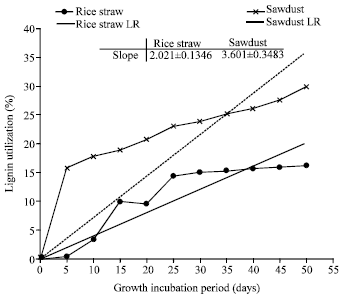
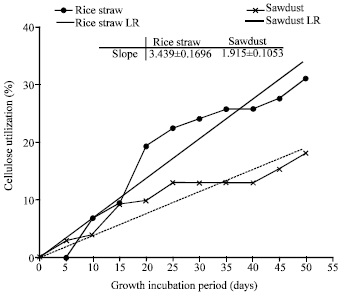
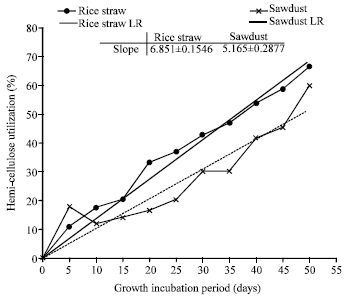
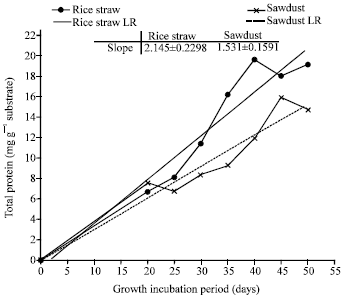
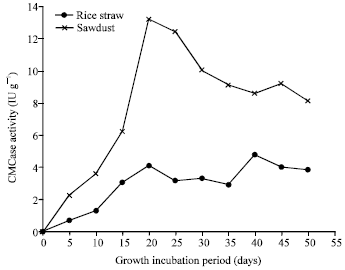
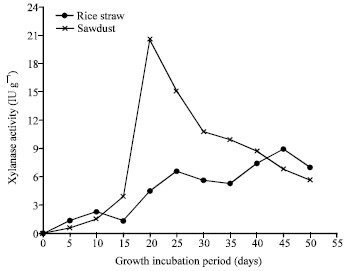
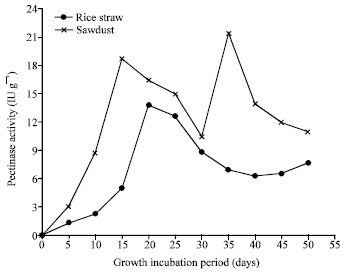
Research Article
Lignocellulolytic Enzymes and Substrate Utilization During Growth and Fruiting of Pleurotus ostreatus on Some Solid Wastes
Department of Botany, Faculty of Science, Mansoura University, Egypt
A. B. El-Tanash
Department of Botany, Faculty of Science, Mansoura University, Egypt
A. M. Temraz
Department of Botany, Faculty of Science, Mansoura University, Egypt