Research Article
Adsorption of Acid Dyes from Wastewater on Saudi Bentonite Clay
Department of Chemistry, Al-Imam Muhammad Bin Saud Islamic University (IMSIU), P.O. Box 90950, Riyadh, 11623, Kingdom of Saudi Arabia
Among different industrial wastewaters, dye color wastes occupy a prominent position. Since many dye pigments contain toxic organic pollutants, the presence of dye colors in wastewater may indicate the existence of toxicants. If an effort is not made to remove pigments, the public assumes that little effort has been expended in removing the other pollutants. A color in the wastewater has to be removed before it is discharged into a water body or intended for land disposal.
Currently, the adsorption process is considered as a useful technology to minimize the extent of water pollution by different types of dyes. The major advantages of an adsorption system for controlling water pollution are a low capital investment in terms of initial cost and land required, simple design, easy operation and high removal efficiency of dye related waste constituents as compared to other methods. The adsorption process also achieves a higher level of dye removal over a wider range of solution conditions and also reduces sludge or solid residuals requiring disposal. Activated carbon is one of the most commonly used adsorbents for wastewater purification in an adsorption process. But, the use of activated carbon is relatively expensive for the removal of dye colors from wastewaters. There are also losses of approximately 15-20% during the regeneration process (McKay et al., 1980). Therefore, the researchers are looking for low-cost materials suitable for the adsorption of dyestuff from wastewater.
Previously, the idea of using low-cost adsorbents for the removal of dyes from wastewater has been investigated extensively. Various low-cost adsorbents were evaluated for their adsorption properties, including wood, sawdust, maize cob, rice bran, rice hull, peat, chitin, lignite, nut husks and hardwood. These materials have been reported to adsorb different pollutants such as dyestuffs and other toxic pollutants (El-Geundi, 1990a; Okieimen et al., 1991; Marshall et al., 1993; Zarraa, 1995; Marshall and Champagne, 1995; Ho and McKay, 1998; McConvey and McKay, 1985; Asfour et al., 1985; McKay, 1987; McKay et al., 1987; El-Geundi, 1990b; Allen et al., 1989, 1994). However, Ho and McKay (1998) specifically studied the suitability of using wood as a natural adsorbent material for the removal of dyestuffs from simulated wastewater.
Besides, adsorption isotherms and adsorption kinetics of dyestuffs on to wood was determined (Ho and McKay, 1998) and the data obtained was correlated using equation based on the Langmuir model and intraparticle diffusion model. The adsorption kinetics of dyestuffs onto wood meal was also investigated in an agitated batch adsorber based on pore diffusion process and external mass transfer (McKay et al., 1985). Asfour et al. (1985) studied the suitability of using hardwood (sawdust) as a natural adsorbent to remove basic dyestuffs from simulated wastewater. They found that the maximum adsorption capacity of hardwood is higher than the softwood. The kinetics of adsorption and adsorption isotherms for the process were also studied.
The adsorption kinetics of the dyestuffs onto chitin was determined in an agitated batch adsorber and fixed-bed systems (McKay, 1987; McKay et al., 1987). In another study, feasibility of using maize cob waste and lignite as natural adsorbent materials for the removal of dyestuffs from simulated wastewater were studied (El-Geundi, 1990a, b; Allen et al., 1989). The adsorption isotherms for the adsorption of basic dyestuffs onto maize cob were studied and the experimental results fitted well to Langmuir, Freundlich and Redlich-Peterson models (El-Geundi, 1990a; Allen et al., 1989). While, El-Geundi (1990b) studied the adsorption kinetics for the adsorption of basic dyestuffs onto maize cobs.
Allen et al. (1994) worked on peat as an adsorbent material for the removal of dyestuffs and metals from simulated wastewater. They also determined the adsorption isotherms as well as the adsorption kinetics of the dyestuffs. They found peat as a successful adsorbent for wastewater treatment.
