ABSTRACT
The present study aimed to study the effect of acute oxidative stress on serum isoenzyme expression and protein profile induced by acute paraquat (PQ) toxicity in female rats. The protective role of α-Lipoic Acid (LA) administration has been challenged. Acute paraquat toxicity was induced by administration of 1/4 LD50 in adult female wistar rats. α-Lipoic acid was orally supplemented 2 h prior PQ administration at a dose equivalent to 10 mg/100 g body weight. Isoenzyme expression of serum enzymes, namely MDH, ACP, G6PDH, ASAT and EST as well as serum protein patterns were determined 24 h post administration. The present data revealed a significant rise in total serum activity of MDH, ACP and ASAT but insignificant change in total activity of G6PDH and EST in PQ group versus the control group. Fractional activities of serum isoenzymes exhibited the same trend as that demonstrated in serum total activity except in case of EST. α-LA administration exhibited a high protective effect against the changes in both total and fractional activities of serum ACP and ASAT but a considerable protection in case of MDH. The present investigation supports the idea that the estimation of the fractional rather than the total activity of an enzyme is more reliable in reflecting the molecular consequences of acute oxidative stress. The present data has also exposed a wid?e-range variation in serum protein patterns duo to PQ and/or lipoic acid treatment, which indirectly reflects a parallel variation in the process of gene expression and/or proteolytic activity.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/rjet.2008.53.66
URL: https://scialert.net/abstract/?doi=rjet.2008.53.66
INTRODUCTION
Recent years have shown considerable interests in the reactions of the oxidants with bio-molecules. Reactive oxygen species are produced by a variety of factors including some chemicals or ionizing radiation as well as the by-products of aerobic respiration (Cadenas, 1989; Halliwell and Gutteridge, 1999; Jensen, 2003; Valko et al., 2005). Reactive Oxygen Species (ROS), such as superoxide, hydroxyl radicals and hydrogen peroxide (H2O2) can damage cellular lipids, proteins and DNA (Siems et al., 1995; Berlett and Stadtman, 1997; Halliwell and Gutteridge, 1999; Marnett, 2000; Bergamini et al., 2004; Stadtman, 2004; Valko et al., 2006, 2007; Hwang and Kim, 2007).
Oxidative stress arises when concentrations of ROS exceed the cellular ability to remove ROS and repair cellular damage and ultimately results in the widespread oxidation of biomolecules, including DNA and proteins. Protein oxidation, in turn, results in tertiary structural alterations that promote protein aggregation and amyloid formation (Shinall et al., 2005). Oxidants-other than the hydroxyl radical-generally exhibit selectivity in the type of biomolecule they react with. If such reactions had the possibility to proceed unchecked, then the result is structural alteration of proteins, inhibition of enzymatic activity and interference with the regulatory functions and eventual cell death (Jensen, 2003).
Paraquat (methyl viologen: 1,1`-dimethyl-4,4`-bipyridinium dichloride), as an effective and widely used herbicide, is considered as a potent redoxcycling chemical agent, which can accept electrons and pass these electrons to oxygen. Thus, it could be participating in generating intracellular superoxide radicals (Margolis et al., 2000; Suntres, 2002; Jung and Kim, 2003; Mollace et al., 2003; Dinis-Oliveira et al., 2006, 2007).
α-Lipoic acid (LA; 1, 2-dithiolane-3-pentanoic acid), which plays an essential role in the mitochondrial dehydrogenase reactions, is present in all kinds of prokaryotic and eukaryotic cells and has recently gained considerable attention as an antioxidant (Biewenga et al., 1997). LA and its reduced form dihydrolipoic acid act as antioxidants not only through free radical quenching but also indirectly through recycling of other cellular antioxidants (Bilska and Wlodek, 2005). Free LA has not been detected in human beings because it is bound to proteins (Hermann et al., 1996). However, after therapeutic applications, free LA can be found in the circulation (Teichert and Preiß, 1995). It is likely that the therapeutic effects originate from free, unbound LA, provided by exogenous offer. Guimarães et al. (2007) reported that LA administered before torsion of the spermatic cord in male rats gave significant protective effects against ischemia/reperfusion injury by decreasing lipid peroxidation and regulating testicular reduced glutathione and plasma total antioxidant power levels.
Previous studies have supported the use of isoenzyme expression and activity, rather than total enzyme activity, for better understanding of the molecular basis of oxidative stress in male animals (El-Zayat and Amer, 2002; El-Zayat, 2007).
