ABSTRACT
The present study has reported the isolation, characterization, antioxidant and antiulcer activity of a new triterpenoid extracted from Cucurbita pepo seeds. The seed extract was subjected to column chromatography for the isolation of constituents. The isolated compound was characterized by Infra red, 1Hydrogen Nuclear Magnetic Resonance, 13Carbon Nuclear Magnetic Resonance which allow the identification of isotopic hydrogen and carbon atoms in a molecule, respectively. The isolated compound was a tetracyclic triterpenoid which possesses basic cucurbitacin skeleton characterized as 19-(10→9β)-abeo-10α-lanost-5-ene. The isolated compound showed maximum antioxidant activity i.e. 72.8±0.15% by 1, 1 diphenyl -2- picryl hydrazyl method at 300 μg mL-1 as compared to ascorbic acid. Further, it was evaluated for anti ulcerogenic activity, extracted triterpenoid showed optimum percentage inhibition i.e., 55.7, 67.1 and 59.1% by Pyloric Ligation, Water Immersion Stress and NSAID (Indomethacin) induced ulcer method at 300 μg mL-1 dose in rats. It can be inferred from the above study that the extracted tetracyclic triterpenoid have potent anti-ulcer activity due to its antioxidant potential and can be used as natural antiulcerogenic agent.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=rjphyto.2011.70.79
INTRODUCTION
Cucurbitaceae family is commonly known as family of gourds, melons or pumpkins generally climbing plant family composing 118 genera and 825 species having wide distribution in warmer region of world. Plants of this family have many medicinal and nutritional benefits. Recent studies revealed that many plant of family cucurbitaceae like Cucurbita pepo and Trichosanthes Cucumeria show potent hepatoprotective activity (Sunilson et al., 2009). Curcurbita pepo (pumpkin) also belongs to Cucurbitaceae family (Shah et al., 2010). It is used as a vegetable for human consumption and also use in traditional medicine (Caili et al., 2006). Seeds of Cucurbita pepo are used in the therapy of minor disorders of the prostate gland and the urinary bladder (Bombardelli and Morazoni, 1997). Cucurbita pepo has received considerable attention in recent years because of the nutritional and health values of the seeds. The seeds are excellent source of protein and also pharmacological activity such as antidiabetic, anti fungal and antioxidant (Atuonwu and Akobundu, 2010). Diets rich in pumpkin seeds have also been associated with lower levels of gastric, breast, lung and colorectal cancer (Huang et al., 2004) and also can be used as a potent vermifuge (Applcquist et al., 2006). Cucurbita maxima (another member of this family) possesses β-sitosterol, beta-carotene and stigmasterol, which are thought to be the major contributing factors against diabetes (Karim et al., 2011)
Seeds and fruit parts of cucurbits are reported to possess purgatives, emetics and antihelmintics properties due to the secondary metabolite cucurbitacin content (Bisognin, 2002). Recently various functional components in the seeds are reported by Toyama et al. (2008) Cucurbitacins are also important functional component found in Cucurbitaceae and constitute a group of diverse triterpenoid substances which are well known for their bitterness and toxicity. Seeds and fruit parts of cucurbits are reported to possess purgatives, emetics and antihelmintics properties due to the secondary metabolite cucurbitacin content (Bisognin, 2002). They are highly oxygenated, tetracyclic triterpenes containing a cucurbitane skeleton characterized as 19-(10→9β)-abeo-10α-lanost-5-ene (Chen et al., 2005). The cucurbitacins are of great interest because of the wide range of biological activities they exhibit in plants and animals. They are predominantly found in the family Cucurbitaceae but are also present in several other families of the plant kingdom (Wang et al., 2007). Despite their toxicity, species of the plants in which they are found have been used for centuries in various pharmacopoeias. A number of compounds of this group have been investigated for their cytotoxicity, hepatoprotective, anti-inflammatory and cardiovascular effects (Bernard and Olayinka, 2010). The cucurbitacins are arbitrarily divided into twelve categories which range from cucurbitacins A to T. The various cucurbitacins differ with respect to oxygen functionalities at various positions (Chen et al., 2009).
