ABSTRACT
Influenza virus capsid contains surface glycoproteins and virus takes the help of topically available proteases to enter into the host cells. As the proteases and the virus capsid glycoproteins are proteins in nature, the aim of this study was to test some tannin rich plant extracts, either alone or in association, so as to neutralize proteases as well as virus glycoproteins to stop further infection. MDCK cells were infected with influenza virus in the presence or absence of MMP (Matrix Metalloproteinases) proteases and various plant extracts using cytopathic virus concentrations to evaluate the role of MMP proteases and tannin rich plant extracts on virus growth in vitro. Results indicate that plant tannins can bind to specific viral proteins and proteases but requires highly specific associations to inhibit total virus activity. These results prove that specific proteases are involved in influenza virus infection and simultaneous neutralization of virus glycoproteins and proteases constitute the most promising approach to treat influenza infection.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijv.2011.53.63
URL: https://scialert.net/abstract/?doi=ijv.2011.53.63
INTRODUCTION
Influenza, commonly referred to as the flu, is a RNA virus, particularly known for causing sore throat and pneumonia in human beings. The virus is transmitted through the air by coughs or sneezes, creating aerosols containing the virus but also through nasal secretions, or through contact with contaminated surfaces causing severe pandemics (Sar et al., 2010).
The mode of progression of a topical viral infection is completely different than a systemic viral infection. During a topical external infection such as the influenza virus, initially a few virus particles come in contact with the cells of the throat mucosa. There are practically no clinical signs at this stage. After initial infection, the virus multiplies in a few cells and millions of new virus particles are than liberated topically to infect new cells and to create visible lesion (Mehle and Doudna, 2010).
Influenza virus has no processing proteases to fuse with the host cell membrane and the virus entry is determined primarily by the host cellular HA (0) processing proteases that proteolytically activate membrane fusion activity (Al-Majhdi, 2007; Tambunan et al., 2008). Matrix metalloproteinases (MMPs) belong to a large family of proteases. At least seven different trypsin-type processing proteases including tryptase Clara and tryptase TL2 have been identified for HA (0) processing but probably there are many others which are not yet identified (Kido et al., 1996, 2008; Delboy et al., 2008). Intracellular virus multipication also encodes up to 11 proteins and this coding capacity demands that the virus use the host cellular machinery for many aspects of its life cycle (Konig et al., 2010), including the help of different intracellular proteases.
This virus therefore, uses some specific MMPs present on the surface of the respiratory tract to enter and to infect throat cells. To restrict viral infection, our body defense mechanisms liberate anti-proteases called secretory leukoproteases in the upper respiratory tract and the pulmonary surfactants in the lower respiratory tract to reduce the amount of free proteases available for viral entry. When proteases activity predominant over the activities of inhibitory compounds, virus infection cannot be stopped (Beppu et al., 1997). Body defence mechanisms are activated to produce antibodies and to stop virus replication but it normally takes 5-10 days. This is the reason why proteases inhibitors are considered as potential future therapeutic agents for the treatment of influenza virus (Kido et al., 2007).
Proteases, also known as proteinases or proteolytic enzymes, are a large group of enzymes found in or outside the cells, particularly in the vicinity of the damaged tissues and play a vital role in protein catabolism by hydrolysis of the peptide bonds that link amino acids together in the polypeptide chain. They are involved in the splitting of the protein molecules. Their main role is to break and to clean the proteinous debris generated during the tissue breakdown because such substances interfere with the tissue repairing process. They are essential to create a favorable environment for subsequent tissue repairs (Ajlia et al., 2010).
According to the mode of action, proteases are divided into four major groups as metalloproteinases or Matrix-Metallo-Proteins (MMPs), serine proteinases, cysteine (thiol) proteinases, and aspartic proteinases but their exact number is not yet known as new proteases are being discovered regularly (Vanaman and Bradshaw, 1999).
Many proteases may be found topically on a virus infected skin or mucus membrane such as MMPs, pepsin, trypsin, chymotrypsin, subtilisin, cystin proteinase (cathepsin B,H,K,L,S), aspartic proteinase (cathepsin D) and clotting factors (R 26, R27). MMPs are particularly abundant in skin wounds as they are involved in the process of topical wound healing. Several proteases have already been identified to mediate influenza virus entry (Yamada et al., 2006). Therefore, identifying the MMPs which are involved in influenza virus infection and their subsequent inhibition may constitute a logical solution to block virus entry into the cells. As there are multiple types of proteases, a non-specific protease inhibitor is essential so as they block all the proteases present on an infected throat surface.