The present investigation aimed to (1) study the protective role of alpha-lipoic acid on acute paraquat-induced oxidative stress changes in isoenzyme patterns and activities of serum malate dehydrogenase (MDH), acid phosphatase (ACP), glucose-6-p-dehydrogenase (G6PDH), aspartate aminotransferase (ASAT) and esterase (EST) as well as serum protein pattern; (2) test the feasibility of using isoenzyme expression and activity beside total enzyme activity as additional markers for better understanding of the molecular basis of acute oxidative stress in female animals. Acute paraquat toxicity (1/4 LD50) has been used as experimental model for induction of acute oxidative stress, via production of superoxide radicals.
MATERIALS AND METHODS
Chemicals and Animal Specimens
Paraquat
Paraquat (methyl viologen: 1,1`-dimethyl-4,4`-bipyridinium) in a concentration of 20% was a product of Kafr El Zayat Chemical Company, Kafr El Zayat, Egypt).
Antioxidant
α-Lipoic acid was generously synthesized and donated by the staff members of the Organic Chemistry Lab, Chemistry Department, Cairo University.
Animals
Adult female wistar rats (Rattus norvegicus) were supplied from the animal house of National Organization for Biological Products and Vaccines, Cairo, Egypt. Animals were kept under natural conditions of illumination and temperature and had free access to standard pelleted diet and tap water. Rats were acclimatized to laboratory conditions for one week before onset of the experiment.
Experimental Design
Twenty adult female albino rats weighing 270-300 g (12 month old) were assigned for the present study. After the acclimatization period, they were divided into 4 groups as follows: The Control group (C) supplied orally with 0.5 mL of 1% DMSO (used as the solvent for α-lipoic acid); Lipoic acid-treated group (LA) given orally 10 mg/100 g body weight of α-lipoic acid dissolved in 0.5 mL of 1% DMSO; Paraquat-treated group (PQ) given orally a dose equivalent to 1/4 LD50 of paraquat (LD50= 23.31 mg/100 g body weight) and Paraquat and Lipoic acid-treated group (LAPQ) supplied orally with 10 mg/100 g body weight of α-lipoic acid dissolved in 0.5 mL 1% DMSO 2 h prior administration of a dose equivalent to 1/4 LD50 of paraquat.
Blood Sampling
Blood samples were collected from the retro-orbital plexus 24 h after treatment. Serum samples were collected and kept frozen at -20°C for subsequent electrophoretic separation of the isoenzymes of MDH, ACP, G6PDH, ASAT, EST as well as serum proteins.
Electrophoretic Separation of Isoenzymes
Serum samples were brought to room temperature one hour prior electrophoretic separation. Isoenzymes of MDH (EC 1.1.1.37), ACP (EC 3.1.3.2), G6PDH (EC 1.1.1.49), ASAT (EC 2.6.1.1) and EST (EC 3.1.1.1) were separated by polyacrylamide gel electrophoresis (PAGE) using specific buffers according to methods described by Guérin and Kerambrun (1982) for MDH and EST, by Beck and Feir (1989) for ACP, by Toncheva et al. (1982) for G6PDH and by Van Der Laarse (1980) for ASAT using Mini Protein II electrophoretic apparatus, Biorad. Isoenzymes were stained with FAST blue or MTT and scanned with Media Sci Image densitometry 700 Biorad and analyzed using the gel pro software (Ver. 3, USA, 1998). Fractional isoenzyme activities were calculated as percentage of total activity and presented in the form of IU/l.
Electrophoretic Separation of Serum Proteins
SDS-PAGE electrophoresis was carried out according to the method of Laemmli (1970). The wide range SDS-PAGE molecular weight pre-stained standard mixture (Biorad) was applied to the first well. Scanning was applied using gel pro software (ver. 3, USA, 1998), for Media Sci Image densitometry 700 Biorad.
Statistical Analysis
Data were statistically analyzed using one-way analysis of variance (ANOVA) followed by a post hoc test (LSD) according to Winter et al. (1991) using the SPSS statistical software package version 10.