Several cucurbitane and hexanorcucurbitane glycosides and other types of triterpenoids have been isolated from the fruits of Cucurbita pepo (Ge et al., 2006). Several phytochemicals such as cucurbitacins, kuguacins have been isolated from the vines and leaves of Momordica charantia (Chen et al., 2009). Several multiflorane triterpenoids have been isolated from the seed extract of Trichosanthes kirilowii. The most predominant ones include karounidiol and its 3-O-benzoate derivative (Akihisa et al., 2001). However, no study has been reported on the constituents of the seeds of Cucurbita pepo. Thus the present study was aimed at isolation of cucurbitane type of triterpenoids from the seeds of Cucurbita pepo followed by the evaluation of its anti ulcer potential.
MATERIALS AND METHODS
Plant material: The seeds were purchased from the Khari Baoli (spice market) Delhi (India) in August 2009. The healthy looking seeds were selected for authentication and voucher specimen number 0357 has been deposited in the Botanical and Environmental Science Department, Guru Nanak Dev University, Amritsar (India). The seeds were cleaned, washed, dried at low temperature and powered.
Drugs and chemicals: All chemical reagents used were of analytical grade which were procured from different companies (Loba Chem, Mumbai and Merck Limited, Mumbai).
Extraction: The powdered seeds were extracted for 72 h with methanol at room temperature. The solvent was filtered off and residue macerated again with the fresh solvent. Both solvents were combined and concentrated under reduced pressure on a rotary evaporator (Heidolph) at 40°C. The concentrated filtrate was suspended in distilled water and partitioned successively with hexane. The aqueous layer was separated and concentrated on water bath. The crude extracts were used for isolation.
Isolation: The isolation was carried out by column chromatography. The methanolic extract (2 mL) was mixed with 5 g of silica gel (60-120 mesh). Then the mixture was evaporated to dryness in rotary evaporator at 40°C. The cotton wool was used as a support for the column. The column was packed by wet packing method. Firstly the slurry of the adsorbent was prepared in the same solvent that was being used in the chromatographic process. The slurry was added to the column gradually up to approximately two thirds of the column height and then the sample mixture was loaded at the top. The gradient elution method was followed. Firstly the column was eluted with a solvent of low polarity i.e. hexane. Then the column was eluted with solvent systems of increasing polarity such as hexane: ethyl acetate (99:1), hexane: ethyl acetate (98:2) and hexane: ethyl acetate (97:3) and so on. Thin Layer Chromatography profiling was done simultaneously in an appropriate solvent system (Bhujbal et al., 2008). The fractions of similar Rf value was pooled and concentrated. The fractions eluted were subjected to TLC profiling again. Fractions giving single spot in the TLC were regarded as pure. Further these fractions were used for the characterization of the compound by IR, 1H NMR and 13C NMR. The isolated cucurbitane triterpenoid was further evaluated for its antioxidant and anti ulcer potential.
Characterization of compound: Four fractions F1, F2, F3 and F4 were collected and subjected to Liebermann-Buchard’s test. F1 fraction gave the positive test which indicates the presence of triterpenoids and the fraction was further used for characterization by IR, 1H NMR and 13C NMR.
The IR spectrum was carried out by placing the liquid sample directly in the sample cell without any prior preparation using Bruker IR Spectrophotometer. 1H NMR spectra and 13C NMR spectra were recorded at 400 MHz on Bruker AC 400F instrument.
DPPH radical scavenging activity: Various methods are used to test antioxidant activity but the most widely used methods are those that involve generation of free radical species which are then neutralized by antioxidant substance (Chanda et al., 2011) The free radical scavenging activity of isolated triterpenoid was determined by using 1,1-diphenyl-2 picryl-hydrazyl (DPPH) method (Sreejayan and Rao, 1996).
Experimental design for pyloric ligation induced gastric ulcer (PL): Animals were divided into 6 groups, each comprising of 6 rats.
| Group I | : | Administered vehicle (normal saline 0.9% w/v, p.o.) 1 h before pyloric ligation on the day of experiment |
| Group II | : | Sham control group subjected to surgical procedure without pyloric ligation |
| Group III | : | Disease control group subjected to pyloric ligation for the induction of ulcer |
| Group IV | : | Administered standard (ranitidine 50 mg kg-1, p.o.) 1 h before pyloric ligation on the day of experiment |
| Group V | : | Administered isolated triterpenoid (150 mg kg-1, p.o.) 1 h before pyloric ligation on the day of experiment |
| Group VI | : | Administered isolated triterpenoid (300 mg kg-1, p.o.) 1 h before pyloric ligation on the day of experiment |
The pylorus was ligated according to the method of Shay et al. (1945) and Bose et al. (2003).
