ABSTRACT
The microstructure of sinters produced from Nigerian Iron ores which consist of hematite and magnetite have been investigated using optical microscopy and X-ray diffraction analyses. The effect of magnetite in the ores is to reduce the quantity of SFCA obtained at any basicity when compared with an ore that is wholly hematite. A microstructure that is essentially SFCA can be obtained in Itakpe ore when basicity is 2.5 and the sintering temperature is 1300°C. The Itakpe iron ore is relatively better than Chokochoko iron ore for production of Sinters of good properties on the basis of microstructures developed during sintering.
PDF Abstract XML References
How to cite this article
URL: https://scialert.net/abstract/?doi=tasr.2007.508.514
INTRODUCTION
For the best results in the Blast Furnace, Sinter as a burden material must posses certain properties which have been found (Loo and Wong, 2005; Li-Heng, 2005, Mansurova et al., 2006; Price and Wasse, 1972; Ahsan et al., 1983; Mukhejee and Whiteman, 1985; Ishirara, 1984; Dawson et al., 1984. Dawson et al., 1985; Matsuno and Hirada, 1979; 1981; Yang, 2005; Yang and Davis, 2005) to be dependent on the mineralogy of the Sinter after sintering. However, the mineralogy of the sinter material in turn depends on the mineralogy of the raw materials that are used to produce the sinter.
The main phases that constitute the microstructure of fluxed sinters are SFCA (SFCA is a complex quaternary phase known as silicoferrite of calcium and aluminium. Its chemical composition is given as 5CaO. X SiO2. yFe2O3. zAl2O3 with x + y + z = 12.} hematite (Fe2O3), magnetite (Fe2O3) and dicalcium silicate (2CaO). Of all these phases, SFCA has been found to be the most desirable in terms of conferring on the sinter the best properties such as high responsibility, high cold strength, good low temperature breakdown resistance and high softening temperature. Recent research efforts have therefore been geared towards maximizing the proportion of this phase in iron ore sinters. It has been found that the formation of the phase is favored by hematite ores and recent studies in Sheffield (Egundebi and Whiteman, 1989) have also shown that the atmosphere during sintering has an effect on its proportion. An oxidizing atmosphere is found to favour the formation of SFCA whereas reducing atmosphere does not. The result in reducing atmosphere has also been confirmed by the fact that the phase does not form when a purely magnetite ore is used.
Proposals have been put forward as to the mechanism of formation of this phase (Egundebi and Whiteman, 1989). The morphology of this phase is also an important parameter in its role as the best phase in sinter. It can be obtained in needle and lath shapes. It is the needle-like shape that gives the sinter a calcium ferrite acicular structure that is preferred. Mechanisms for obtaining one in preference to the other have also been suggested (Egundebi and Whiteman, 1989).
In this study, the effect of the mixture of hematite and magnetite coexisting in an ore, as is the case in Nigerian ores, on the evolution of microstructure (notably SFCA) is being investigated. This aims also at confirming that magnetite inhibits the formation of SFCA.
| Table 1: | Chemical and mineralogical analysis of the ores |
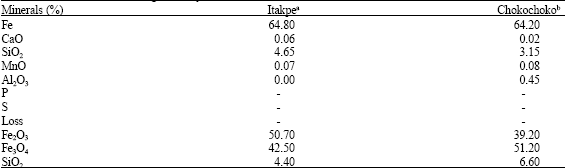 | |
| A Concentrates of the Ore, B The Mineralogical analysis are for the run-of-mine | |
| Table 2: | Experimental mixes |
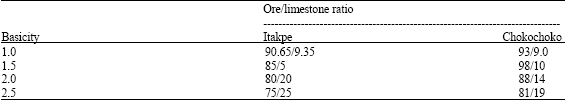 | |
MATERIALS AND METHODS
For this study, concentrates of iron ores from two Nigerian deposits of Itakpe and Chokochoko were used. The ores consists of hematite, magnetite and silica as shown by their x-ray diffraction patterns. The mineralogical and chemical analyses of the ores are shown in Table 1.
The ores were ground and sieved; -75 µm size fractions were used in the experiments. A commercial grade limestone containing about 7% SiO2 was used and this was ground to the same size fractions as the ores. Mixtures of limestone and ores were prepared to give samples of basicities *1, 1.5, 2 and 2.5. These basicities however correspond to different Ore/limestone ratios (Table 2) for the two ores because of differences in their chemical analyses. Thorough mixing was carried out on the samples using pestle and mortar.