RESULTS
Effect on Total and Isoenzyme Activity of Serum Malate Dehydrogenease (MDH)
Almost normal value of MDH activity has been recorded in LA-treated group versus the control value (Table 1). On the other hand, acute PQ-toxicity showed a significant increase in total serum MDH activity 24 h post administration as compared to both C and LA groups. α-Lipoic acid treatment 2 h prior paraquat administration (LAPQ) has shown a considerable improvement despite the fact that significantly higher levels still be demonstrated versus the control and LA values. This finding can be supported by the fact that a significant reduction in total serum MDH could be seen in LAPQ versus PQ group.
| Table 1: | Effect of acute toxicity of paraquat (1/4LD50) with or without α-lipoic acid supplementation on total and fractional isoenzyme activities of malate dehydrogenase (MDH) and acid phosphatase (ACP) in serum of adult female rat |
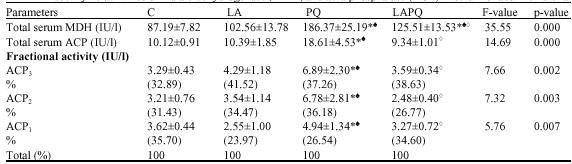 | |
| Data are presented as Mean±SD. No. of samples per group = 5. * = Significant difference versus C group at p<0.05 (ANOVA, LSD), ♠= Significant difference versus LA at p<0.05 (ANOVA, LSD), ◊ = Significant difference versus PQ at p<0.05 (ANOVA, LSD) | |
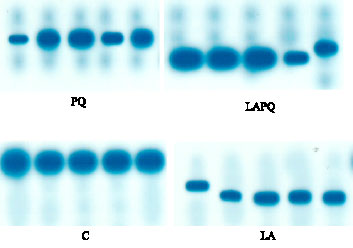 | |
| Fig. 1: | Isoenzyme pattern of serum malate dehydrogenase (MDH) in adult female rat as affected by acute paraquat toxicity and/or α-lipoic acid administration for 24 h. Individual lanes represent individual samples per group. C = Control group; LA = Lipoic acid group; PQ = Paraquat-treated group; LAPQ = Lipoic acid and paraquat-treated group |
The electrophoretic pattern of serum MDH revealed one single phenotype in the gel with highly similar electrophoretic mobility in the control group. Slight changes in the electrophoretic mobility could be demonstrated in LA and LAPQ groups versus the control one (Fig. 1).
As only one phenotype of MDH is expressed, the effect of oxidant and/or antioxidant is considered the same as that demonstrated in total serum MDH activity.
Effect on Total and Isoenzyme Activity of Serum Acid Phosphatase (ACP)
LA and LAPQ groups exhibited normal values versus the C group. On the other hand, acute PQ-toxicity showed a significant increase in total serum ACP activity 24 h post administration as compared to both C and LA groups (Table 1). Statistical analysis has also revealed a significant reduction in the total ACP activity in LAPQ group versus PQ-intoxicated group.
As regards ACP serum isoenzyme pattern and activity, Fig. 2 showed 3 distinct phenotypes on the gel with the same electrophoretic mobility in the control (C) and PQ groups. In opposition, LA group exhibited changes in ACP2 only while LAPQ group exhibited pronounced changes in the electrophoretic mobility of ACP2 and ACP3. In both LA and LAPQ groups, normal expression of the
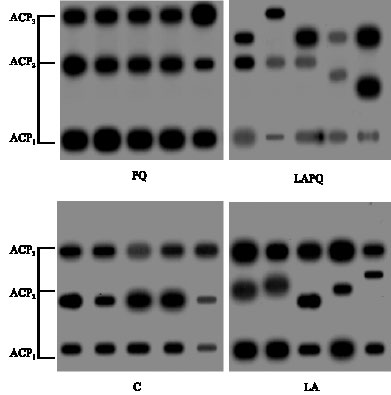 | |
| Fig. 2: | Isoenzyme pattern of serum acid phosphatase (ACP) in adult female rat as affected by acute paraquat toxicity and/or α-lipoic acid administration for 24 h. Individual lanes represent individual samples per group. C = Control group; LA = Lipoic acid group; PQ = Paraquat-treated group; LAPQ = Lipoic acid and paraquat-treated group |
three isoenzymes has been demonstrated against the respective control values (Table 1). On the other side, fractional activities of the three separated isoenzymes have shown a significant increase in PQ group versus both C and LA values. Statistical analysis has also revealed a significant reduction in the three fractional activities in LAPQ group versus PQ-toxicated group.
Effect on Total and Isoenzyme Activity of Serum Glucose-6-p-Dehydrogenase (G6PDH)
All treated groups (LA, PQ and LAPQ) exhibited a nonsignificant change in total and fractional activities of serum G6PDH 24 h after treatment versus the control (C) group (Table 2).
Two distinct phenotypes for serum G6PDH have been demonstrated in the gel with the same electrophoretic mobility in the control (C) group (Fig. 3). A slight change in electrophoretic mobility of serum G6PDH isoenzymes has been encountered in all treated groups versus the C group.
