been considered unavoidable contaminants in food chain, the Food and Drug Administration (FDA) of USA has established an action 732 level for total aflatoxins which is at 20 ppb for all foods, including animal feeds (Munkvold et al., 2005). Of the currently identified many types of aflatoxins, aflatoxin B1, B2, G1 and G2 occur naturally and are significant contaminants of a wide variety of foods and feeds (Juan et al., 2008). Samples of wheat, corn and other grains based product were analysed for the presence of aflatoxins in this study as these food were commonly consumed by saudian without any awareness of their safety from any local organizations. The aim of this study was to screen the content of aflatoxins in samples of wheat, corn and other grains based products available in Saudia Arabia that were widely consumed in huge amounts, as well as providing sensitive, accurate and reproducible analytical method for the detection of aflatoxins to assess the exposure of consumers to the toxins in order to bring them to the attention of the importance of monitoring the levels of aflatoxins in the wheat, corn and other grains based products. So, this study aimed to determine the occurrence and load of fungi in wheat and other grains offered for sale to consumers retail stores at Riyadh region in Saudi Arabia.
Samples: Fungal infected were collected from different regions such as: Wadi Al-Dawser, Dhurmah, Al-Qassim, Najran, Yemen, Al-Sharjah, Dubai, Turkey, India, Asuteralia, Ethiopia. Wheat samples include brown wheat from Wadi Al-Dawser, Dhurma, Al-Qassim, Najran, Yemen and Dubai, groats from Turkey and Dubai, grains from Austria and Al-Sharjah, millet from India and corn from India and Ethiopia. Sixteen samples were collected from storage shops. The collected samples were put into sterile polythene bags and sealed properly. The samples were stored and analyzed the day after collection. They were brought into laboratory for further processing (Sekar et al., 2008).
Isolation and identification of fungi: About 1 g of the collected samples were washed aseptically with ten successive 10 mL volumes of sterile distilled water and was surface sterilized using KOH 10% and then rinsed with sterile distilled water. Czapec (dox) agar media were prepared and chloramphenicol (500 mg L-1) was added to inhibit bacterial growth. About 1 g of the washed samples was inoculated randomly in each of the Czapec (dox) plate. For each sample, three replicates were maintained. The plates were incubated at room temperature and examined daily for growth and sporulation for 5 days. After 5 days of incubation the different fungal colonies were transferred into fresh Czapec (dox) plates. The fungi isolated were identified by a drop of distilled water placed on a clean glass slide and a loopful of the fungal colony was taken and placed on the slide. With the help of sterile needles, the fungal mycelia were teased gently and a cover slip was placed over the drop of distilled water. The slide is then observed under 40X power in microscope and identified based on morphological characteristics (Sekar et al., 2008).
Extraction and clean-up: A 30 g of ground sample was mixed with 100 mL of mixture of methanol and distilled water at ratio of 80:20 and shaked for 1 h. The extract was filtered through Advantec filter paper No.131. A 9 mL of filtrate was then transferred into a test tube and clean-up cartridge which was obtained from Romer Labs. Inc. (Binder et al., 2007).
Chromatography: HPLC is the preferred method for analyzing aflatoxins. A variety of separation and quantitation modes using reverse-phase chromatography (RPLC) have been developed. The RPLC employs a nonpolar bonded silica surface and a polar mobile phase. For the analysis of aflatoxins, silica-based HPLC columns bonded with C8 or C18 groups are used with mobile phases consisting of binary or ternary mixtures of polar solvents. Commonly used solvent mixtures include deionized water, methanol and acetonitrile. In the reversed phase mode, the elution order of the common aflatoxins is G2, G1, B2 and B1. Aflatoxins may be separated and detected by UV detection (Sekar et al., 2008).
The results of isolation of some species of microorganisms from 16 wheat and small grains samples collected from different Mills in Riyadh, a total of three genera of fungi were isolated (Table 1). Fungi are remarkable organisms that readily produce a wide range of natural products called secondary metabolites. Some are beneficial (e.g., antibiotics) to human kind, while others are deleterious (e.g. mycotoxins). Fungi that exhibit filamentous growth and have a relatively complex morphology produce most secondary metabolites. The production of these secondary metabolites usually commences late in the growth of the fungus, often upon entering the stationary phase (Sekar et al., 2008). The collected samples were grown on Czapec (dox) plates. After 5 days incubation, the results of inoculation of the sample on Czapec (dox) plates are tabulated in Table 2. It is shown that some of the following wheat samples contain fungi above or below the permissible number in terms of PFUs per gram of sample.
| Table 1: | Percentage of fungal growth |
 | |
| Table 2: | Growth after 5 days incubation on Czapec (dox) (PFUs g-1) |
 | |
| Table 3: | Colony morphology of fungi on Czapec (dox) plates and results of wet mount microscopic observation |
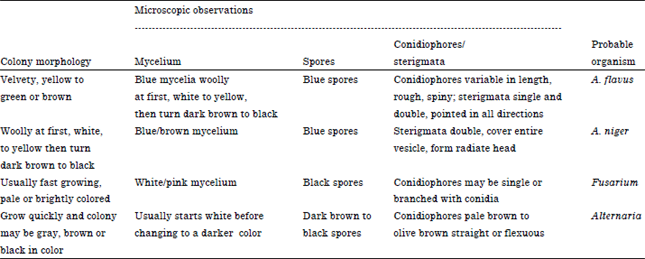 | |
For identification by morphology, LCB wet mount was prepared and the following morphologies were observed. The results of LCB wet mount preparation are shown on Table 3.
