Research Article
Histopathological and Ultrastructural Changes in Renal Corpuscle of Female Rats Topical Application by P-phenylene Diamine
Department of Biology, Science Faculty for Girls, King Abdulaziz University, Jeddah, Saudi Arabia
Para-phenylene Diamine (PPD) is a common ingredient in most of the hair dye preparations used for accelerates the process of dyeing and can caused local toxic effects or systemic when applied topically and/or ingested (Singh et al., 2008). PPD is derivative of aniline aromatic amine, a colourless solid when pure. It is used primarily as a fur and hair dye and as a chemical intermediate in the production of numerous substances, including dyes and polymers (HSDB, 2009). Previous studies by Chugh et al. (1982) and Hashim et al. (1992) reported that the PPD frequently used by women for beauty in Africa and the Middle East and Indian sub-continent.
Hashim et al. (1992) demonstrated that, PPD is mixed with henna leaves of Lawsonia alba, as color enhancement to decorate the hands and feet in special Sudanese social events, such as wedding ceremonies. Whereas, a serious PPD intoxication problems were recorded in Morocco and India due to the most popular hair dyes contain PPD, among other ingredients (Anuradha et al., 2004; Filali et al., 2006). Similarly, Kumar (2010) revealed that some product sold as henna also contains PPD P articularly black henna.
Moreover, Singh et al. (2008) found that acute exposure to high level of PPD may cause severe dermatitis, eye irritation and tearing, Asthma, renal failure, vertigo tremors, convulsions and coma, while ingestion of PPD produces rapid developments of edema of face, neck, pharynx tongue and larynx with respiratory distress which often needs tracheostomy. As also stated by Kumar (2010) in the later stages Rhabdomyolysis and acute tubular necrosis with acute renal failure and hepatic failure develops, accompanying by bleeding tendency, subcounjunctival hemorrhage and bleeding from mucus membrane.
Certain studies were investigated the dermal absorption potential of PPD in humans and animals under exposure conditions that mimicked the intended-use conditions for such hair dyes (NIOSH, 2010). Hence, under the intended-use conditions, dermal absorption of 0.54 to 2.7% in volunteers (Wolfram and Maibach, 1985; Goetz et al., 1988; Steiling et al., 2001; Hueber-Becker et al., 2004) and 2.7% in monkeys (Maibach and Wolfram, 1981; Wolfram and Maibach, 1985; Steiling et al., 2001) has been reported. Dermal absorption of 2.7% has been noted in human cadaver skin (Dressler, 1999). The degree of dermal absorption was reported as 0.93% (Steiling et al., 2001) in excised pig skin. Hueber-Becker et al. (2004) reported similar dermal absorption values, of 2.44 and 3.39% in vitro, in human and pig skin, respectively.
In Sudan, over a 10-year period (1995-2005), 3159 patients were reported to suffer from PPD poisoning; among these were 568 (18%) children below the age of 14 years (Filali et al., 2006; Hamdouk et al., 2008; Abdelraheem et al., 2009). A study from Morocco described 374 cases of PPD poisoning in adults and children over a 10-year period (Filali et al., 2006). A report from Tunisia showed similar results (Kallel et al., 2005) and a report from Saudi Arabia documented a suicide attempt with PPD in a 14 year old female (Ashar, 2003; Abdelraheem et al., 2009).
Skin exposure to chemicals available in the environment or for cosmetic purposes is a new initiative in the field of toxicology research. Thus is more important especially in the case of chemicals that find their way into the human system through the skin. Beside to, no report has been available regarding PPD chronic dermal exposure mediated kidney histopathology. Since, the most reports are pointed out the nephrotoxicity in patients and focused on the pathophysiology and clinical features of PPD poisoning (Gleason et al., 1963; Sampath and Yesudas, 2009). Therefore, the aim of this study was to evaluate toxic effects of PPD on glomerular histological, subcellular structure and kidney function in female rats.
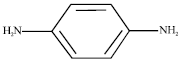
Chemical: The tested chemical P-phenylene Diamine (PPD),CAS No: 106-50-3), Batch: 99E483. Molecular Weight (MW): 108 and 98% purity was purchased from commercial markets in Jeddah of Saudi Arabia.