One another way to block virus entry into the cells is to directly neutralize the virus particles on the infected surface. Influenza virus contains several glycoproteins on the surface coat and blocking viral glycoproteins may help to neutralize the infectivity of the whole virus particle (Ali et al., 2000).
As all the proteases which help influenza virus entry into the cells are proteins in nature and as virus surface glycoproteins are also protein in nature, the aim of this study was to identify the main MMPs involved in topical influenza virus infection and to test some plant extracts rich in tannins, for their virus or protease binding properties in vitro.
Plant extracts rich in tannins were selected as antiviral test products because tannins are specific with respect to their protein binding properties and each plant contains a wide variety of tannins (Frazier et al., 2010). Taking into consideration the variety of proteases and virus surface glycoproteins involved in influenza virus entry and the need to block all the proteins simultaneously, plant extracts were also associated with each other to maximize the anti-viral effects.
MATERIALS AND METHODS
Selection of plants and preparation of plant extracts: One hundred and thirty one tannin rich known plants were selected and plant extracts were prepared using the part of the plant which contains maximum amount of condensed tannins. This research was performed between April 2007 up to November 2011.
As hydrosoluble tannins are not suitable for topical application, plant extracts were enriched in condensed tannins. Briefly, initially the plant material was mixed with water at 1:12 solid to water ratio and stirred between 200-300°F at 2-50 psi pressure for 1 h. The aqueous solution obtained was than filtered through a polymeric membrane to remove large particles. The solution was purified by adding 3% bentonite w/w and removed by filtration. The extract was than kept in contact with an adsorbent material (non ionic resin packed in a column) and the tannins retained on the adsorbent material were eluted with a polar solvent. The concentrated tannins extract was then dried by atomization (120°C) to obtain a tannin rich dried and soluble plant extract. The percentage of tannin in the dried extract varied between 15-34% depending upon the initial richness of the plant in tannins and the part of the plant used. These dried extracts were solubilised in water (10 mg mL-1) for all the experiments. Individual plant extracts were tested at a concentration of 50 μg mL-1 and half the concentration of each plant extract (25 μg mL-1) was used to prepare association of plant extracts.
Initially, the maximum non-cytotoxic concentrations of each plant were determined by exposing different type of cells with plant extract concentrations ranging between 5 to 2000 μg mL-1 to verify that the selected extracts are not cytotoxic at least up to a concentration of 50 μg mL-1 in the culture medium.
On the basis of initial results, when a plant extract was found active, different species of plants extracts in the same family as well as associations of two or more active plant extracts were also checked for their activity.
Cell cultures: Cell culture models where cells remain exposed to the external environment were used to mimic topical viral infections. MDCK (Madine Durby monkey kidney) cells sensitive to influenza virus infection were initially grown in 75 cm2 tissue culture flasks (Corning, USA) in Dulbecco’s Modified Eagle’s medium (DMEM, PAA cell culture, France) supplemented with 10% Fetal Calf Serum (FCS) and antibiotics. All cells were cultivated at 37°C with 5% CO2 employing standard cell culture methods as described elsewhere (Shankar et al., 2009; Shrivastava et al., 1993; Matlin et al., 1981).
For experiments, cells were grown in 96-well tissue culture plates. Once a cell monolayer of desired confluence was obtained, further experiments were conducted using specific cell culture Medium devoid of proteases (MMPs) in a serum free medium.
Virus source: Cells and viruses were purchased from American Type of Culture Collection (ATCC), USA.
Selection of proteases: In the absence of complete knowledge regarding the proteases involved in facilitating viral entry into the cells, initial screening was conducted with individual MMPs to check whether a specific MMP is involved in virus growth. Similarly, associations of MMPs were also studied and the best association of MMPs enhancing maximum virus growth was used for further experiments.
The purified human MMPs (proteases) and the protease assay kits were purchased from AnaSpec, Inc, USA. The MMP 1 (interstitial and fibroblast collagenase, Ref.72004 and 71128); MMP 2 (gelatinase A, Ref. 72005 and 71151); MMP 3 (stromelysin-1, transin1, Ref. 72006 and 71153); MMP7 (proenzyme, Ref. 72007), MMP-9 (gelatinase B, collagenase IV, Ref. 72009 and 71134), MMP-10 (stromelysin 2 Ref. 72067 and 72024) and MMP-12 (elastase, Ref. 72010 and 71137), were used at concentration of 0.5 μg mL-1 either with the viral suspension or in the cell culture medium.