The natural clay has been investigated as an adsorbent for the removal of lead from drinking water (Orumwense, 1996) in order to determine the kinetics of adsorption and adsorption isotherms at different temperatures. The results showed that lead removal is favored by a low concentration, a high temperature and an acidic pH. It was concluded that natural clay is a good adsorbent for lead ions. In the area of metallic effluents, research was carried for the adsorption of cadmium, zinc and Cr (VI) ions on natural clay from aqueous solution (Pradas et al., 1994a; Singh et al., 1992). The experimental data were fitted to Langmuir equation in order to calculate the adsorption capacities of the adsorbents. The removal of dyestuffs from simulated wastewater has been studied using natural clay as an adsorbent (El-Geundi, 1996). He determined that the rate of adsorption is controlled initially by the boundary layer film. The adsorption kinetics of dyestuffs onto natural clay were also determined in batch adsorbers (El-Geundi, 1991).
Removal of chlorophyll-a from acetone solution using natural and activated bentonite as adsorbents was investigated by Pradas et al. (1994b). Another study used the Freundlich model with the experimental results in order to calculate the adsorption capacities of pristine and HCl-activated montmorillonite as adsorbents for adsorption of acid dye from wastewater (Lin et al., 2004). They studied different parameters such as liquid flow rate, initial dye concentration and bed height. It was found that, the adsorption capacity of acid dye onto pristine clay was enhanced by activating the clay by HCl. Also, hectorite clay was modified using cetyldimethylbenzylammonium chloride and cetylpyridinium chloride to adsorb Acid Red 151 from wastewater (Mumin et al., 2007). Recently, the effect of pH on the adsorption process was investigated. It was found that the adsorption capacity was 208.33 and 169.49 mg g-1 for the modified cetyldimethylbenzylammonium chloride-hectorite and cetylpyridinium chloride, respectively. Spahn and Schlunder (1975) investigated the adsorption of Congo red dye from wastewater on clay for the effect of different parameters such as, pH, temperature and the mass of adsorbent. They found an increase in the amount of adsorption with increasing the initial concentration and decreasing the pH values with a removal efficiency of more than 98%.
Adsorption of methylene blue from wastewater on activated clay was investigated by El- Mouzdahir et al. (2010) by treating the clay by thermal method by heating at 300°C for 2 h and by acid treatment with nitric acid. They found the maximum adsorption capacity of clay as 500 mg g-1. Recently, natural clay was also used as an adsorbent for adsorption of acid red 88 from waste water (Akar and Uysal, 2010). They studied different parameters such as adsorbent amount, contact time, initial pH, initial dye concentration, flow rate and column initial depth. It was found that, pseudo-second-order kinetic and Langmuir isotherm models can be used to describe the adsorption process. From the study of literature, it was found that though the research has been accomplished on the natural clay as an adsorbent but little has been done on Saudi bentonite clay for removing acid dyes from wastewaters under local conditions.
In recent years, the increasing demand for water in Saudi Arabia has focused attention on the problem of dyes removal from wastewater. Many researchers have studied natural local adsorbents (clay, date pits powder, solid waste from water treatment plants) suitable for use in this process. Investigations have been struggling to determine whether cheap, commercially available materials hold promise in the treatment of wastewater. The initial research on a low-cost material has included the natural clay. The aim of this study is to investigate the application of a Saudi natural clay as an adsorbent material for the removal of acid dyes from wastewater. Also, determine the feasibility of using Saudi clay as an adsorbent for removing acid dyes from waste water. Finally, determine equilibrium and kinetic parameters necessary to design batch adsorbers.
Materials
Adsorbent: Bentonite clay was used as an adsorbent this study. The clay was obtained from Jeddah city, West of Saudi Arabia. The Saudi bentonite clay was analyzed chemically and the results are given in Table 1 and 2.
The mineralogical analysis of the bentonite clay was done by RXD. The clay contains Kaolinite (9%), montmorillonite (82%) and quartz and illite (9%).
| Table 1: | Chemical analysis of bentonite clay by XRF |
 | |
 | |
| Fig. 1: | Acid blue 25 |
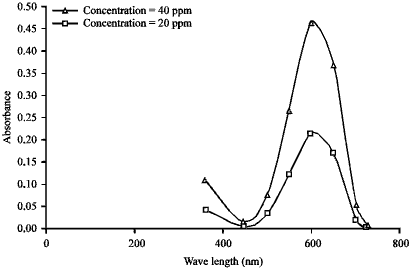 | |
| Fig. 2: | Determination of the maximum wavelength value for AB25 |
| Table 2: | Characteristic properties of bentonite clay |
 | |
Adsorbate: The adsorbate and its associated structures are shown in Fig. 1.
