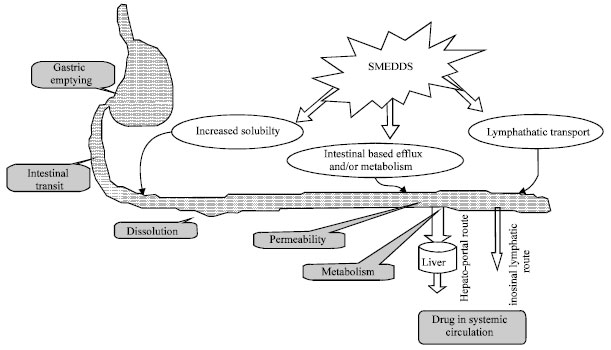
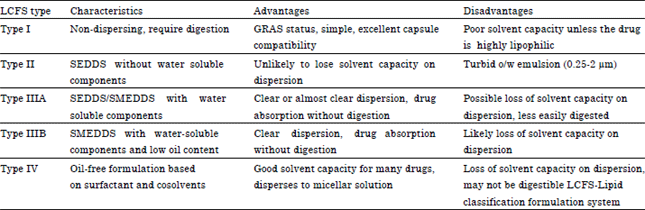
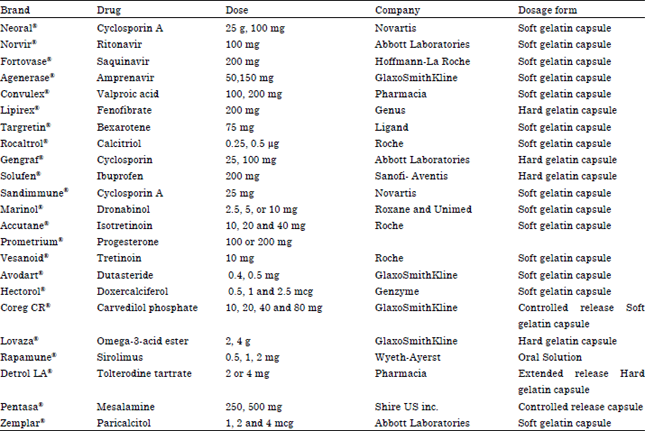
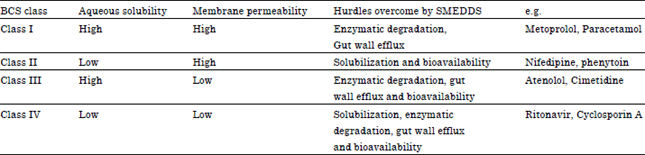
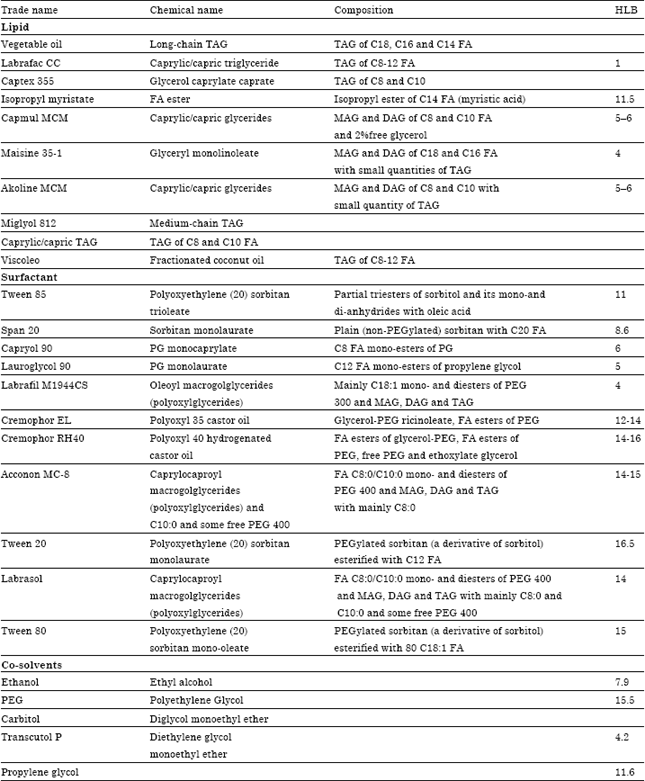
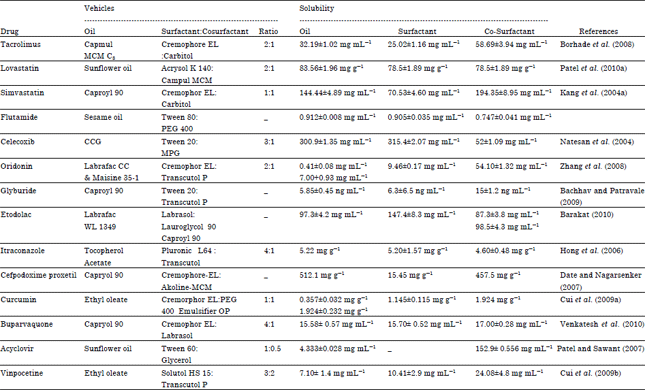
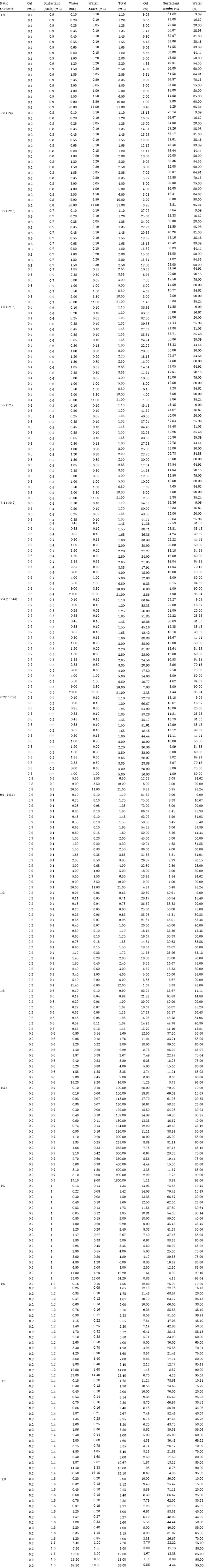
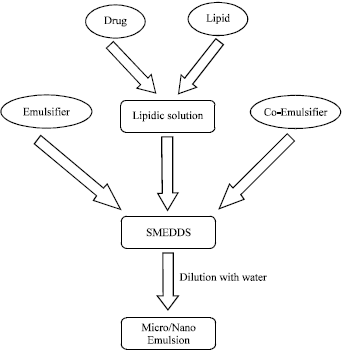
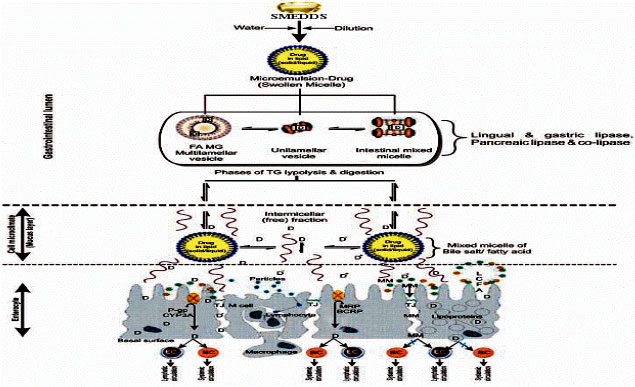
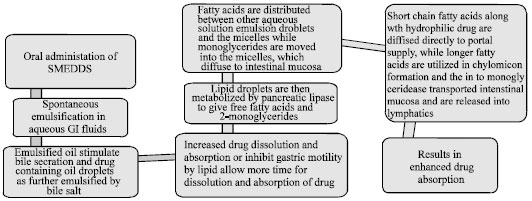
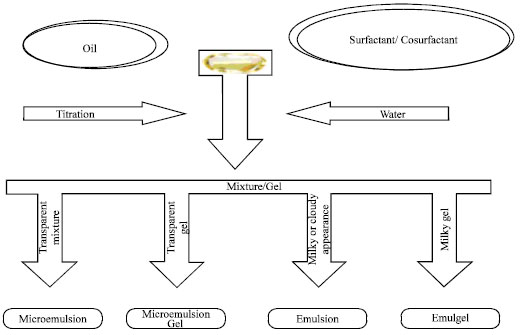
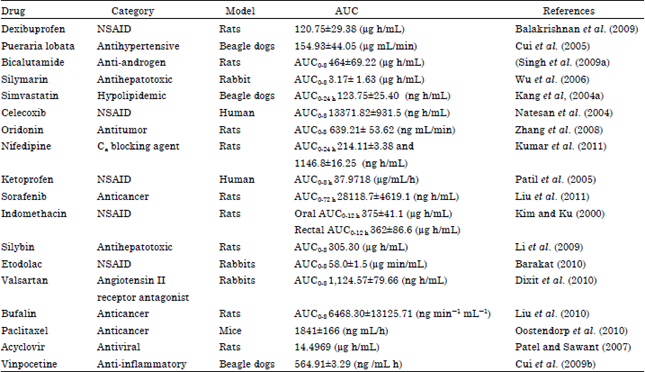
Review Article
A Review on Novel Therapeutic Strategies for the Enhancement of Solubility for Hydrophobic Drugs through Lipid and Surfactant Based Self Micro Emulsifying Drug Delivery System: A Novel Approach
Rugnta College of Pharmaceutical Sciences and Research, Kohka-Kurud Road, Bhilai, Chhattisgarh, India
Tapan Kumar Giri
Rugnta College of Pharmaceutical Sciences and Research, Kohka-Kurud Road, Bhilai, Chhattisgarh, India
Dulal Krishna Tripathi
Rugnta College of Pharmaceutical Sciences and Research, Kohka-Kurud Road, Bhilai, Chhattisgarh, India
Ajazuddin
Rugnta College of Pharmaceutical Sciences and Research, Kohka-Kurud Road, Bhilai, Chhattisgarh, India
Amit Alexander
Rugnta College of Pharmaceutical Sciences and Research, Kohka-Kurud Road, Bhilai, Chhattisgarh, India
LiveDNA: 91.5283
ORCID: 0000-0003-0391-7650