Virus titer: To determine 50 or 100% virus tissue culture infective dose (TCID50 and TCID100), MDCK cells were grown in confluency in 96-well tissue culture plates, washed with PBS and then infected with 10-fold virus dilutions in a serum free and MMP free tissue culture medium. Cells were then incubated at 37°C for 1 h. After initial incubation with virus, inoculum was removed; cells were washed again with PBS and further incubated for 72 h with fresh medium. Cell death was evaluated with MTT vital staining. Virus titer was also evaluated using standard influenza virus haemagglutination assay. Plates were kept for 24 h at -20°C, thawed to induce cell lyses, centrifuged (500 g for 5 min), 50 μL sample of supernatant was drawn from each well, transferred to a new 96-well plate and virus was titrated by haemagglutination (HA) test with a 0.5% suspension of chicken red blood cells. The TCID doses were calculated by the method of Reed and Muench (1938).
Research for the proteases involved in viral entry into the cells: To search the MMP proteases involved in virus entry into cells, TCID50 concentration of virus was preincubated with 0.5 μg mL-1 of corresponding MMP in a protease and serum free medium for 1 h. MDCK cell cultures were then exposed to this mixture and cell cultures were further incubated for 72 h at 37°C to allow virus growth. The percent increase in the virus growth (indicating a helping role of MMP in viral entry) was determined for each type of MMP or MMP association.
If the virus growth was identical to the corresponding non-MMP added virus controls, the MMP was considered not involved in the process of viral entry into the cells but if the virus growth was superior to the corresponding non MMP added virus controls, the percent increase in virus growth was determine to evaluate the extent of MMP or the association of MMPs involvement in virus entry into the host cells. Values represent mean of minimum 3 experiments±SEM.
Evaluation of the anti-protease activity of plant extracts: The in vitro anti-protease activity of different tannin rich plant extracts was adapted from the method described by Diaz-Nido et al. (1991). In short, a fixed concentration of each plant extract (50 μg mL-1) or an association of plant extracts (25 μg mL-1 each) was pre-incubated for 1 h in a test tube with the influenza virus growth promoting MMPs (MMPs 1, 2, 7, 9) at a concentration of 0, 5 μg/mL/MMP. After 1 h pre-incubation, the plant extract-MMP suspension was exposed to cell cultures pre-infected before 1 h with TCID100 concentrations of influenza virus. The cells cultures were washed before exposure to ensure that there are no free virus particles on the surface of the cultures. The untreated cells served as cell controls while only virus treated cells served as virus controls.
Result interpretation: It was assumed that if the plant extract neutralize the MMPs, the protease will not be available for virus entry into the cell thereby reducing the virus infectivity and consequently the cell death.
Virus glycoprotein inhibiting properties of the plant extracts alone or in association: The experimental procedure was similar to the procedure followed for the evaluation of the anti-protease activity of the plants with the exception that influenza virus (TCID100 concentration) was pre-incubated with the plant extracts and the MMPs were added in the cell culture medium only during the phase of virus replication. Untreated cells and only virus treated cells served as negative and positive controls.
Virus glycoprotein neutralization was evaluated by quantifying virus titer with haemagglutination test to quantify reduction in the viral growth, which was proportional to the virus glycoprotein neutralization.
Total anti-viral activity of individual plant extracts or the association of plant extracts: The antiviral activity was determined using a slightly modified method as described by Xiao et al. (2008). In short, a fixed concentration of an individual plant extract (50 μg mL-1) or a synergistic association of plant extracts (each 25 μg mL-1 each) was pre-incubated for 1 h before cell exposure with a fixed concentration of MMPs (0.5 μg mL-1) and herpes virus (TCID100). Percent mean reduction in cell death indicating reduction in virus growth compared to the corresponding controls was calculated to determine the total anti-viral activity.
Data analysis: All the experiments were conducted in triplicate (n = 16 wells per experiment) and the mean percent change (±SEM) in cell viability or virus growth compared with the corresponding controls was determined. In each case, statistical analysis was performed with one-way ANOVA with the paired student’s t-test. Inhibitory activity is expressed as percent increase in cell survival, which was proportional to the reduction in virus multiplication.