Analytic work
Maximum wavelength: The maximum wavelength value for acid blue 25 (AB25) was 600 nm determined by spectrophotometer (Fig. 2).
Calibration curve: A known concentrations ranging from 5-60 mg L-1 of acid blue 25 were prepared to measure the absorbance of the samples and to obtain the calibration curve for the acid blue 25 as shown in Fig. 3.
Batch experimental studies
Equilibrium experiments: Equilibrium data were obtained by placing a 0.5 g of clay in 50 mL dye solution in bottles in an agitation shaker bath. In each experiment, the dye concentrations ranged from 15-467 mg L-1.
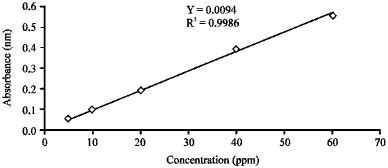 | |
| Fig. 3: | Calibration curve for AB25 |
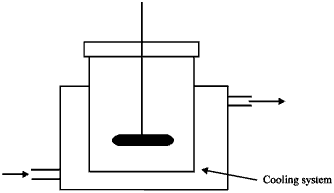 | |
| Fig. 4: | Vessel reactor used in the experiment |
The temperatures of the process were 20, 40, 60 and 75°C. It was observed that 4 h in the constant agitation bath was sufficient for the Acid Blue 25/clay solutions to reach equilibrium. Dye adsorbed on clay was calculated as follows:
| (1) |
The equilibrium curves were obtained by plotting qe versus ce.
Adsorption rate experiments: The contact time experiments provided the kinetic data in the form of concentration decay versus time curves. In the contact-time experimental run, a weighed amount of clay was brought into contact with 1.7 L of a dye solution with a predetermined initial concentration as shown in Fig. 4. When the adsorbate and adsorbent came into contact, the mixing begins at a prefixed impeller agitation speed. At regular time intervals, 3 mL samples of solution were taken by a syringe and stored in sample bottles, sealed to prevent evaporation and dye concentration of each sample is analyzed.
Kinetic experiments: The kinetic experiments were done by using 2 L vessel reactor with cooling system as shown in Fig. 4.
Kinetic experiments on acid blue 25: The vessel reactor was used to study the adsorption rate as a function in the following parameters.
Influence of agitation speed: The adsorption rate was determined using a mass of clay (3.4 g), temperature (20°C), a particle size (75-150 μm) and mixed with 1.7 L of a dye concentration solution (200 mg L-1). In these experiments, the agitation speed ranged from 200-800 RPM. Samples (3 mL) were taken by a syringe at different time intervals. Then the concentration of samples was determined as described previously. The experimental data were plotted as a dimensionless concentrations versus time.
Influence of particle size: The adsorption rate was determined using the same conditions as stated in the agitation speed experiment except that the agitation speed was fixed at 400 RPM. Different particle size ranges (75-150, 250-295, 295-600 and 600-850 μm) were used. The treatment of the collected samples were the same as described in the first section (agitation speed).
Influence of temperature: The adsorption rate was determined using the same conditions as stated above at an agitation speed of 400 RPM. In these experiments, the temperature was 20, 40 and 60°C. The treatment of the collected samples was the same as described in the agitation speed experiment.
Mathematical models
Adsorption isotherms: Two types of isotherms were in this study as summarized below.
Langmuir isotherm: The Langmuir equation assumed that the adsorption of dye on clay is a monolayer and is applied to evaluate the maximum capacity of the clay (McConvey and McKay, 1985). The Langmuir isotherm equation is described as follows:
| (2) |
However, the Eq. 2 can be written in a linear form to determine the constants aL and KL as shown in Eq. 3:
| (3) |
A plot of Ce/qe versus Ce provided the constants, aL and KL for the system.