Chemical formula: C6H8N2 (free base), C6H8N2. 2HCl (dihydrochloride), C6H8N2. H2SO4 (sulfate) (NIOSH, 2010).
Structural formula:
 |
Physical form: White to light purple powder (NIOSH, 2010).
Animals and their treatment: According to OECD (2010a, b) guideline on the design and conduct of chronic toxicity and the Council of Europe (2006); recommendations on rodents housing, the study was conducted in Wistar rats (Rattus norvegicus albinus) 35 weeks old female rats (n = 80), with an average body weight of about 282.67±7.10 g were obtained from the Animal House of the King Fahd Center for Medical Research, King Abdulaziz University in Jeddah of Saudi Arabia. The Animals housed under a 12 h light/12 h dark cycle in stainless steel cages and allotted randomly to four groups (n = 20). Group I and II was the control groups, while groups III and IV were topical application of 0.005 or 0.001 mL (0.5 and 1 mg/kg/b.wt.), respectively of PPD dissolved in double (degassed purified water) in a shaved of approximately 10% of the total body surface area of interscapular skin for 30 min per day, 7 days per week, for a period of 6 months (OECD, 1981; Hayes, 1994; Derelanko and Hollinge, 1995; OECD, 2004). Since, the control groups received degassed purified water painted on their dorsal side as in other PPD treated animals. The rats were observed daily for signs of toxicity and for mortality. Each week they were weighed. At termination of the study all animals were sacrificed by cervical dislocation and the kidneys were excised. Absolute and relative weights were recorded at necropsy for kidneys. Macroscopic and microscopic examination of tissues from control and treated animals were performed.
Biochemical assays: At the end of the trail 24 h after the experiment, the blood samples were collected and analyzed for serum Urea Nitrogen (UN), Creatinine (Cre), Na+ and Cl¯ were determined according to the procedures recommended by the manufacturer of the kits employed according to Chromy et al. (2008) procedures.
Histological study: After bleeding, the rats were sacrificed by cervical dislocation and the abdominal cavity was opened up to expose the kidneys which were quickly dissected out and fixed in 10% formal saline for routine histological techniques. The tissues were dehydrated in an ascending grade of alcohol (ethanol), cleared in xylene and embedded in paraffin wax. Serial sections of 2 microns thick were obtained using a rotator microtome. The deparaffinized sections were stained routinely with hematoxylin and eosin (Bancroft and Gamble, 2002). Photomicrographs of the desired sections were made by light microscope for further observations.
Electron microscopy: Samples of kidney were fixed in 2.5% glutaraldehyde and 0.25 m sodium cacodylate, post-fixed in 1% osmium tetroxide and embedded in Spurr’s epoxy. Ultrathin sections were picked up on nickel grids, stained with uranyl acetate/lead citrate (Woods and Stirling, 2002) and examined in a Philips TEM 100 microscope. At least six (x 2600) fields from each of at least four glomeruli per rat were photographed.
Statistics: The experimental data were analyzed with SPSS18.0 software and performed using student’s t-test on files procedure with significance set at p<0.05. The analytical data are presented as Mean±SEM.
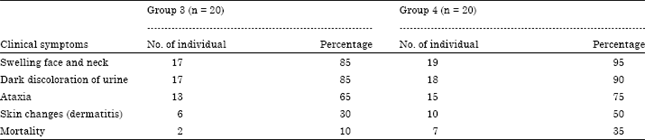
Clinical signs and mortality: As seen in Table 1, there were significant positive association between the concentration of PPD administered and mortality and clinical signs of either groups.
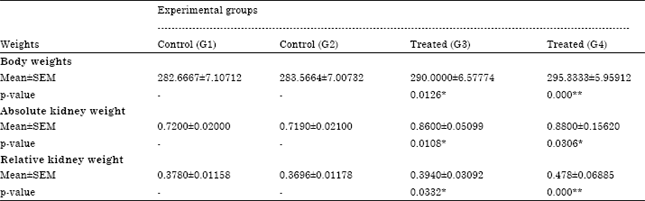
Body and kidney weights: Body weights and the mean absolute and body-weight-related kidney weights were affected by the treatment, significantly increased (p<0.05) for animals given 0.5 mg/kg/day and highly significantly (p<0.01) for group treated 1 mg/kg/day in relation to their respective controls (Table 2).