RESULTS
The results in Table 1 clearly show that the addition of certain MMPs in the cell culture medium of cells infected with TCID50 of influenza virus stimulates the growth of virus. Addition of 0.5 μg mL-1 of MMPs 2, 7 and 9 increased the growth of influenza virus by nearly 50% while with MMP1, the virus growth was increased by 63% compared to non-MMP added virus controls. An association of MMP 1, 2, 7 and 9 markedly stimulated the virus growth increasing the amount of virus produced by 81% (±8.78) compared to the corresponding to the controls. On the contrary, MMP 3, 10 and 12 seems not involved in enhancing influenza virus host cell infection.
| Table 1: | Mean % cell death, proportional to the role of MMP in virus growth, compared to the non-MMP treated virus controls in vitro |
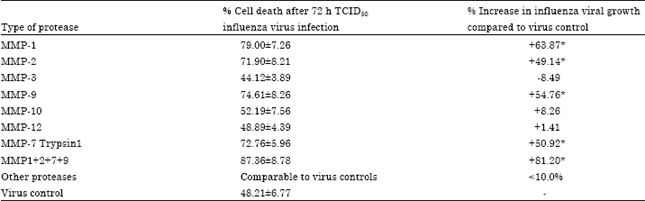 | |
| *p<0.05 compared to the controls | |
On the basis of these results, an association of MMPs 1, 2, 7 and 9 was used (0.5 μg mL-1 each) to conduct further experiments as influenza virus MMPs.
The mean results (±SEM) of the three experiments (minimum n = 48) for individual plant extracts pre-incubated either with influenza virus selected MMPs or with the virus suspension or with MMPs and virus suspension to evaluate antiprotease, antivirus glycoprotein or total antiviral activity, respectively are shown in the Fig. 1.
Among 131 plant extracts tested, 123 had no effect on the virus glycoprotein or proteases (results not shown) as only the extracts of E. purpurea and C. sinensis had some anti-protease activity while the extracts of Vaccinium macrocarpon, Vitis vinifera, A. hippocastanum, S. officinalis and S. nigra showed slight binding with the influenza virus glycoproteins. The extract of Centella asiatica was not very active on these two parameters but had showed some total antiviral potential. None of these individual plant extracts was able to inhibit virus growth above 43%. We observed that all the 8 plant extracts had simultaneously antiprotease and virus glycoprotein binding properties and that their total antiviral activity is always higher and not additive than their antiprotease or virus neutralizing activity.
When two or more plant extracts were associated at non-cytotoxic concentrations of 25 μg mL-1, none of the associations inhibited markedly the protease activity or virus glycoproteins but surprisingly the association of certain plant extracts comprising the extracts of Vitis vinifera, Echinacea purpurea, Vaccinium macrocarpon, Sambucus nigra and Camellia sinensis showed very significant total antiviral activity (Fig. 2).
As shown in the Fig. 2, best results were obtained with the association of Green tea+Vitis vinifera+Sambuscus nigra and the associations of Vaccinium macrocarpon with Sambucus nigra tannins which inhibited on an average only 30-40% MMP activity or about 30% virus glycoproteins but above 90% total virus activity. These results are not additive and prove that plant extracts are specific with respect to their anti-protease and anti-virus glycoprotein inhibiting properties and only specific associations of certain plant extracts are capable to block total virus activity.