The experiments were carried out in a horizontal tube furnace which can be heated up to 1400°C by resistance heating. Sintering was carried out in air (i.e., with the furnace open) at temperatures of 1250°C and 1300°C using 4 min sintering times at each temperature. These two temperatures were chosen since it has been found out form previous works that sintering below 1200°C and above 1300°C does not give good sinters.
Quantitative determination of SFCA phase in the samples was carried out using X-ray diffraction and an internal standard method. A standard sample of SFCA, used for obtaining a calibration graph, was first prepared using pure samples of Fe2O3, CaO, SiO2 and Al2O3 in a Au/Pt/Rh crucible. Pure lime was used as the internal standard and its proportion in each of the experimental samples was 20%.
RESULTS
The results obtained using both Itakpe and Chokochoko ores for sintering are as shown in Fig. 1 for Itakpe ore and Fig. 2 for Chokochoko Ore.
In the Itakpe ore sinters produced at 1250°C, the presence of SFCA is revealed in the x-ray diffraction patterns and the micrographs (Fig. 2). In addition other phases are also analyzed from the x-ray diffraction patterns. These include hematite, dicalcium silicate and dicalcium ferrite. However, at 1300°C the x-ray diffraction patterns do not show any evidence of SFCA except at basicities of 2 and 2.5, whereas needles of SFCA are present in the micrographs (Fig. 1e-f).
 | |
| Fig. 1: | Optical micrographs of sinters produced from itakpe ore at the temperatures and basicities shown x 100 the microstructures consist of relict hematite, magnetite, SFCA, dicalcium silicate + glass |
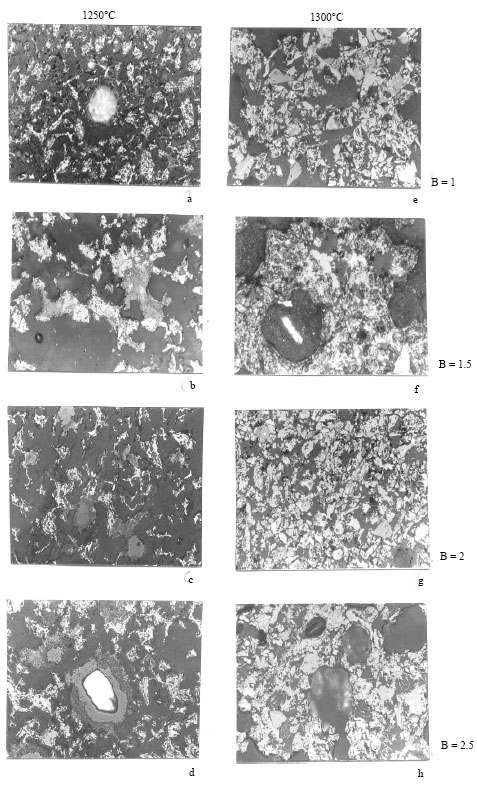 | |
| Fig. 2: | Optical micrographs of sinters produced from chokochok ore at the temperatures and basicities shown x100. The microstructures consist of relict hematite, magnetite, SFCA, dicalcium silicate + glass. The white patches in a, d and f are unreacted lime. Relative to microstructure in Fig. 1, the structures in this ore |
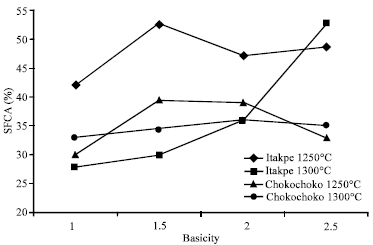 | |
| Fig. 3: | Percentage SFCA vs basicity |
In the Chokochoko ore sinters produced under the same conditions, much less SFCA are obtained (than in Itakpe Ore) as shown in Fig. 2 in both the x-ray diffraction patterns and the micrograph. From the diffraction patterns analyses more SFCA appears to be formed at 1250°C than at 1300°C. Magnetite is also observed in these sinters.
A summary of the results is presented in Fig. 3 in which the proportion of SFCA in the samples have been quantitatively estimated using x-ray diffraction internal standard method described in the experimental techniques. The results in this figure show that in the Itakpe ore sinter, the proportion of SFCA increases with increase in basicity at 1250°C. When the sintering temperature increases to 1300°C, same trend is still observed but SFCA contents are lower at basicities of 1, 1.5 and 2 but at basicity of 2.5, instead of a decrease, there is an increase over what is obtained at 1250°C. In the Chokochoko Ore sinters however, Fig. 3 also shows that less SFCA content is present than in Itakpe Ore sinters and there is not much significant difference between sintering at 1250 and 1300°C.