Microorganism propagules get on grain in different ways, most often with dust from soil, from the surface of plant remnants during harvesting, transportation, storage and processing (Klich, 2002). Mold spores present in wheat survive for several years and therefore, care should be taken in the storage of wheat (Christensen and Cohen, 1950). Table 2 shows the mean values of total fungal counts obtained with the direct plating technique. These results are in agreement with the results reported by Cabanas et al. (2008) in their study on wheat flour from Spanish markets. Dilution plating is the technique recommended for fungal enumeration in flours and direct plating is considered to be the more effective technique for mycological examination of particulate foods such as grains or nuts and wheat samples (ICFM, 2006; Cabanas et al., 2008). Cabanas et al. (2008) reported that the total mold counts obtained from wheat flour samples in Spain (<10-1.6; 103 CFU = g) are similar to those reported by other authors. In wheat flours, the fungal counts reported from various countries varied about 102-6; 103 CFU = g (Weidenborner et al., 2000; Berghofer et al., 2003). In Malaysia, total fungal count in wheat flour samples ranged from 102 CFU = g sample to slightly more than 104 CFU = g sample (Abdullah et al., 1998). In Spain, the maximum mold count limit for wheat flour for human consumption is 1; 104 CFU = g (Real Decreto 1286 = 1984). Recently, in the quality guidelines proposed for Australian flour the acceptable quality limit for yeasts and molds was <103 CFU = g (Berghofer et al., 2003). In Germany, 51 species belonging to 14 different genera were isolated from whole wheat flour and white wheat flour and total fungal counts of the whole wheat flour amounted to 1833 molds and the white wheat four contained 1730 CFU/2 g_1 (Weidenborner et al., 2000). In Algeria, total fungal count in wheat flour samples was 275 CFU/g_1 (Riba et al., 2008). Mycotoxins occur and exert their toxic effect in extremely small quantities in food stuffs. Mycotoxin literally means “fungus poison” and the fungi that produce mycotoxins do not have to be present to cause harm (Anonymous, 2003). The mycotoxin produced by each of these fungi may differ from each other in chemical formula, products in which they occur, conditions under which they are produced, their effects on various animals and humans and in degree of toxicity (Agrios, 1978).
| Table 4: | Validation data for extraction of wheat from HPLC |
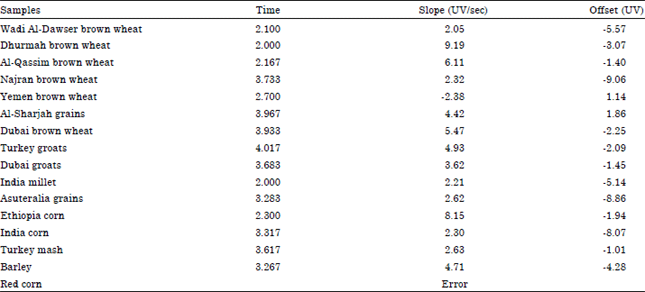 | |
Several different fungi, however, produce some of the same or closely related toxins. Fuzarium, Alternaria and Aspergillus species particularly produce similar toxins (Amadi and Adeniyi, 2009). Many of the isolates produced toxins but were not identified within the scope of this study. Validation data for extraction of wheat from HPLC are given in Table 4. This gives relevant information about the presence of mycotoxins in a run time from 2-4 min.
This study represents the investigation of toxigenic fungi occurrence in wheat and other grains. Alternaria were the most common group of encountered fungi and Aspergillus and Fusarium could be isolated from more than half of the tested samples. Their presence together with that of other potentially toxigenic molds indicates a risk of mycotoxins contamination and demands an exhaustive survey of the mycotoxins occurrence as well as the elaboration of specifc regulations for this commodity (Storari et al., 2012). The monitoring and elimination of mycotoxin in consumer products should be implemented and frequently conducted by the industry and government in order to bring the consumers and manufacturers to the concerns of quality and public health (Hong et al., 2010).
The samples of imported wheat in Saudi Arabia, it was found that samples containing common Alternaria, Aspergillus and Fusarium. It was detected fungi produce toxins device by HPLC and results indicated that the fungi are mostly produced aflatoxin, so verify their existence before consumption in order to preserve the health of the consumer.
We take our pleasure in expressing our sincere thanks to Eshrag Alsuhaibany Ghaida Alnughaimshi and Meznah Almusallam, Department of biology, College of Science.