 | |
| Fig. 1: | Mean % inhibition of protease activity, virus glycoprotein and total influenza virus activity with individual (I) plant extracts at a concentration of 50 μg mL-1. All experiments represent means of at least 3 experiments±SEM (n = 16 per experiment). Ep: Echinacea purpurea, Cs: Camellia sinensis, Vm: Vaccinium macrocarpon fruit, Vv: Vitis vinifera seed, Ca: Centella asiatica, Ah: Asculus hippocastanum, Sa: Salvia officinalis, Sn: Sambucus nigra. Other individual plant extract showed activity <10% (results not shown) |
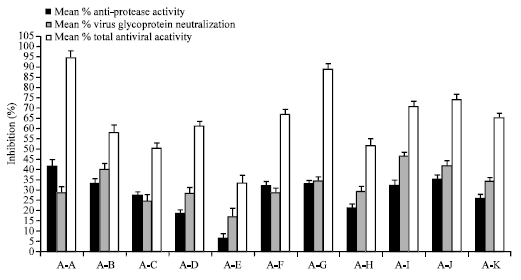 | |
| Fig. 2: | Mean % inhibition of protease activity, virus glycoprotein and total influenza virus activity with best plant extract associations (A) tested as a concentration of 25 μg mL-1 each. All experiments represent means of at least 3 experiments±SEM (n = 16 per experiment). AA = Gt+Vv+Sn; AB=Ep+Cs; AC= Sn+Cs; AD = Ep+Sn; AE = Vv+Cs; AF = Vv+Ca; AG = Vm+Sn; AH = Gt+Sn; AI = Ep+Vv+Sn; AJ = Ep+Vv; AK = Gt+Vv. Other associations of plant extracts tested showed total virus inhibition <33% compared to the controls (results not shown) |
DISCUSSION
Influenza spreads around the world in seasonal epidemics, resulting in the deaths of between 250,000 and 500,000 people every year and millions in pandemic years. As this infection primarily involve throat mucosal cells, virus multiplication generates a large amount of free virus particles all over the throat surface. These free virus particles continue attacking new healthy cells and maintain the infection up to the time body defense mechanisms are not activated to neutralize the viruses. The differences in the surface glycoprotein structures confer different morphology and antigenicity in the same family of virus and help the virus to enter the host cell (Soltanialvar et al., 2010; Kido et al., 2008). For example, three different types of influenza virus, dubbed A, B and C have been identified with HA (hemagglutinin) and NA (neuraminidase) as main surface glycoproteins. Thirteen major types of HA and 9 major antigenic determinants of NA have already been identified. This shows that the viral capsid may contain a very large variety of glycoproteins on the surface coat. Furthermore, all the viral glycoproteins are not yet discovered and there is continuous research on the presence of new virus glycoproteins and their role in viral multiplication and infection. This is the reason why it is practically impossible to develop a specific viral glycoprotein inhibiting drug to treat topical viral infection (Murineddu et al., 2010).
Continuous infection by the free virus particles present on the throat surface weakens local immunity and creates a favorable ground for a secondary bacterial infection. In the absence of any specific topical antiviral drug, all treatment efforts are directed to stop secondary bacterial infection, to reduce throat pain, to decongest nasal passage or to stop other symptomatic manifestations of flu without interfering with the basic cause of influenza. An ideal treatment should neutralize all the virus particles on the throat surface so as to stop further virus growth and the infection but neutralizing a constantly mutating virus is not very easy (Leneva et al., 2009). Due to the complexity of the structure of the influenza virus with various surface glycoproteins, vaccines are also not very effective and require regular antigenic update (Konig et al., 2010).
In the absence of processing structure to enter into the host cell, influenza virus takes the help of processing proteases to enter into the cells. Different type of proteases are found in and around a damaged tissue as they are primarily involved in cleaning the damaged surface, in tissue remodeling and in the wound repair process but the amount of MMPs is particularly high on the virus infected surfaces. Multiple cell types, including macrophages, fibroblasts, neutrophils, epithelial cells secrete proteases during early inflammatory stage and endothelial cells following specific biochemical signals such as inflammatory cytokines or the presence of bacterial endotoxins.
Although the role of many MMP proteases is still remains unknown (Hayden and Pavia, 2006), their role in intracellular virus entry is proven. Therefore, the use of protease inhibitors to block the virus entry into the host cells is an ideal approach to stop topical viral infection but there are many proteases and no chemical entity is yet discovered which can block all the proteases at a time (Gupta and Kumaran, 2006).
Currently available targets for antiviral therapy include almost exclusively the intracellular antiviral agents which are often used in combination to maximize efficiency (Hsieh and Hsu, 2007). Most of these drugs are used orally and target the viral enzymes or intracellular proteases (Hsieh and Hsu, 2007; Alymova et al., 2005). For example, intracellular nuraminidase inhibitors are used to treat the influenza, A, B and C type of enveloped viruses with 2 main classes of drugs by oral route: the adamantanes which interfere with viral uncoating inside the cells and are effective against influenza A type of viruses only while the newer class Zanamivir or Oseltamivir (Tamiflu) which interfere with the release of intracellular progeny viruses and require early oral administration to stop further virus growth. All these intracellularly active drugs are found to be ineffective on asymptomatic influenza (Jefferson et al., 2010). Furthermore, all these drugs can only be used once the infection is established or as a preventive treatment as these drugs have no effect on the free virus present on the infected throat surface.