Experimental design for water immersion stress induced gastric ulcer (WIS): Animals were divided into 5 groups, each comprising of 6 rats.
| Group I | : | Administered vehicle (normal saline 0.9% w/v, p.o.) 1 h before water immersion stress |
| Group II | : | Disease control group subjected to water immersion stress for the induction of gastric ulcers |
| Group III | : | Administered standard (ranitidine 50 mg kg-1, p.o.) 1 h before water immersion stress |
| Group IV | : | Administered isolated triterpenoid (150 mg kg-1, p.o.) 1 h before water immersion stress |
| Group V | : | Administered isolated triterpenoid (300 mg kg-1, p.o.) 1 h before water immersion stress |
The rats were fasted 24 h prior to experiment and test samples were administered 1 h before stress induction. Rats were immobilized in a stress cage and then immersed to the level of the xiphoid in a water bath at 23±0.2°C for 4 h (Hayase and Takeuchi, 1986). After 4 h animals were removed and sacrificed. The stomach of each animal was removed and cut open along the greater curvature and pinned on wooden board after washing it with running tap water. Then ulcerative index and percentage ulcer protection were calculated.
Experimental design for NSAID (Indomethacin) induced ulcer model (NIU): Animals were divided into 5 groups, each comprising of 6 rats.
| Group I | : | Administered vehicle (normal saline 0.9% w/v, p.o.) 30 min before Indomethacin induced ulcers |
| Group II | : | Disease control group administered Indomethacin (25 mg kg-1 p.o.) for the induction of gastric ulcers |
| Group III | : | Administered standard (ranitidine 50 mg kg-1, p.o.) 30 min before Indomethacin induced ulcers |
| Group IV | : | Administered isolated triterpenoid (150 mg kg-1, p.o.) 30 min before Indomethacin induced ulcers |
| Group V | : | Administered isolated triterpenoid (300 mg kg-1, p.o.) 30 min before Indomethacin induced ulcers |
Normal saline, ranitidine, isolated triterpenoid were given orally and 30 min later Indomethacin was administrated to all the groups. Six hours later, the animals were killed by decapitation. The stomachs were removed, opened along the great curvature and washed with tap water to remove gastric contents and examined under a dissecting microscope with square-grid eyepiece to assess the formation of ulcers. For each stomach, ulcerated and total areas were measured as mm2 and the ulcer indexes for each stomach were calculated (Dengiz and Gursan, 2005).
Estimation of gastric volume and free and total activity changes in PL mode: Four hours after ligation, stomachs were dissected out and contents were collected into measuring cylinder to measure the volume of gastric contents. The gastric contents were centrifuged and subjected to titration for estimation of free and total acidity. One mL of the supernatant liquid was pipette out and diluted to 10 mL with distilled water. The solution was titrated against 0.01 N NaOH using Topfer’s reagent (Sachin and Archana, 2009) as indicator, to the endpoint when the solution turned to orange colour. The volume of NaOH needed was taken as corresponding to the free acidity. Titration was further continued by adding 1% solution of phenolphthalein till the solution gained pink color. The volume of NaOH required was noted and was taken as corresponding to the total acidity. The sum of the two titrations was total acidity (Rajkapoor et al., 2002). Acidity was expressed as:
Estimation of gastric ulcerative index changes in PL, WIS, NIU model: Ulcerative index was measured by method of Takagi et al. (1969).
Statistical analysis: All the results were expressed as Mean±Standard Error of Means (SEM). The data was statistically analyzed by one way Analysis of Variance (ANOVA) followed by Tukey’s multiple range tests by using Sigmastat Version-2.0 Software. The p-value<0.05 was considered to be statistically significant.
RESULTS
The IR spectrum was carried out by Bruker IR Spectrophotometer. 1H NMR spectra and 13C NMR spectra were recorded at 400 MHz on Bruker AC 400F instrument.