| Table 1: | Clinical symptoms and the mortality recorded in groups 3 and 4 of PDD treatment rats after 24 h-6 months |
 | |
| Table 2: | Body weight and kidney weight (absolute and relative) of experimental rats after 6 month topical application of PPD |
 | |
| *,**Significant at p<0.05 and 0.01, respectively | |
Effects of PPD on renal function: In the serum, significantly (p<0.01)increased Urea Nitrogen (UN) was present in groups III and IV and the serum of group IV had significant (p<0.01) elevated creatinine (Cre) and decreased Na+ and Cl- (Table 3).
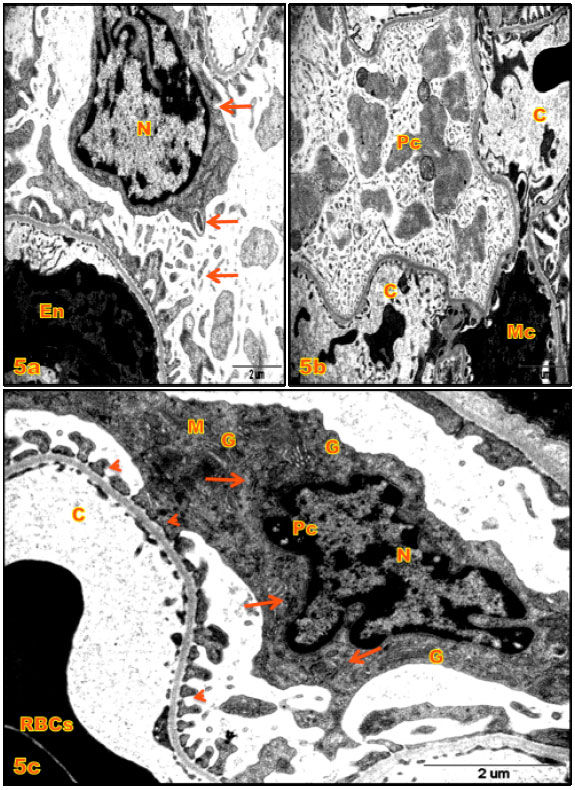
Histological results: No histopathology was observed in the control groups I and II of female rats kidneys. Cortical zone of rats kidneys displayed urinary corpuscles and glomeruli (Fig. 1a, b). Glomeruli of kidney corpuscles were made up of parietal cells and podocytes with foot processes, the Glomerular Basement Membrane (GBM), foot processes and filtration slits were observed in normal structure (Fig. 2a, 3a, b).
The results of light microscopy of 18 and 13 specimens of groups III and IV, respectively revealed microscopical damage caused by PPD treatment. Such changes an existence of hyperemia with glomerular hypertrophy associated with swelling of glomerular lining epithelium and mesangial matrix expansion in group III (Fig. 1c, d). However, lobulation of glomerular tufts with glomerular cells vacuolation as well as changes in glomerular capillaries indicated haemorrhagic lesions associated by diffused hyaline and thickening of Glomerular Basement Membrane (GBM) were seen in group IV (Fig. 1e, f).
In contrast, electron microscopic changes in the group III confirm glomeruli podocytes and mesangial cells proliferation with increase mesangial matrix (Fig. 2b, c). Severe podocytes pedicles fusion and lysis association with GBM indistinct layers with spikes and electron dense deposits on destructed endothelium side were observed (Fig. 3c).