Freundlich isotherm: The Freundlich isotherm model describes the experimental data for heterogeneous surface. The Freundlich form is written as follows:
| (4) |
The constants KF and n were found by linearizing the equation:
| (5) |
The favorable adsorption is indicated by values of n greater than 1 (n > 1) (El-Geundi, 1990a).
Rate limiting steps: Three steps were assumed to obtain the rate limiting steps as follows:
| Step 1: | Mass transfer of dye from solution to the adsorbent surface |
| Step 2: | Internal diffusion of dye by pore diffusion mechanism, or surface diffusion mechanism |
| Step 3: | Adsorption at the internal surface |
In this process, it was assumed that step 3 occurs very fast and was neglected in the analysis of adsorption rate. Therefore, there will be only two rate limiting steps i.e., external mass transfer and intra-particle diffusion.
Single mass transfer models
External mass transfer: The external mass transfer is the controlling step, therefore, the external mass transfer coefficient, kf was determined by using the initial mass transfer (Spahn and Schlunder, 1975) as described as follows:
| (6) |
where, Ap is the total surface area of the clay and can be written as follows:
| (7) |
Consequently, by sketching a tangent line (round t = 0) to the curve of dimensionless concentration (Ct/Co) against time, the external mass transfer coefficient can be determined.
Intra-particle diffusion: The mass transport model is based only on intra-particle diffusion. So, the fractional dye uptake can be calculated from Eq. 8 as follows according to McKay (1983):
| (8) |
As shown in Eq. 8, there is a linear relationship between fractional dye uptake against (D t/r2)½. Since the diffusivity and the clay particle size are assumed constant during any given experiment, the dye uptake is linearly dependent on the square root of time. Consequently a rate parameter D was defined as the slope of the graph of the amount of dye adsorbed per gram of clay, qt, against t½. But in all the diffusion studies, rate processes are usually expressed in terms of the square root of time (McKay, 1983).
Equilibrium of acid blue 25: Many experimental isotherms were carried on clay and acid blue 25 (AB25) as summarized as follows:
Influence of particle size
| Mass of clay | = | 0.5 g |
| Solution volume | = | 0.05 L |
| Temperature | = | 20°C |
| Initial dye concentration | = | 15-467 mg L-1 |
| Particle size range | = | 75-150 μm |
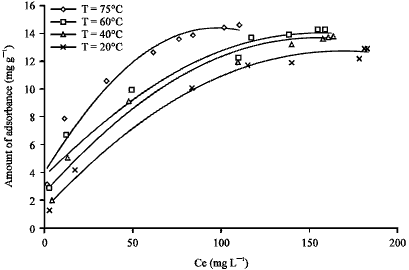 | |
| Fig. 5: | Effect of temperature on adsorption isotherm for AB25 on clay |
Influence of temperature
| Mass of clay | = | 0.5 g |
| Solution volume | = | 0.05 L |
| Particle size range | = | 75-150 μm |
| Initial dye concentration | = | 15-315 mg L-1 |
| Temperature | = | 20, 40, 60, 75°C |
The equilibrium isotherm results are presented in Fig. 5.