Future antiviral research is mostly directed to search new intracellular virus growth inhibitors (Reeves and Piefer, 2005; Briz et al., 2006; Vermeire et al., 2004; Este, 2003). The inhibitors of interactions between the cellular proteins and the viral proteins is also envisaged for topical virus infections (Este, 2003), without taking into consideration the role of proteolytic enzymes.
All the currently used or future drug candidates are chemically synthesized molecules which interfere with the cellular metabolic process with multiple toxic and side effects. Peptides cannot be used for the treatment of topical infections because being protein in nature; peptides are easily neutralized by the proteases.
The use of topical antiviral agents may have the advantage of suppressing viral infectivity, virus growth, new cell infection and cellular cytotoxicity as well as side effects and systemic toxicity (Dasanu and Alexandrescu, 2010). A number of compounds are being developed to specifically target each of these steps leading to virus entry and some compounds have reached early clinical development but their widespread use as topical antiviral agents is suspected due to their toxicity, the absence of efficacy or their peptide nature prone to degradation by proteolysis (Boriskin et al., 2008; Shi et al., 2007; Rossignol et al., 2009). Therefore, there is an urgent need to find a safe drug capable to act topically in multiple ways to neutralize maximum amount of free virus as well as the proteases on the throat surface.
The only natural substances which are known to possess strong affinity for different type of proteins are the plant tannins or curcumin (Ao et al., 2008). Hundreds of tannins are identified in plants with minor structural variations which confer tannin the properties to bind with specific proteins (Deaville et al., 2007; King and Young, 1999).
The results of this study prove that the MMP 1,2,7 and 9 are the major MMPs involved in facilitating topical virus entry and that only certain specific plant tannins can bind with these MMPs. Most of the plant extracts tested in this study were rich in tannins but hardly 6% plant tannins were capable to bind with either proteases or with the virus glycoproteins. These individual plant extracts cannot be used for the treatment of viral throat infection as their total antiviral activity always remained less than 50% and taking into consideration the amount of free virus particles present on an infected throat surface, blocking the activity of 50% virus particles may not significantly reduce the infectious process. This is probably the reason why none of the individual herbal preparations have shown any significant topical antiviral effects (Fink et al., 2009; Namba et al., 1998).
The incapacity of any individual plant extract to block more than 50% total viral infection also indicate that tannin binding with proteases or with the virus glycoproteins is highly specific and requires a dual and synergistic mechanism of neutralizing virus MMPs on one hand and the virus entry on the other hand to stop virus infection. This synergistic antiviral effects of certain plant tannins may be related to the fact that being specific in nature, some tannins bind to the proteases, others to the viral glycoproteins and this combined effect hampers most of the virus particles to infect new cells.
The results of this study prove that in the absence of any antiviral drug, the specificity of plant tannins to bind with the proteins may be a safe and reliable approach to treat topical viral infections. Taking into consideration the number of plant species present on the earth and the variability in their tannin composition, it is suggested to test anti-viral properties of other plant tannins and to verify these findings clinically.
These results constitute a part of study presented in the patent PCT/EP2010/050236 (Shrivastava and Shrivastava, 2009).
CONCLUSION
The results of this study clearly show that some specific proteases are involved in the topical infection of influenza virus and that the influenza virus can be neutralized with specific tannins. Neutralizing free virus particles present on the virus infected surface is one of the best solutions to treat topical viral infections.
REFERENCES
- Ajlia, S.A.S.H., F.A.A. Majid, A. Suvik, M.A.W. Effendy and H.S. Nouri, 2010. Efficacy of papain-based wound cleanser in promoting wound regeneration. Pak. J. Biol. Sci., 13: 596-603.
CrossRefDirect Link - Ali, A., R.T. Avalos, E. Ponimaskin and D.P. Nayak, 2000. Influenza virus assembly: Effect of influenza virus glycoproteins on the membrane association of M1 protein. J. Virol., 74: 8709-8719.
Direct Link - Al-Majhdi, F.N., 2007. Structure of the sialic acid binding site in influenza a virus: Hemagglutinin. J. Boil. Sci., 7: 113-122.
CrossRefDirect Link - Alymova, I.V., G. Taylor and A. Portner, 2005. Neuraminidase inhibitors as antiviral agents. Curr. Drug Targets Infect. Disord., 5: 401-409.
CrossRef - Beppu, Y., Y. Imamura, M. Tashiro, T. Towatari, H. Ariga and H. Kido, 1997. Human mucus protease inhibitor in airway fluids is a potential defensive compound against infection with influenza A and Sendai viruses. J. Biochem., 121: 309-316.