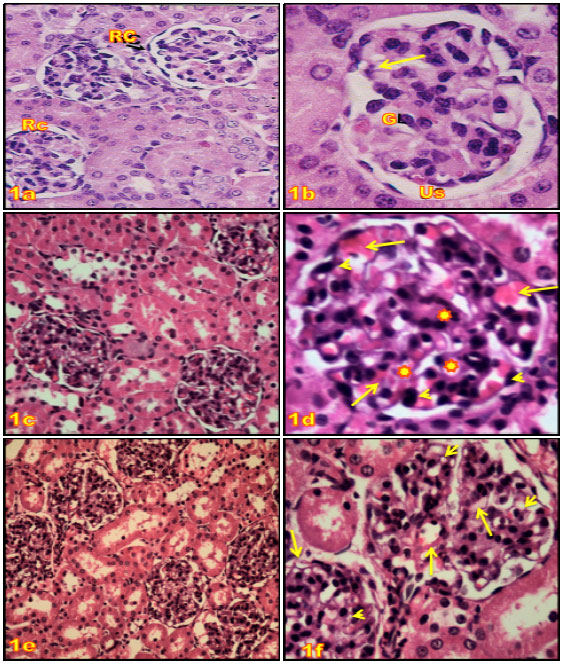 | |
| Fig. 1(a-f): | Photomicrographs of kidney sections of control groups rats (G1 and G2) (H and E); (a) Showing the normal histological structure of the renal corpuscles (Rc) and renal tubules in the cortical portion; x 400, (b): High power from previous section showing glomerulus (G) and distinct urinary space (Us); x 1000, (c, d) Sections of treated kidney (G3), (c) Showing glomeruli hypertrophy with alliterative urinary space; x400, (d) High power from previous section showing glomerulus hyperemia (arrows) with alliterative urinary space and swelling the lining epithelial (►) and mesangial matrix expansion ( |
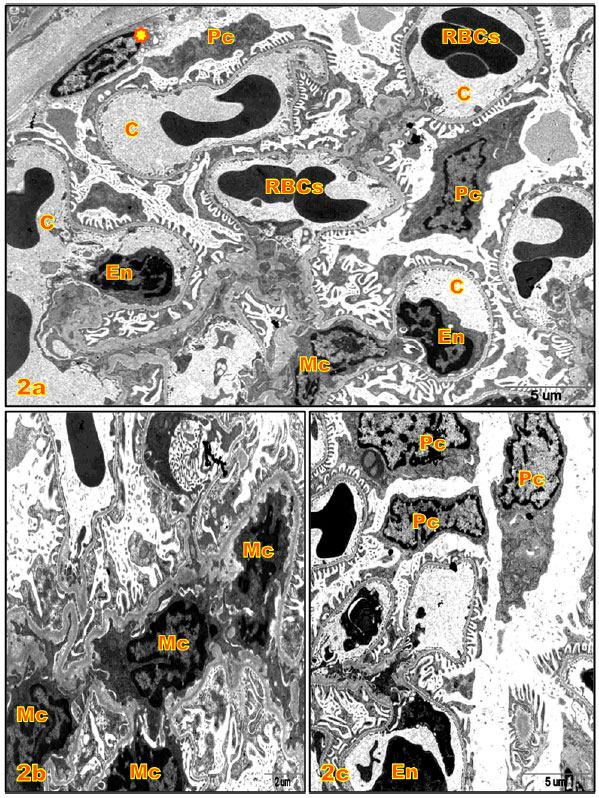 | |
| Fig. 2(a-c): | Electron micrographs (E.M.) of renal corpuscles of kidney cortex of rats; (a) E.M. of a part of renal corpuscle of control rat showing capillary loops (C) endothelial lining cells (En); RBCs; mesangial cells (Mc); parietal cell ( |
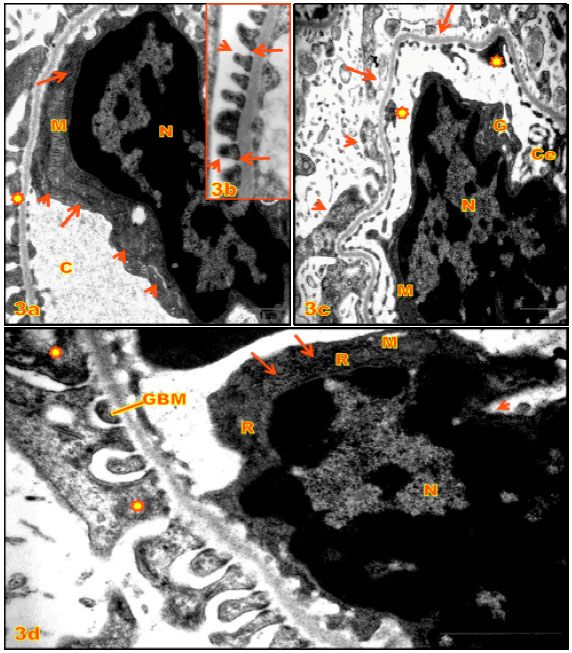 | |
| Fig. 3(a-d): | (a, b) E.M. of renal corpuscles of kidney cortex of control rats; (a) A part of capillary loops (C) showing endothelial cell with large irregular nucleus (N) and scanty cytoplasm, elongated mitochondria (M), RER (arrows), Golgi bodies (►); glomerular basement membrane ( |
| Table 3: | Serum creatinine and serum urea of experimental animals kidney after 6 month topical application of PPD |
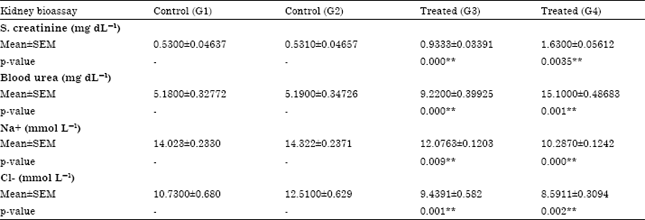 | |
| *p<0.05, **p<0.01 | |
Moreover, marked endothelium nucleus swollen accompanied by inactive chromatin condensation, nuclear membrane ruptured, mitochondrial damage and scatter of RER ribosomes (Fig. 3d) as comparing to control groups (Fig. 3a, b).
