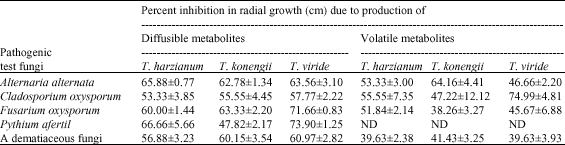
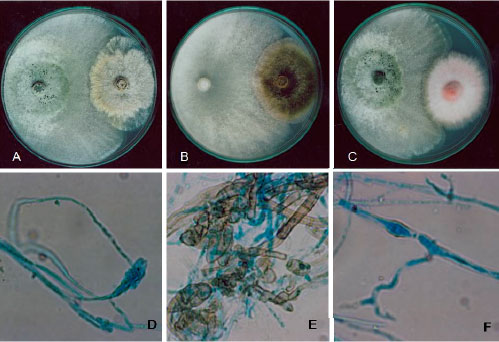
Research Article
Isolation of Cold Tolerant Antifungal Strains of Trichoderma sp. From Glacial Sites of Indian Himalayan Region
Biotechnological Applications Group, GB Pant Institute of Himalayan Environment and Development, Kosi-Katarmal, Almora, Uttarakhand, 263 643, India
A. Pandey
Biotechnological Applications Group, GB Pant Institute of Himalayan Environment and Development, Kosi-Katarmal, Almora, Uttarakhand, 263 643, India