Research Article
Studies on the Use of Tea Leaves as Pharmaceutical Adsorbent
Department of Chemistry, Panjab University, Cbandigarh-160014, India
Karamvir Sidhu
Department of Chemistry, Panjab University, Cbandigarh-160014, India
Tea (Camellia sinensis) is a tropical evergreen shrub. Black, green and oolong are considered to be the basic types of tea. Black tea, prepared from green tea leaves that have been allowed to oxidize, is the most common form of tea worldwide. All tea leaves contain four basic types of constituents: vitamins, essential oils, polyphenols and caffeine (Hara, 2001). Aside from water, tea is the most widely consumed beverage in the world. Current research has shown that tea contains specific antioxidants and health promoting ingredients (Higdon and Frei, 2003), lowering the risk of heart disease (Davies et al., 2003), stroke (Ikeda et al., 2007) and certain types of cancer like oral, pancreatic and prostate (Mukhtar and Ahmad, 2000). Green tea has also been reported to have antidiabetic properties; it enhances insulin activity and the predominant active ingredient is epigallocatechin gallate (Anderson and Polansky, 2002). Most of the work reported in the literature refers to the medicinal uses of tea and tea extract. However, in the present study it was thought of interest to explore the possibility of using tea leaves as pharmaceutical adsorbent. Tea leaves, being natural, biodegradable, non-toxic, easily available and cost effective, offer a number of advantages over other drug carriers. Drug delivery through medicated tea and use of tea leaves as antidote for drugs has not been reported before. Adsorption of a wide range of drugs on tea leaves has been studied under different experimental conditions using ultraviolet absorption spectroscopic technique for drug analysis.
Materials
Drugs
Cefotaxime sodium (CS), paracetamol (Para), caffeine (Caf), chlorpromazine hydrochloride (Cpz) and metronidazole (Metro) were obtained as gift samples from various manufacturers. Black dried tea leaves were procured locally. All other reagents were of analytical grade and were used without further purification. Water used was double distilled in an all glass apparatus. 0.1M phosphate buffer of pH 7.4 was prepared by mixing 19 mL of 0.2M NaH2PO4 with 81 mL of 0.2 M Na2HPO4 and diluting the mixture to 200 mL with water. In the pH range 2-10, 0.1M Na2HPO4-HCl/NaOH buffers were used.
Drug Analysis
Ultraviolet absorption spectroscopic technique was used for drug analysis. Phosphate buffer was used as solvent. Since buffer could extract active constituents of tea leaves which adsorbed in the UV range (270 nm), extinction coefficients of all the drugs were determined in phosphate buffer containing tea extract, prepared by mixing 0.001 g tea leaves with 10 mL buffer for 24 h. All absorbance data was corrected for the absorbance of tea extract.
Adsorption of Drugs on Tea Leaves
For studying the adsorption of drugs on tea leaves, 1 mg of tea leaves were mixed with 10 mL of drug solutions of increasing concentrations prepared in 0.1M phosphate buffer (pH 7.4). The contents were mixed and allowed to stand at 25°C for 24 h for attainment of adsorption equilibrium. The tea leaves were removed by filtration and the filtrate was analysed for drug concentration by ultraviolet absorption spectroscopic technique after appropriate dilution. The initial drug concentration (Ci) was varied from 50-2000 μM. The drug concentration at equilibrium was calculated as Ce = {(A-ATE) x dilution factor}/ε, where A and ATE are the absorbance of drug solution and tea extract, respectively and ε is the molar extinction coefficient of the drug. The amount of drug adsorbed per gram tea leaves (x/m) was calculated as x/m (mg g-1) = [(Ci - Ce) x 10-3 x MW x 10]/1000 w, where 10 is the volume of drug solution taken for adsorption and w, the weight of tea leaves was 0.001g in each case. To study the effect of pH on drug adsorption, concentration and temperature were kept fixed at 1000 μM and 25°C, respectively and pH was varied from 2-10 using 0.1M Na2HPO4-HCl/NaOH buffer. Kinetics of adsorption was studied at pH 7.4 and 25°C, keeping drug concentration at 1000 μM. The rate of adsorption, d(x/m)/dt, was calculated at 3 h time period. Experiments were also carried out at 37°C for 1000 μM drug at pH 7.4. Enthalpy of adsorption (ΔH ) was calculated using modified form of Van’t Hoff equation, ln {(x/m)2/(x/m)1} = ΔH/R {(T2-T1)/ T2}. The study was conducted in the department of chemistry, Panjab University, Chandigarh, India during the year 2006.