DISCUSSION
The results of this study can only be discussed when compared with results obtained when a two sinters are likely to give the same results when used in the blast furnace. However, when the conditions under which they have been produced are compared (1250°C, basicity 1.9), the microstructure in the sinter from pure hematite ore has been obtained more easily and at a less cost than the one from a mixture of hematite and magnetite. The higher temperature and more time needed purely hematite Ore is used. The micrograph obtained from a previous work (Price and Wasse, 1972) in which a purely hematite ore is used is shown in Fig. 4. When this micrograph is compared with that of Fig. 1h. (Itakpe Ore, 1300°C and basicity of 2.5), virtually the same phases are observed and the in the case of Itakpe Ore mean more coke would be needed in the raw mix to produce this sinter and therefore higher cost of production. The only reason for this is the magnetite content of the Itakpe Ore which inhibits the development of SFCA in sinters. The effect of magnetite had earlier been likened to that of sintering in reducing atmosphere when Fe2+ ions in the sinter melt lead to lowering of the viscosity of the melt since Fe2+ is a network modifier. Also, it has been proposed that SFCA does not precipitate out of a low viscous melt (Egundebi and Whiteman, 1989; Egundebi, 1987). Therefore, wherever magnetite is present in an ore, the proportion of SFCA in the final sinter piece will be reduced.
From Fig. 3, that the % SFCA is still rising at 1300°C and basicity of 2.5 ever above what was obtained at 1205°C, appears to be an anomaly. The reason for this is the increase in basicity (or specifically the proportion of CaO in the mix) which has counteracted the effect of rising temperature in lowering viscosity.
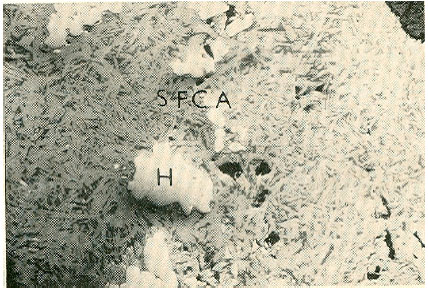 | |
| Fig. 4: | Optical micrograph of sinter produced from 100% hematite ore at temperature of 1250°C in air x100 the microstructure consists essentially needle SFCA, dicalcium silicate and relict hematite (H) |
Though Ca2+ is a network modifier like Fe2+, its effect in lowering melt viscosity is modified by the fact that it increases the melting point of the melt and the effective viscosity is higher than what it should be. Therefore, at a certain proportion of CaO, the effect of increasing CaO in the mix and hence increase in basicity, is more than effect of increase in temperature. So in this work, at basicity of mix greater than 2, melt viscosity rises with increase in temperature (up to 1300°C) and the % SFCA in the sinter also rises. Sintering at a basicity of 2.5 might be considered expensive but it is the only way out for the effective utilization of the itakpe Ore to get optimum results in the blast furnace.
The results of this study are by no means conclusive since presence of Al2O3 and MgO would modify the microstructure and thereby the properties of sinter and these would come in especially from coke during industrial sintering. Coke is also the source of reducing gas and this means that proportion of SFCA that could be obtained in real sintering will be less than experimental one.
CONCLUSIONS
| • | Under similar sintering conditions, the Itakpe iron ore is better than Chokochoko ore for producing sinters that would give optimum performance in the blast furnace. This is on the basis of the microstructures developed during sintering. |
| • | Magnetite contained in the ores lowers the proportion of SFCA that would have otherwise been obtained in the sinters. The higher the proportion of Magnetite, the lower the SFCA content. |
| • | A sinter that contains a high proportion of SFCA can be obtained from Itakpe Ore, but at a much higher cost than if the ore had been wholly hematite. |
REFERENCES
- Heng, H.L., 2005. Effect of raw material composition on the sintering properties. ISIJ Int., 45: 551-559.
CrossRef - Loo, C.E. and D.J. Wong, 2005. Fundamental factors determining laboratory sintering results. ISIJ Int., 45: 449-458.
Direct Link - Mansurova, N.R., T.Y. Malysheva, V.V. Korovushkin, M.F. Gibadulin, V.P. Lekin and V.A. Gostenin, 2006. Comparative analysis of the mineralogical composition and metallurgical properties of sinters obtained from iron-ore-bearing charges of different origins. Metallurgist, 50: 510-518.
CrossRefDirect Link - Yang, L.X., 2005. Sintering fundamentals of magnetite alone and blended with hematite and hematite goethite ore. ISIJ Int., 45: 469-476.
Direct Link