Batch kinetic of acid blue 25: Kinetic experiments were carried on clay and acid blue 25 as summarized as follows:
Influence of agitation speed
| Solution volume | = | 1.7 L |
| Particle size | = | 75-150 μm |
| Initial dye concentration | = | 200 mg L-1 |
| Temperature | = | 20°C |
| Mass of clay | = | 3.4 g |
| Agitation speeds | = | 200, 400, 600, 800 RPM |
Influence of particle size
| Solution volume | = | 1.7 L |
| Agitation speed | = | 400 RPM |
| Mass of clay | = | 3.4 g |
| Initial dye concentration | = | 200 mg -1 |
| Temperature | = | 20°C |
| Particle size range | = | 75-150, 250-295, 295-600, 600-850 μm |
Influence of temperature
| Solution volume | = | 1.7 L |
| Agitation speed | = | 400 RPM |
| Mass of clay | = | 3.4 g |
| Initial dye concentration | = | 200 mg L-1 |
| Particle size range | = | 75-150 μm |
| Temperature range | = | 20, 40, 60°C |
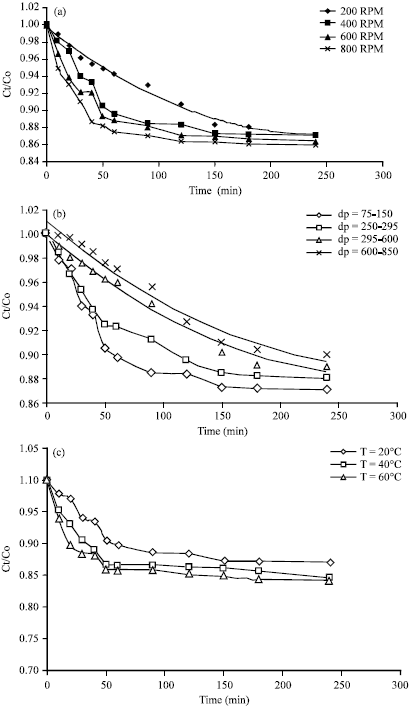 | |
| Fig. 6(a-c): | Effect of (a) Agitation speed, (b) Particle size range and (c) Temperature on the adsorption of AB25 on clay |
The results of kinetic experiments are presented in Fig. 6a-c.
Mechanism of adsorption: The main components of the experimental clay were SiO2 and Al2O3 (Table 1). The adsorption of the acid blue 25 occurs on the positive sites of the clay as a result of the electrostatic attraction between these sites and the anions of the dye.
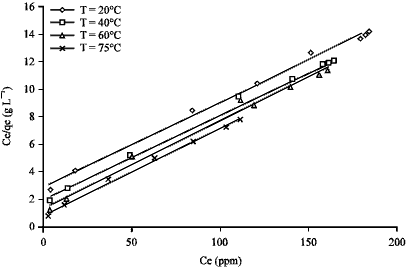 | |
| Fig. 7: | Effect of temperature on Langmuir isotherm plots for AB25 on clay |
The formation of positive sites on the clay results from the presence of alumina but the adsorption of acid dye will be low because of the small number of positive sites.
Equilibrium study
Adsorption equilibrium: The adsorption process reached the equilibrium within 4 h, therefore, the equilibrium experiments were carried at least for 5 h to insure that the equilibrium state was attained. The major components of clay were Si and Al. In contact with water, clay becomes positively charged due to the presence of alumina and at a pH lower than 9. Like most dyes, acid blue 25 ionizes in solution, becoming an anionic colored component. Because of the dye’s negative charge, the acid dye anion has an electrostatic attractive force, but the amount of adsorption remains low due to limited number of positive sites on the clay particles.
Influence of temperature on adsorption equilibrium: Data in Fig. 5 shows that the adsorption capacity of acid blue 25 on clay increased with increasing the temperature. Also, the mobility of the dye ion increases when the temperature is increased. Furthermore, the increasing capacity for acid dye adsorption on clay with increasing temperature indicates that the number of active sites available increases with temperature (Boki et al., 1992).
Isotherm models
Langmuir isotherm: The Langmuir equation in the linear form is given as follows:
| (9) |
Figure 7 shows the linear relationship between Ce/qe and Ce at different temperatures. The equilibrium parameters KL and aL were calculated from the slope and intercept of the straight lines and presented in Table 3.
Freundlich isotherm: The Freundlich isotherm model is written in linear form as follows:
| (10) |
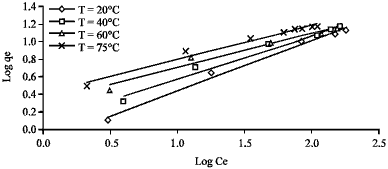 | |
| Fig. 8: | Effect of temperature on Freundlich isotherm plots for AB25 on clay |
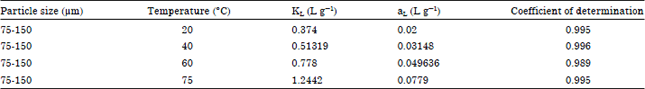
| Table 3: | Langmuir constants for the acid blue 25 adsorption process |
 | |
| Table 4: | Freundlich constants for the acid blue 25 adsorption process |
 | |
Figure 8 demonstrates the linear relationship between log qe and log Ce at different temperatures. The equilibrium constants were calculated and presented in Table 4.