Adsorption of an antibiotic (cefotaxime sodium), analgesic and antipyretic drug (paracetamol), central nervous system stimulant (caffeine), antipsychotic drug (chlorpromazine HCl) and anti-infective drug (metronidazole) on tea leaves has been studied. Extinction coefficients of various drugs in phosphate buffer (pH 7.4) containing tea extract (0.001 g/10 mL) at the respective λmax values are recorded in Table 1. The amount adsorbed (x/m) versus Ce plots for the adsorption of various drugs on tea leaves at 25°C are shown in Fig. 1. All the drugs could be adsorbed on tea leaves and the adsorption was found to be monomolecular (Type I). The maximum adsorption capacity was observed for chlorpromazine HCl and cefotaxime sodium followed by caffeine and paracetamol. Least adsorption capacity was observed for metronidazole. It is seen that at the highest drug concentration used, 179-457 mg of various drugs could be adsorbed per gram tea leaves at 25°C. Thus tea leaves are good adsorbents for these drugs. Cefotaxime sodium and paracetamol are acidic drugs with pKa values 3.4 and 9.7, respectively. Chlorpromazine hydrochloride, metronidazole and caffeine are predominantly basic drugs with pKa values 9.3, 2.6 and 3.6, respectively. Accordingly at pH 7.4, cefotaxime sodium and chlorpromazine hydrochloride are fully ionized whereas the other three drugs exist in predominantly unionized form. Therefore, at this pH, acidic drug, cefotaxime sodium is negatively charged, basic drug chlorpromazine hydrochloride is positively charged while other three drugs are neutral. Hence at near neutral pH, cationic, anionic as well as neutral species have high affinity for the tea surface.
Adsorption data was found to obey both Langmuir (x/m = abCe/(1+bCe), where a and b are constants) and Freundlich (x/m = K Ce1/n, where K and n are constants) adsorption isotherms. Constants of the Langmuir and Freundlich isotherms, obtained from their linearized forms are given in Table 2 for various drugs. Constant ‘a’ of Langmuir adsorption isotherm is a measure of the adsorptive capacity of the adsorbent for the adsorbate under examination and ‘b’ is a constant related to the enthalpy of adsorption (Florence and Attwood, 1998). Constant 1/n of the Freundlich adsorption isotherm is a dimensionless parameter related to the intensity of adsorption (Florence and Attwood, 1998).
Effect of pH
Adsorption data was also obtained at different pH values varying from 2-10 at 1000 μM drug concentration and 25°C. x/m versus pH plots for different drugs are shown in Fig. 2. The extent of adsorption was found to increase with increase in pH in the case of all the drugs.
| Table 1: | Extinction coefficients of various drugs |
 | |
| *Extinction coefficients have been determined in phosphate buffer (pH 7.4) containing tea extract (0.001 g/10 mL) | |
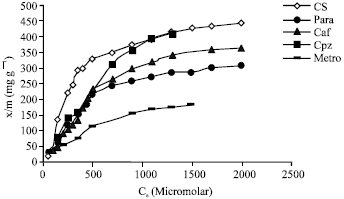 | |
| Fig. 1: | Adsorption isotherms for various drugs |
| Table 2: | Adsorption parameters for Langmuir and Friendlich adsorption isotherms |
 | |
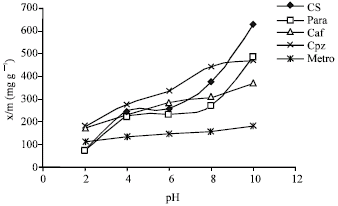 | |
| Fig. 2: | Effect of pH on the adsorption of different drugs on tea leaves |
However, the increase was much larger in the case of acidic drugs as compared to the basic drugs. With increase in pH, the acidic drugs (cefotaxime sodium and paracetamol) should become more and more negatively charged and therefore, it may be concluded that negatively charged species are preferentially adsorbed by tea leaves as the pH is increased. Basic drugs (chlorpromazine hydrochloride, caffeine and metronidazole), on the other hand, should become neutral as the pH is increased. Since adsorption capacity of basic drugs also increases with increase in pH, although the increase is smaller, it appears that the tea surface has some preference for neutral species as well, as the pH is increased. At the highest pH used, 181-631 mg of drugs could be adsorbed per gram tea leaves. Thus tea leaves are better adsorbents at higher pH values.