IR (Liquid): It showed characteristic peaks at 3207 cm-1, 2928 cm-1, 2833 cm-1, 1514 cm-1, 1720 cm-1, 1023 cm-1 indicating the presence of ketone (C = O) and alcoholic (R-OH) group.
1HNMR (400 MHz, CDCl3): δ 0.66-1.18 (m, 21H, -CH3), δ 1.22-1.48 (m, 4H, H7, H8, H9, H10), δ 1.49-1.59 (m, 5H, H3, H11, H18, H19, H20), δ 1.81-1.87 (m, 3H, H2, H4, H5), δ 1.98-2.29 (m, 5H, H12, H13, H15, H16, H17), δ 3.55 (m, 1H, H14), δ 5.12-5.19 (brs, 3H, -OH), δ 5.33-5.34 (s, 2H, H1,H2).
13C NMR (400 MHz, CDCl3): δ 19.88, 21.25, 24.08, 24.31, 25.55, 28.25, 28.92, 30.31, 31.46, 31.55, 33.86, 36.25, 36.54, 38.27, 39.78, 41.49, 42.43, 45.75, 49.15, 51.82, 56.89, 57.78, 71.91, 124.73, 124.84 (C = C), 129.39, 129.56 (C = C), 139.72 (C = O), 141.67 (C = O).
The DEPT spectra revealed the presence of 29 carbon signals due to seven primary, five secondary, nine tertiary and eight quaternary carbons (including two carbonyl carbons) which were assigned to a triterpene skeleton. Careful analysis of the NMR data indicated that it was a highly oxygenated cucurbitane derivative. The structure of the isolated compound was elucidated by detailed 1H and 13C NMR spectroscopy and by comparison of the spectral data with published results. The structure of Triterpenoid is shown in Fig. 1.
The triterpenoid was further evaluated for its antioxidant and anti ulcer potential in various rat models. The percentage scavenging of DPPH was 72.8% at 300 μg mL-1 as comparable to standard (ascorbic acid) (Table 1). In pyloric ligated rats, there was an increase in the gastric volume, free and total acidity and ulcerative index as compared to the sham control group. The triterpenoid showed reduction in gastric secretion, free and total acidity and ulcerative index. But only highest dose i.e., 300 mg kg-1 showed significant reduction in the above parameters which was comparable to the standard drug ranitidine (Table 2). It caused 39.1 and 55.7% inhibition of ulcers at the dose of 150 and 300 mg kg-1, respectively in PL model.
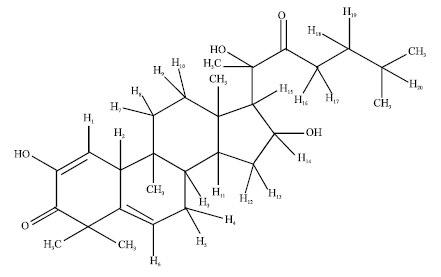 | |
| Fig. 1: | Structure of triterpenoid |
| Table 1: | Percentage scavenging of DPPH radical |
 | |
| Values are the average of triplicate experiments and represented as Mean±SEM | |
| Table 2: | Effect of isolated triterpenoid on gastric secretion, free acidity and total acidity in pylorus ligation induced gastric ulcer in rats |
 | |
| Values are Mean±SEM, n = 6 animals in each group; ap<0.05 as compared with sham control group. bp<0.05 compared with disease control groups, cp<0.05 compared with ranitidine treated group | |
In water immersion stress induced ulcer model the triterpenoid showed reduction in ulcerative index but only highest dose i.e., 300 mg kg-1 showed significant reduction which was comparable to the standard drug ranitidine (Table 3). It caused 52.1 and 67.1% inhibition of ulcers at the dose of 150 and 300 mg kg-1, respectively in water immersion stress induced model (Table 3).