Goodness of the fit: The data from the equilibrium models (Langmuir and Freundlich) and the data from equilibrium experiments were plotted. The coefficient of determination (R2) was calculated to assess the goodness of the fit between the equilibrium models and the experimental data. The R2 values are given in Table 3 and 4. However, considerable variations were observed between these two models as clear from the values of R2. It was found that, Langmuire model fits the experimental data well.
Kinetic study (single mass transfer model)
External mass transfer model
Influence of agitation speed: The experimental results from the kinetic study for the adsorption of acid blue 25 on clay with varied degrees of agitation speed are shown in Fig. 6a. The external mass transfer coefficients (kf) were calculated form Eq. 6. It was observed that increasing the agitation speed caused a reduction in the film resistance to mass transfer and increased the external mass transfer coefficients as shown in Table 5. Therefore, it can safely be concluded that the adsorption rate is controlled by the agitation speed.
Influence of particle size: Experimental results from the kinetic study for the adsorption of acid blue 25 on clay with different particle size are shown in Fig. 6b.
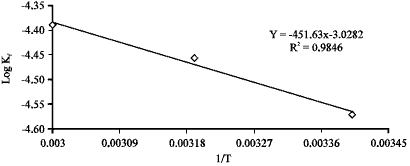 | |
| Fig. 9: | Plot of log Kf against 1/T for the adsorption of acid blue 25 on clay |
| Table 5: | External mass transfer coefficients, Kf, at different agitation speeds |
 | |
| Table 6: | External mass transfer coefficients, Kf, at different clay particle sizes |
 | |
| Table 7: | External mass transfer coefficients, Kf, at different temperatures |
 | |
The external mass transfer coefficients (kf) were calculated from Eq. 6. It was observed that the external mass transfer coefficient (kf) decreased with increasing the particle size. This may due to the the size of the particle as the large particles move slowly in the solution than the small particle, hence, the shear on the particle will be less. Consequently, the film thickness is thicker on the large than that the small particle (Table 6).
Influence of temperature: Experimental results from the kinetic study for the adsorption of acid blue 25 on clay with different temperatures are shown in Fig. 6c. External mass transfer coefficients (kf) were calculated as given in Table 7. It was observed that, with increasing temperature, the external mass transfer coefficients increased thus resulting in increased adsorption rate of dye on clay. This may be due to increase in mobility and dye diffusion with increasing the solution temperature thus producing higher concentration gradients. Similar conclusions were reported by Boki et al. (1992) who concluded that the number of active sites available for dye adsorption increases with temperature.
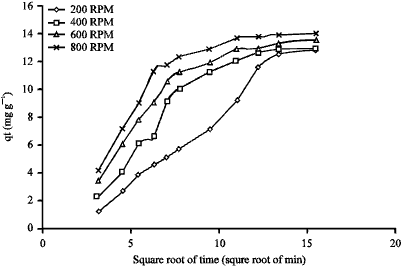 | |
| Fig. 10: | Plot of qt against square root of time for various agitation speeds during the adsorption of AB25 on clay |
| Table 8: | Macropore rate parameters, D, for different agitation speeds |
 | |
Figure 9 shows a linear relationship between log kf versus 1/T. The activation energy can be calculated using the Arrhenius equation for specific rate constants:
| (11) |
Logarithmically:
| (12) |
The activation energy (E) for the acid dye was calculated from the slope of the straight line in Fig. 9 and by using Eq. 4. It was found that the activation energy is 8.65 kJ mol-1.
Intra-particle diffusion model: The diffusion model is used based on intra-particle diffusion as the rate controlling step for adsorption. A plot of qt versus t½ can assess the effect of intraparticle diffusion on adsorption. The study results are in agreement with many researchers who represented good linearization of adsorption data by plotting qt against t½ (Juang and Swei, 1996; Juang et al., 1996; Sekaran et al., 1995). The macro-pore rate parameter (D) was calculated from the slope of the linear portion.