Effect of Time
To study the kinetics of adsorption process, the amount of drug adsorbed at different time intervals was determined. The concentration of drug was kept fixed at 1000 μM and the temperature was maintained at 25°C. The tea leaves were found to have high affinity for the drugs since the adsorption was fairly rapid. 80-90% of adsorption occurred in the first 3-4 h in most cases. In order to compare the affinity of different drugs for the tea surface, rate of adsorption at 3 h was calculated (Table 3). The rate of adsorption of chlorpromazine hydrochloride and cefotaxime sodium was found to be nearly three times higher than the other three drugs. Thus these two drugs have high affinity for tea surface since the adsorption capacity as well as adsorption rate are large.
Effect of Temperature
The adsorption data for various drugs at 1000 μM drug concentration and pH 7.4 was also determined at two different temperatures. The amount of drug adsorbed (x/m) at room temperature (25°C) and physiological temperature (37°C) is given in Table 3. The amount adsorbed decreased with increase in temperature in each case. This is expected since adsorption is an exothermic process. The enthalpy of adsorption was found to lie in the range -8.723 to -11.765 kJ mol-1 (Table 3).
| Table 3: | Effect of time and temperature on adsorption of drugs by tea leaves |
 | |
| * At 1000 μM initial drug concentration | |
| Table 4: | Amount adsorbed (x/m) and tea leaves required per dose for various drugs |
 | |
| *Reported x/m values are at 1000 μM initial drug concentration. ** Commonly used doses of various drugs are mentioned | |
Pharmaceutical Applications
An attempt has also been made to explore the possible pharmaceutical applications of the adsorption results discussed above. The amount adsorbed (x/m) at 1000 μM initial drug concentration and the weight of tea leaves required per dose for various drugs at pH 7.4 and 10 are given in Table 4. It is seen that for the commonly used doses, a very small quantity of tea leaves (0.061-1.887 g at pH 7.4 and 0.059-1.105 g at pH 10) are required per dose. Thus tea leaves are good adsorbents for all the drugs. These adsorption characteristics can be explored for the design of drug delivery systems as well as use of tea leaves as safe, natural and effective antidote for these drugs.
The results are specially significant in the case of antipsychotic drug, chlorpromazine hydrochloride, where the dose required is much smaller (25 mg) and the adsorptive capacity is high. Thus only 0.06 g is required per dose and therefore, tea leaves are an excellent adsorbent for this drug. The toxicity of antipsychotic and antidepressant drugs due to overdose or accidental intake is commonly encountered (James and Kearns, 1995). Thus tea leaves can be used as safe antidote for chlorpromazine hydrochloride with no side effects.
The adsorption was found to be monomolecular and at the highest drug concentration used, the adsorptive capacity at 25°C was in the range 179-457 mg g-1 tea leaves at pH 7.4. The adsorptive capacity increased with increase in pH. At the highest pH studied, 181-631 mg of drugs could be adsorbed per gram tea leaves. Adsorption was fairly rapid, 80-90% adsorption occurred in 3-4 h. The enthalpy of adsorption was in the range -8.723 to -11.765 kJ mol-1. The weight of tea leaves required per dose for the commonly used doses of these drugs was found to be 0.061-1.887 g at pH 7.4 and 0.059-1.105 g at pH 10. A small quantity of tea leaves required per dose showed that tea leaves are good adsorbents for these drugs and the data can be explored for the design of drug delivery systems as well as use of tea leaves as safe, natural and effective antidote for these drugs. The results are specially significant in the case of antipsychotic drug, chlorpromazine hydrochloride, where the dose required is much smaller (25 mg) and the adsorptive capacity is high. Thus only 0.06 g is required per dose and therefore, tea leaves are an excellent adsorbent for this drug. The present study leads to an important conclusion that tea leaves have the potential to be used as a safe and effective pharmaceutical adsorbent for the studied drugs.