Pretreatment with the Cucurbita pepo extract, significantly (p<0.01) reduced the volume of gastric secretions 1.35±0.89, 2.51±0.56 and 1.91±0.67 at the doses of 50, 150 and 300 mg kg-1 respectively. In addition, total acidity and free acidity were also reduced significantly (p<0.01) in a dose dependant manner. Further it is observed that pyloric ligation has caused gastric ulcerations and pretreatment with Cucurbita pepo extract has reduced them significantly (p<0.01) in a dose dependent manner. The gastroprotection offered by the test extract was comparable to that of the standard drug Ranitidine. In NSAID (Indomethacin) induced ulcer model the triterpenoid showed reduction in ulcerative index but only highest dose i.e., 300 mg kg-1 showed significant reduction which was comparable to the standard drug ranitidine (Table 3).
| Table 3: | Effect of isolated triterpenoid on ulcerative index and percentage inhibition in PL, WIS and NSAID induced gastric ulcer in rats |
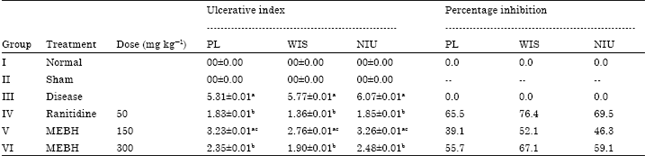 | |
| Values are Mean±SEM, n = 6 animals in each group; ap<0.05 compared with sham control group, bp<0.05 compared with PL and WIS groups respective coloums, cp<0.05 compared with ranitidine treated group. PL: Pyloros ligation, WIS: Water immersion stress, NSAID: Non-steroidal anti-inflammatory drug | |
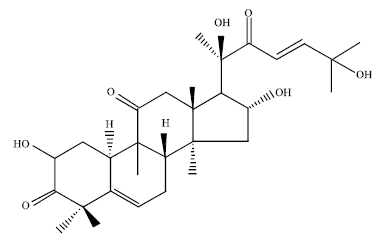 | |
| Fig. 2: | Structure of cucurbitacin D |
It caused 46.3 and 59.1% inhibition of ulcers at the dose of 150 and 300 mg kg-1, respectively in NSAID (Indomethacin) induced ulcer model.
DISCUSSION
Cucurbitacin B is isolated from fruits and roots of Trichosanthes kirilowii Maximowicz (Cucurbitaceae family). Naturally occurring cucurbitacins are cytotoxic terpene sterols containing a cucurbitane skeleton characterized by a 19-(10→9β)-abeo-10α-lanost-5-ene. These Cucurbitacins exhibit antiproliferative and cytotoxic anticancer activity both in vitro and in vivo (Yin et al., 2008). New trinorcucurbitane triterpene isolated from the methyl alcohol extract of the stems of Momordica charantia (Chen et al., 2010).
The isolated compound is a tetracyclic triterpenoid. It possess the basic cucurbitacin skeleton 19-(10→9β)-abeo-10α-lanost-5-ene. The Structure resembles with the cucurbitacins isolated from various plants of the family Cucurbitaceae such as cucurbitacin D (Fig. 2). The isolated compound differs from cucurbitacin D with respect to a ketone group, a double bond and an OH group at C-24.
The triterpenoid was further evaluated for its antioxidant and in vivo antiulcer activity in pyloric ligation, water immersion stress and NSAID induced ulcer models. Ulcer index parameter was used for the evaluation of anti-ulcer activity since ulcer formation is directly related to factors such as gastric volume, free and total acidity (Khayum et al., 2009). The ulcer formation in each of these models occurs by different mechanisms. Hence, it is not possible to propose a single mechanism for antiulcer effect. The reflex or neurogenic effect has also play an important role in the formation of gastric ulcer in this model (Goswani et al., 1997). In water immersion stress induced ulcer model ulcers are formed as a result of disturbance of gastric secretion, alteration in microcirculation of gastric mucosa and abnormal gastric motility (Kitagawa et al., 1979). It has been suggested that active oxygen species may be involved in the pathogenesis of gastric mucosal injuries (Szelenyi and Brune, 1988). The isolated triterpenoid showed dose dependent antiulcer effect in pyloric ligation, water immersion stress and indomethacin induced ulcer models.
The induction of stress generates free radicals which cause mucosal damage and change in antioxidant enzymes (Das and Banerjee, 1993). Consequently, some radical scavengers have shown to possess a protective effect against the mucosal injuries induced by active oxygen species (Oka et al., 1991). The decrease in ulcerative index suggests the ability of the compound to protect the gastric mucosa against free radical mediated tissue injury. Thus the action of isolated triterpenoid may be through free radical scavenging mechanism.