PubMed - Boriskin, Y.S., I.A. Leneva, E.I. Pecheur and S.J. Polyak, 2008. Arbidol: A broad-spectrum antiviral compound that blocks viral fusion. Curr. Med. Chem., 15: 997-1005.
PubMed - Briz, V., E. Poveda and V. Soriano, 2006. HIV entry into the cells-mechanisms and therapeutic possibilities. Med. Clin. (Barc)., 126: 341-348.
PubMed - Dasanu, C.A. and D.T. Alexandrescu, 2010. Prophylactic antivirals may be helpful in prevention of varicella-zoster virus reactivation in myeloma, but are they safe. J. Oncol. Pharm. Pract., 16: 266-268.
CrossRef - Deaville, E.R., R.J. Green, I. Mueller-Harvey, I. Willoughby and R.A. Frazier, 2007. Hydrolyzable tannin structures influence relative globular and random coil protein binding strengths. J. Agric. Food. Chem., 55: 4554-4561.
CrossRef - Delboy, M.G., D.G. Roller and A.V. Nicola, 2008. Cellular Proteasome activity facilitates herpes simplex virus entry at a postpenetration step. J. Virol., 82: 3381-3390.
CrossRef - Diaz-Nido, J., R. Armas-Portela and J. Avila, 1991. Addition of protease inhibitors to culture medium of neuroblastoma cells induces both neurite outgrowth and phosphorylation of microtubule-associated protein MAP-1B. J. Cell Sci., 98: 409-414.
PubMed - Este, J.A., 2003. Virus entry as a target for anti-HIV intervention. Curr. Med. Chem., 10: 1617-1632.
PubMed - Fink, R.C., B. Roschek Jr. and R.S. Alberte, 2009. HIV type-1 entry inhibitors with a new mode of action. Antivir. Chem. Chemother., 19: 243-255.
PubMed - Frazier, R.A., E.R. Deaville, R.J. Green, E. Stringano, I. Willoughby, J. Plant and I. Mueller-Harvey, 2010. Interactions of tea tannins and condensed tannins with proteins. J. Pharm. Biomed. Anal., 51: 490-495.
CrossRef - Gupta, S.P. and S. Kumaran, 2006. Quantitative structure-activity relationship studies on matrix metalloproteinase inhibitors: Piperazine, piperidine and diazepine hydroxamic acid analogs. Asian J. Biochem., 1: 211-223.
CrossRefDirect Link - Hayden, F.G. and A.T. Pavia, 2006. Antiviral management of seasonal and pandemic influenza. J. Infect. Dis., 194: S119-S126.
CrossRef - Hsieh, H.P. and J.T.A. Hsu, 2007. Strategies of development of antiviral agents directed against influenza virus replication. Curr. Pharm. Des., 13: 3531-3542.
CrossRef - Matlin, K.S., H. Reggio, A. Helenius and K. Simons, 1981. Infectious entry pathway of influenza virus in a canine kidney cell line. J. Cell. Biol., 91: 601-613.
PubMed - Kido, H., Y. Niwa, Y. Beppu and T. Towatari, 1996. Cellular proteases involved in the pathogenicity of enveloped animal viruses, human immunodeficiency virus, influenza virus A and Sendai virus. Adv. Enzyme Regul., 36: 325-347.
CrossRef - Kido, H., Y. Okumura, E. Takahashi, H.Y. Pan and S. Wang et al., 2008. Host envelope glycoprotein processing proteases are indispensable for entry into human cells by seasonal and highky pathogenic avian influenza viruses. J. Mol. Genet. Med., 3: 167-175.
PubMed - Kido, H., Y. Okumura, H. Yamada, T.Q. Le and M. Yano, 2007. Proteases essential for human influenza virus entry into cells and their inhibitors as potential therapeutic agents. Curr. Pharm. Des., 13: 405-414.
CrossRef - King, A. and G. Young, 1999. Characteristics and occurrence of phenolic phytochemicals. J. Am. Dietetic Assoc., 99: 213-218.
CrossRefPubMedDirect Link - Konig, R., S. Stertz, Y. Zhou, A. Inoue and H.H. Hoffmann et al., 2010. Human host factors required for influenza virus replication. Nature, 463: 813-817.