Effect on Total and Isoenzyme Activity of Serum Aspartate Aminotrasnferase (ASAT)
The present data revealed a nonsignificant change in total and fractional serum activities of ASAT in both LA and LAPQ as compared to the respective control values (Table 2). In opposition, PQ group exhibited a significant increase in total and fractional serum ASAT activities versus both C and LA groups. Meanwhile, a significant reduction has been demonstrated in LAPQ group versus PQ-treated group.
As shown in Fig. 4, two distinct phenotypes of serum ASAT have been detected in the gel with the same electrophorteic mobility in the control (C) group. The three treated groups (LA, PQ and LAPQ) exposed a pronounced change in the mobility of ASAT1, especially in PQ-treated group.
| Table 2: | Effect of acute toxicity of paraquat (1/4LD50) with or without α-lipoic acid supplementation on total and fractional isoenzyme activities of glucose-6-p-dehydrogenase (G6PDH) and aspartate aminotransferase (ASAT) in serum of adult female rat |
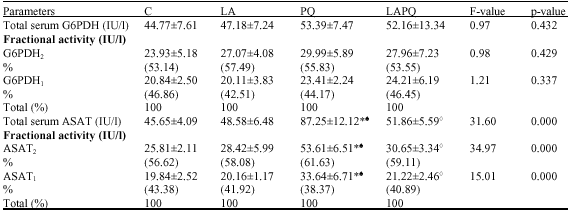 | |
| Data are presented as mean±SD. No. of samples per group = 5, * = Significant difference versus C group at p<0.05 (ANOVA, LSD), ♠= Significant difference versus LA at p<0.05 (ANOVA, LSD), ◊ = Significant difference versus PQ at p<0.05 (ANOVA, LSD) | |
 | |
| Fig. 3: | Isoenzyme pattern of serum glucose-6-p-dehydrogenase (G6PDH) in adult female rat as affected by acute paraquat toxicity and/or α-lipoic acid administration for 24 h. Individual lanes represent individual samples per group. C = Control group; LA = Lipoic acid group; PQ = Paraquat-treated group; LAPQ = Lipoic acid and paraquat-treated group |
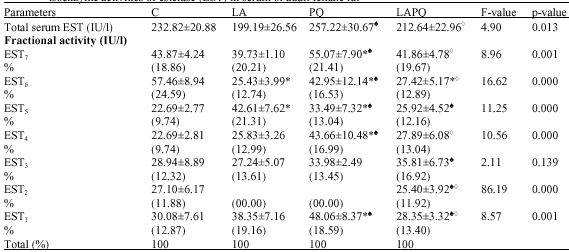
Effect on Total and Isoenzyme Activity of Serum Esterase (EST)
As shown in Table 3, all treated groups (LA, PQ and LAPQ) exhibited a nonsignificant change in total serum activity of EST as compared to the control (C) group. However, the statistical analysis revealed a significant rise in PQ group versus LA group. In contrast, a significant reduction has been demonstrated in LAPQ group versus PQ group.
As concerns serum EST isoenzyme pattern, the present data revealed seven distinct phenotypes in the gel with the same electrophoretic mobility in the control (C) group (Fig. 5). In LA and PQ groups, only six isoenzymes have been detected in the gel, EST2 has not been expressed at all. In addition, LA and LAPQ groups exhibited a pronounced change in the mobility of EST3.
The present data has shown a significant elevation in EST5 but a significant reduction in EST6 in LA group versus the respective control values (Table 3). On the other hand, PQ group exhibited a significant rise in the fractional activities of EST1, EST4 and EST7 as compared to both C and LA
| Table 3: | Effect of acute toxicity of paraquat (1/4LD50) with or without α-lipoic acid supplementation on total and fractional isoenzyme activities of esterase (EST) in serum of adult female rat |
 | |
| Data are presented as mean±SD. No. of samples per group = 5, * = Significant difference versus C group at p<0.05 (ANOVA, LSD), ♠= Significant difference versus LA at p<0.05 (ANOVA, LSD), ◊ = Significant difference versus PQ at p<0.05 (ANOVA, LSD) | |
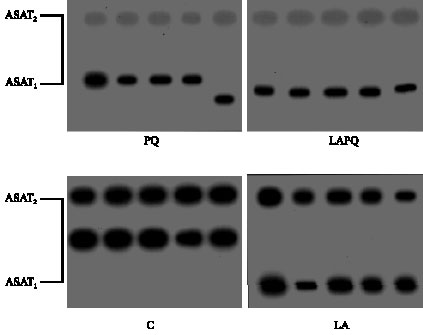 | |
| Fig. 4: | Isoenzyme pattern of serum aspartate transaminase (AST) in adult female rat as affected by acute paraquat toxicity and/or lipoic acid administration for 24 h. Individual lanes represent individual samples per group. C = Control group; LA = Lipoic acid group; PQ = Paraquat-treated group; LAPQ = Lipoic acid and paraquat-treated group |
values. Meanwhile, a significant rise in EST5 but a significant reduction in EST6 has been detected in PQ group versus the respective control values. For these two isoenzymes EST5, EST6, the picture was opposite in PQ group versus LA group. EST3 isoenzyme expression seemed to be resistant to acute PQ- toxicity (Table 3).