CONCLUSION
In the present study, it may be concluded that the triterpenoids extracted from the seeds of Cucurbita pepo possessed potent anti-ulcer activity due to its antioxidant potential and can be used as a future natural antiulcerogenic agent.
ACKNOWLEDGMENTS
Thanks to Professor A.C. Rana and all faculty members of Rayat Institute of Pharmacy for their encouragement and support. We are also grateful to Rayat and Bahra Educational and Research Trust for their unconditional help to carryout this project.
REFERENCES
- Akihisa, T., H. Tokuda, E. Ichiishi, T. Mukainaka and M. Toriumi et al., 2001. Anti-tumor promoting effects of multiflorane-type triterpenoids and cytotoxic activity of karounidiol against human cancer cell lines. Cancer Lett., 173: 9-14.
PubMed - Applcquist, W.L., B. Avula, B.T. Schancberg, Y.H. Wang and I.A. Khan, 2006. Comparative fatty acid content of seeds of four Cucurbita species grown in a common (shared) garden. J. Food Compos. Anal., 19: 606-611.
CrossRef - Atuonwu, A.C. and E.N.T. Akobundu, 2010. Nutritional and sensory quality of cookies supplemented with defatted pumpkin (Cucurbita pepo) seed flour. Pak. J. Nutr., 9: 672-677.
CrossRefDirect Link - Bernard, S.A. and O.A. Olayinka, 2010. Search for a novel antioxidant, anti inflammatory/analgesic or anti-proliferative drug: Cucurbitacins hold the ace. J. Med. Plants Res., 4: 2821-2826.
Direct Link - Bhujbal, S.S., S.S. Chitlange, A.A. Suralkar, D.B. Shinde and M.J. Patil, 2008. Antiinflammatory activity of an isolated flavanoid fraction from Celosia argentea linn. J. Med. Plants Res., 2: 52-54.
Direct Link - Bisognin, D.A., 2002. Origin and evolution of cultivated cucurbits. Ciencia Rural, 32: 715-723.
CrossRefDirect Link - Bose, M., V.M. Motghare, G.N. Dakhale and A.V. Turankar, 2003. Antiulcer activity of levcromakalim and nicorandil in albino rats: A comparative study. Pol. J. Pharmacol., 55: 91-95.
PubMed - Fu, C., H. Shi and Q. Li, 2006. A review on pharmacological activities and utilization technologies of pumpkin. Plant Foods Hum. Nutr., 61: 70-77.
CrossRefPubMedDirect Link - Chanda, S., R. Dave and M. Kaneria, 2011. In vitro antioxidant property of some Indian medicinal plants. Res. J. Med. Plant, 5: 169-179.
CrossRefDirect Link - Chen, J.C., M.H. Chiu, R.L. Nie, G.A. Cordell and S.X. Qiu, 2005. Cucurbitacins and cucurbitane glycosides: Structures and biological activities. Nat. Prod. Rep., 22: 386-399.
CrossRefPubMedDirect Link - Chen, J.C., W.Q. Liu, L. Lu, M.H. Qiu and Y.T. Zheng et al., 2009. Kuguacins F-S, cucurbitane triterpenoids from Momordica charantia. Phytochemistry, 70: 133-140.
CrossRefDirect Link - Chen, C.R., Y.W. Liao, L. Wang, Y.H. Kuo and H.J. Liu et al., 2010. Cucurbitane triterpenoids from Momordica charantia and their cytoprotective activity in tert-butyl hydroperoxide-induced hepatotoxicity of HepG2 cells. Chem. Pharm. Bull., 58: 1639-1642.
CrossRefPubMedDirect Link - Das, D. and R.K. Banerjee, 1993. Effect of stress on the antioxidant enzymes and gastric ulceration. Mol. Cell. Biochem., 125: 115-125.
CrossRefDirect Link - Dengiz, G.O. and N. Gursan, 2005. Effects of Momordica charantia L. (Cucurbitaceae) on indomethacin induced ulcer model in rats. Turk. J. Gastroenterol., 16: 85-88.