CrossRef - Leneva, I.A., R.J. Russell, Y.S. Boriskin and A.J. Hay, 2009. Characteristics of arbidol-resistant mutants of influenza virus: Implications for the mechanism of anti-influenza action of arbidol. Antiviral Res., 81: 132-140.
CrossRef - Mehle, A. and J.A. Doudna, 2010. A host of factors regulating influenza virus replication. Viruses, 2: 566-573.
CrossRef - Murineddu, G., C. Murruzzu and G.A. Pinna, 2010. An overview on different classes of viral entry and Respiratory Syncitial Virus (RSV) fusion inhibitors. Curr. Med. Chem., 17: 1067-1091.
CrossRef - Namba, T., M. Kurokawa, S. Kadota and K. Shiraki, 1998. Development of antiviral therapeutic agents from traditional medicines. Yakugaku Zasshi., 118: 383-400.
PubMed - Reed, L.J. and H. Muench, 1938. A simple method of estimating fifty percent endpoints. Am. J. Epidemiol., 27: 493-497.
CrossRefDirect Link - Reeves, J.D. and A.J. Piefer, 2005. Emerging drug targets for antiretroviral therapy. Drugs, 65: 1747-1766.
PubMed - Rossignol, J.F., S. La Frazia, L. Chiappa, A. Ciucci and M.G. Santoro, 2009. Thiazolides, a new class of anti-influenza molecules targeting viral hemagglutinin at the post-translational level. J. Biol. Chem., 284: 29798-29808.
CrossRef - Sar, T.T., P.T. Aernan and R.S. Houmsou, 2010. H1N1 influenza epidemic: Public health implications for Nigeria. Int. J. Virol., 6: 1-6.
CrossRefDirect Link - Shi, L., H. Xiong, J. He, H. Deng and Q. Li et al., 2007. Antiviral activity of arbidol against influenza A virus, respiratory syncytial virus, rhinovirus, coxsackie virus and adenovirus in vitro and in vivo. Arch. Virol., 152: 1447-1455.
CrossRef - Shrivastava, R., A. Chevalier, M. Slaoui and G. John, 1993. An in vitro method using vascular smooth muscle cells to study the effect of compounds on cell proliferation and intracellular lipid accumulation. Methods Find Exp. Clin. Pharmacol., 15: 345-350.
PubMed - Tambunan, U.S.F., O. Hikmawan and T.A. Tockary, 2008. In silico mutation study of haemagglutinin and neuraminidase on banten province strain influenza a H5N1 virus. Trends Bioinform., 1: 18-24.
CrossRefDirect Link - Vermeire, K., D. Schols and T.W. Bell, 2004. CD4 down-modulating compounds with potent anti-HIV activity. Curr. Pharm. Des., 10: 1795-1803.
PubMed - Vanaman, T.C. and R.A. Bradshaw, 1999. Proteases in cellular regulation minireview series. J. Biol. Chem., 274: 20047-20063.
CrossRef - Xiao, X., Z.Q. Yang, L.Q. Shi, J. Liu and W. Chen, 2008. Antiviral effect of epigallocatechin gallate (EGCG) on influenza A virus. Zhongguo Zhong Yao Za Zhi, 33: 2678-2682.
PubMed - Yamada, H., Q.T. Le, A. Kousaka, Y. Higashi, M. Tsukane and H. Kido, 2006. Sendai virus infection up-regulates trypsin I and matrix metalloproteinase, triggering viral multiplication and matrix degradation in rat lungs and lung L2 cells. Arch. Virol., 151: 2529-2537.
CrossRef - Soltanialvar, M., H. Shoushtari, M. Bozorgmehrifard, S. Charkhkar and F. Eshratabadi, 2010. Molecular characterization of hemagglutinin and neuraminidase genes of H9N2 avian influenza viruses isolated from commercial broiler chicken in Iran. J. Biol. Sci., 10: 145-150.
CrossRef - Shankar, B.P., R.N.S. Gowda, B. Pattnaik, B.H.M. Prabhu and R.P. Kamal et al., 2009. Assessment of pathogenic potential of avian influenza viruses by MDCK cell culture. Int. J. Poult. Sci., 8: 462-464.
CrossRefDirect Link - Ao, C., A. Li, A.A. Elzaawely and S. Tawata, 2008. MMP-13 Inhibitory activity of thirteen selected plant species from okinawa. Int. J. Pharmacol., 4: 202-207.
CrossRefDirect Link