On the other hand, PPD caused severe various renal corpuscles histological changes in group IV, in general, necrotic of glomerular podocytes, mesangial cells and endothelial cell with detachment accompanied by floppy and vacuolated of cytoplasmic extension. GBM deterioration and lysis led to capillaries collapsed and filled with deformed Red Blood Cells (RBCs), neutrophils, macrophages and cellular debris were been observed (Fig. 4a-c),which suggests opening of the endothelial junction of the glomerular vessels. Moreover, the podocytes manifested more intensive deterioration than those observed in group III which indicated affected and deformed nucleus, pedicles ruptured lead to cytoplasmic lysis with organelles damage (Fig. 5a, b) as comparing to control (Fig. 5c).
Hair dye containing PPD is widely used in Saudi Arabia for hair coloration and is added to Henna to accentuate the colour when used on the skin. The toxicity of PPD occurs through skin absorption. A number of earlier report suggested that a fraction of topically applied PPD alone or in combination with an oxidizing agent reach systemic circulation after percutaneous absorption (Steiling et al., 2001). The lethal dose of PPD is not known; estimates vary from 7-10 g (Kumar, 2010). From the present results it was evident that the treatment of PPD caused a swelling face and neck, dark discoloration of urine, ataxia and mortality in rats of treated groups III and IV, all these clinical sings were matched with Filali et al. (2006) observed. In addition to, the characteristic chocolate brown colour of the urine, swelling of head and neck and ataxia in current study could be confirmative evidence of PPD poisoning in female rats and were matched with many previous studies reported oedema of head and neck experimentally in rabbits and cats by intraperitoneal injection of PPD HCI (Gleason et al., 1963). Urine discoloration and ataxia in male rats by PPD topical application (Waggas, 2011). PPD causes contact dermatitis in susceptible individuals (Filali et al., 2006). Allergic reactions causing dermatitis, urticaria and asthma have also been reported (Nethercott et al., 1986; Seidnari et al., 1990; Abdelraheema et al., 2010).