PubMed - Goswani, S., S. Jain and D. Santani, 1997. Antiulcer activity of cromakalim against experimentally induced gastric and duodenal ulcers in rats and guinea pigs. J. Pharm. Pharmacol., 49: 195-199.
PubMed - Wang, J., X. Zhou, Y. Cao, J. Xiao, E. Ma, Y. Deng and D. Chen, 2007. The antitumor activities of Cucurbitacin Liposome for injection both in vitro and in vivo. Asian J. Trad. Med., 2: 98-103.
Direct Link - Hayase, M. and K. Takeuchi, 1986. Gastric acid secretion and lesion formation in rats under water-immersion stress. Dig. Dis. Sci., 31: 166-171.
CrossRefPubMedDirect Link - Huang, X.E., K. Hirose, K. Wakai, K. Matsuo and H. Ito et al., 2004. Comparison of lifestyle risk factors by family history for gastric, breast, lung and colorectal cancer. Asia. Pac. J. Cancer Prev., 5: 419-427.
PubMedDirect Link - Khayum, M.A., K. Nandakumar, T.S. Gouda, S.M. Khalid, V.N. Rao and S.M.S. Kumar, 2009. Antiulcer activity of stem extracts of Tinospora malabarica (Lamk.). Pharmacologyonline, 1: 885-890.
Direct Link - Kitagawa, H., M. Fjuwara and Y. Osumi, 1979. Effect of water immersion stress on gastric secreton and mucosal blood flow in rats. Gastroenterology, 77: 298-302.
PubMed - Sunilson, J.A.J., M. Muthappan, A. Das, R. Suraj, R. Varatharajan and P. Promwichit, 2009. Hepatoprotective activity of Coccinia grandis leaves against carbon tetrachloride induced hepatic injury in rats. Int. J. Pharmacol., 5: 222-227.
CrossRefDirect Link - Oka, S., K. Ogino, T. Hobara, S. Yoshimura, Y. Okazaki, T. Takemoto and Y. Iida, 1991. Effects of various mucosal protective drugs on diethyldithiocarbamate-induced antral ulcer in rats. Eur. J. Pharmacol., 197: 99-102.
PubMed - Rajkapoor, B., R. Anandan and B. Jayakar, 2002. Antiulcer effect of Nigella sativa Linn. against gastric ulcer in rats. Curr. Sci., 82: 177-179.
Direct Link - Sachin, S.S. and R.J. Archana, 2009. Antiulcer activity of methanolic extract of Erythrina indica Lam. leaves in experimental animals. Pharmacognosy Res., 6: 396-401.
Direct Link - Shay, H., S.A. Komarow, S.S. Fels, D. Meranze, M. Gruenstein and H. Siplet, 1945. A simple method for the uniform production of gastric ulceration in the rat. Gastroenterology, 5: 43-61.
Direct Link - Shah, B.N., A.K. Seth and R.V. Desai, 2010. Phytopharmacological profile of Lagenaria siceraria: A review. Asian J. Plant Sci., 9: 152-157.
CrossRefDirect Link - Sreejayan, N. and M.N. Rao, 1996. Free radical scavenging activity of curcuminoids. Drug Res., 46: 169-171.
PubMed - Szelenyi, I. and K. Brune, 1988. Possible role of oxygen free radicals in ethanol-induced gastric mucosal damage in rats. Dig. Dis. Sci., 33: 865-871.
PubMedDirect Link - Takagi, K., S. Okabe and R. Saziki, 1969. A new method for the production of chronic gastric ulcer in rats and the effect of several drugs on its healing. Jpn. J. Pharmacol., 19: 418-426.
PubMed - Karim, A., M.N. Sohail, S. Munir and S. Sattar, 2011. Pharmacology and phytochemistry of Pakistani herbs and herbal drugs used for treatment of diabetes. Int. J. Pharmacol., 7: 419-439.
CrossRef - Toyama, J., H. Tanaka, A. Horie, T. Uchiyama and R. Akashi, 2008. Purification and characterization of Anti-H lectin from the seed of Momordica charantia and the inter-specific differences of hemagglutinating activity in Cucurbitaceae. Asian J. Plant Sci., 7: 647-653.
CrossRefDirect Link