Influence of agitation speed: Figure 10 shows the kinetic results at different agitation speeds. The resulting plot is linear due to the fact that intraparticle diffusion is dominant in the adsorption controlling step. The macropore rate parameters (D) were calculated at different agitation speeds and represented in Table 8.
The D values increased with increasing the agitation speed. This may be due to the fact as the adsorption process is controlled by two steps i.e., first by the external mass transfer mechanism and secondly by the intra-particle diffusion mechanism.
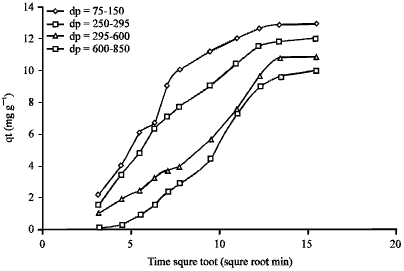 | |
| Fig. 11: | Plot of qt against time square root for various particle size range during the adsorption of AB25 on clay |
 | |
| Fig. 12: | Plor of qt against time square root for various temperature during the adsorption of AB25 on clay |
The external mass resistance decreased with increasing the agitation speed, so the dye became more concentrated at surface of the adsorbent producing a greater driving force at the internal surface resulting in higher values of macropore rate parameters.
Influence of particle size: The influence of the clay particle size on the adsorption of acid blue 25 is shown in Fig. 11. The resulting plot is linear. The macropore rate parameters were calculated as presented in Table 9. It was observed that the adsorption of dye increased with a decrease in the particle size. This may be attributed to the adsorption surface area which is large for the small particles.
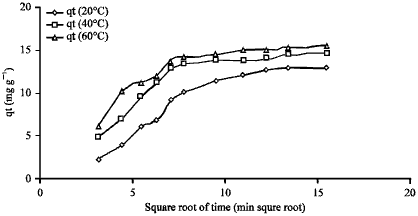
Influence of temperature: The influence of the temperatures on the adsorption of acid blue 25 is shown in Fig. 12. The resulting plot is linear. The macropore rate parameters were calculated and presented in Table 10. It was noticed that the values of macropore rate parameters increased with increasing the temperature of the solution.
| Table 9: | Macropore rate parameters, D, for different particle sizes |
 | |
| Table 10: | Acropore rate parameters, D, for different temperatures |
 | |
The study findings are in line with those of Boki et al. (1992) who indicated that the number of active sites available for adsorption increased with increasing the solution temperature.
The adsorption of acid dye on bentonite clay increased with increasing the temperature. A comparison showed a good agreement between the theoretical isotherms and the experimental data. The increase in the temperature increased the clay active sites resulting an increase in the adsorption rate. Although, the external mass transfer coefficient was determined but with limited success. Overall, the intraparticle diffusion is a major rate-controlling step in the rate of acid dye removal from aqueous solutions.
NOMENCLATURE
| aL | : | Langmuir isotherm constant (L mg-1) |
| Ap | : | Particle surface area (cm2) |
| Ar | : | Arrhenius constant |
| Co | : | Initial solution concentration (mg L-1) |
| Ce | : | Equilibrium solution concentration (mg L-1) |
| Cs | : | Solution concentration at particle surface (mg L-1) |
| Ct | : | Solution concentration at time t (mg L-1) |
| dp | : | Particle size (μm) |
| E | : | Activation energy (kJ/mol) |
| D | : | Macropore rate parameter (mg dye/g clay t-0.5) |
| Kf | : | External mass transfer coefficient (cm sec-1) |
| KF | : | Freundlich isotherm constant (L g-1) |
| KL | : | Langmuir isotherm constant (L g-1) |
| M | : | Mass of adsorbent |
| n | : | Freundlich isotherm constant |
| q | : | Solid-phase concentration (mg g-1) |
| qe | : | Equilibrium solid-phase concentration (mg g-1) |
| r | : | Radial distance from the center of particle, 0≤r≤R (cm) |
| T | : | Absolute temperature (K) |
| t | : | Time |
| V | : | Solution volume (L) |
| ρp | : | Particle density (g cm-3) |