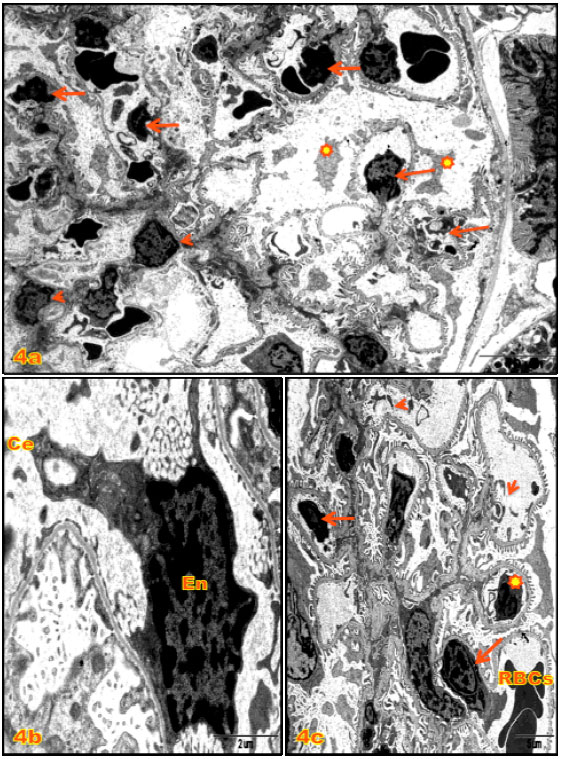 | |
| Fig. 4(a-c): | Electron micrographs (E.M.) of renal corpuscles of kidney cortex of 1 mg kg-1 PPD treated rats (G4); (a) Showing necrotic of glomerular podocytes ( |
 | |
| Fig. 5(a-c): | (a,b) Electron micrographs (E.M.) of renal corpuscles of kidney cortex of 1 mg kg-1 PPD treated rats (G4), (a) Showing necrotic endothelial cell (En) and podocyte with deformed nucleus (N) and pedicles ruptured (arrows); x7900, (b) Showing cytoplasmic lysis of the podocytes (Pc) with organelles damage; x7900, (c) E.M. of podocytes (Pc) of control rat showing intact pedicles (►) irregular nucleus (N), mitochondria (M), RER (arrows) and Golgi apparatus (G); x10500 |
Mortality was 10 and 35% in groups III and IV, respectively, these findings are in agreement with previous reports concerning male rats, mortality was 21.1% in study by Filali et al. (2006) and 41.9 in study by Kumar (2010) and 22% in study by Yagi et al. (1991). The mortality in present study probably because of prolong PPD exposure period. Hence, Prasad et al. (2011) reported the most common cause of death in patients is acute renal failure which occurs as a result of rhabdomyolysis due to the nephrotoxic components of PPD. Rhabdomyolysis is the main cause of acute renal failure and the morbidity and mortality are high once renal failure develops (Sampath and Yesudas, 2009).
Changes in serum UN, Cre, Na+ and Cl¯ are associated with impairment of renal function (Kumar et al., 1988). Also the present investigation show that chronic frequent PPD exposure is deleterious to kidney structure and function of female rats. The elevated serum levels of UN and Cre and the decreased serum levels of serum Na+ and Cl¯ indicate reduced ability of the kidney to eliminate the toxic metabolic substances and reabsorb the metal and non-metal ions. These changes in serum biochemical parameters are consistent with renal histological structure degenerations which are in agreement with previous reports concerning male rats (Waggas, 2011). Moreover, there are some reports have been pointed out the changes in the biochemical profile in patients who have consumed the hair dye containing PPD (Ashraf et al., 1994; Sampathkumar et al., 2007; Gandhe et al., 2009; Chrispal et al., 2010) and that biochemical changes reflect tissue damage (Prasad et al., 2011).
On the other hand, body weights and the mean relative kidney weights were significantly increased by the PPD treatment in present study. Bharali and Dutta (2009) study showed both absolute and relative liver weight of rats exhibits a dose response increase after PPD treatment. Another studies revealed the topical application in both sexes of Wistar rats at dose level of 5 mg/kg/day or greater, mean absolute and relative liver and kidney weights raised in males rats (Wakelin et al., 1998). However, that PPD administered to Wistar rats with 0.05 mL once a week for 18 months, resulted in a slight decrease in the body weights of the males after 30 weeks of exposure and no such effects were found among the females (Rojanapo et al., 1986). A 13 week study was conducted in Crl: CD(SD)BR rats, the body weights and body weight gain were unaffected by PPD treatment and the mean absolute and body-weight-related kidney weights were significantly increased of females given 8 and 16 mg/kg/day (Toxicol Laboratories Ltd., Herefordshire England, 1995).
Results presented here suggest that PPD lead to moderate and severe microscopic and ultra-structural glomerular degenerative changes in both treated groups III and IV. These observations were agreement with Prasad et al. (2011) who confirmed that the degree of the tissue damage is related to the dose of the poison. Suliman et al. (1995) indicated that chronic application of the PPD to the skin lead over many years to glomerular injury and glomerulosclerosis in human. In a study of 19 patients who poison with PPD and underwent renal biopsy, glomerular injury was the most common (94%) finding, followed by interstitial lesions (84%) and tubular lesions (79%) (Abdelraheem et al., 2009).
The cause of renal injury is probably direct nephrotoxicity of PPD compound (Kumar, 2010). PPD induces over production of free radicals subsequently results in lipid peroxidation. The lipid peroxidation is initiated by an existing free radical, by light, metal ions or by toxins (Sutrapu et al., 2010). The metabolic products of PPD have a high urinary excretion rate and their oxidation produces quinone-diamine is a potentially nephrotoxic substance (Ashraf et al., 1994). Likewise, Malondialdehyde (MDA) is one of the aldehyde products of lipid peroxidation (Sutrapu et al., 2010). Experimental studies in guinea pigs dermal exposed to PPD revealed an increased in MDA which indicates lipid peroxidation, suggesting that increased free radicals formation is responsible for histopathologically tissue damage in many organs and nephrotoxicity (Picardo et al., 1992; Bhargava, 2008; Sutrapu et al., 2010; Waggas, 2011). Hence, the results of this study suggest that PPD causes glomerular hypertrophy, capillaries congestion associated by diffused hyaline and thickening of GBM. Similarly, the PPD mediated hepatotoxicity is evident from histological observation (hypertrophy, hyperplasia of portal tract, hepatocytes necrosis, hemorrhages, fibrin deposition within central vein and around the hepatic cords, fibrinous exudates as well as inflammation of the portal tract (Bharali and Dutta, 2009).
The present study indicates that the selected dose of PPD on chronic topical exposure causes glomerular podocytes, mesangial cells and endothelial cell necrosis in treated group IV, are consistent with similar observations on skeletal and cardiac muscle necrosis was experimentally induced by N-methylated PPD in rats (Ashraf et al., 1994; Munday et al., 1989). Coagulation necrosis of skeletal muscles was shown in autopsies in cases of PPD poisoning (Prasad et al., 2011) and in guinea pigs (Chugh et al., 1982; Munday et al., 1989). Liver necrosis has also been reported in PPD poisoning cases (Singla et al., 2005).
Correspond to present work, the previous studies have reported ultrastructural changes in rats glomeruli at treatment with various pollutants, pesticides and toxins represented in glomerular cells such as swollen of endothelial nuclei (Abd El-Aal and Fares, 1993) mesangial cells proliferation (Ramadan, 1996), podocytes vacuolation with pedicles fusion and GBM indistinct layers with electron dense deposits (El-Banhawy et al., 1994; Rahmy and Hemmaid, 1999; Ahmed, 2000), smooth endoplasmic reticulum proliferation, mitochondrial, Golgi apparatus, RER damage with nuclei pyknosis (Ramadan, 1996; Luty et al., 1998; Latuszynska et al., 1999; Rahmy and Hemmaid, 1999). PPD promotes calcium release and the leakage of calcium ions from the smooth endoplasmic reticulum, thus causing irreversible change in the endoplasmic reticulum structure (Chugh et al., 1982; Prasad et al., 2011). PPD also damages function of cell membranes and mitochondrial membranes, ribosomes, membrane receptor sites and cell death induced by DNA fragmentation and lipid peroxidation (Waggas, 2011). Furthermore, Sutrapu et al. (2010) demonstrated that PPD breaks single strand DNA in human lymphocytes.
Meanwhile, acute and chronic nephrotitis under dermal applied toxic substances is pathologically characterized by a prominent infiltration of neutrophils and macrophages (Luty et al., 1998; Latuszynska et al., 1999) and this histological feature is predominantly found in the glomerular tissue of the PPD treated rats in recent study. This confirms the PPD auto-oxidation that increased free radicals (superoxide and hydrogen peroxide) formation. Free radicals play an important role in several pathological conditions through multiple mechanisms, including excitotoxicity, metabolic dysfunction and disturbance of intracellular homeostasis (Munday, 1992; NIOSH, 2010).
These studies have shown that PPD causes various glomerular histological and subcellular structure changes, resulting in impairment of renal function and metabolism. These results therefore provide valuable insight on the effects of chronic PPD exposure in rats.
chikwuogwo w.Paul Reply
I cannot ignore this Master piece that Laila A.Hummadi produced.