In LAPQ group, a significant reduction has been detected in EST6 versus the control value. In comparison with LA group, a significant reduction in EST1 and EST5 but a significant rise in EST3 has been demonstrated in LAPQ group. Meanwhile, EST1, EST4, EST6 and EST7 showed a significant reduction versus PQ-treated group.
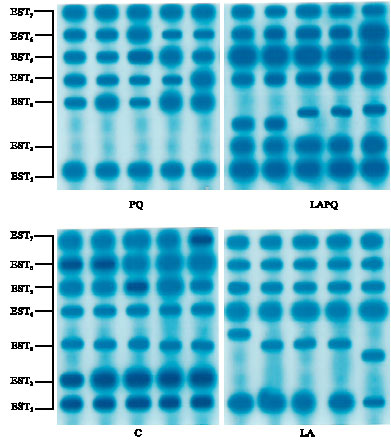 | |
| Fig. 5: | Isoenzyme pattern of serum esterase (EST) in adult female rat as affected by acute paraquat toxicity and/or lipoic acid administration for 24 h. Individual lanes represent individual samples per group. C = Control group; LA = Lipoic acid group; PQ = Paraquat-treated group; LAPQ = Lipoic acid and paraquat-treated group |
| Table 4: | Effect of acute toxicity of paraquat (1/4LD50) with or without α-lipoic acid supplementation on serum protein electrophoretic patterns in adult female rat |
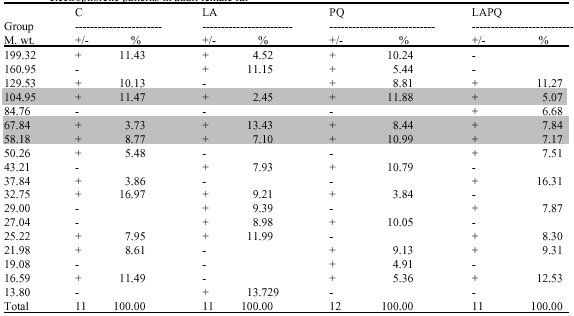 | |
| + = Expressed, - = Not expressed | |
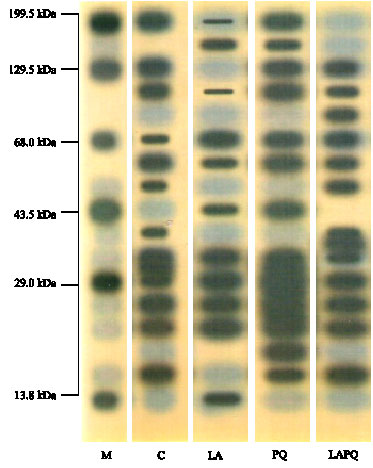 | |
| Fig. 6: | Serum protein electrophoretic patterns in female rat as affected by acute paraquat toxicity and/or lipoic acid administration for 24 h. Individual lanes represent a pool of five samples per group. M = Marker; C = Control group; LA = Lipoic acid group; PQ = Paraquat-treated group; LAPQ = Lipoic acid and paraquat-treated group |
Effect on Serum Protein Patterns
As seen in Table 4 and Fig. 6, the electrophoretic separation of serum protein showed a wide molecular weight range in the different groups with a maximum of 199.32 kDa and a minimum of 13.8 kDa. The banding pattern showed 12 distinct bands in PQ-intoxicated group but only 11 bands in other experimental groups. Only the 3 bands of 104.95, 67.84 and 58.18 kDa were expressed in all experimental groups (highlighted in Table 4). They seem resistant to the effect of PQ and/or lipoic acid. The 104.95 kDa band was similarly expressed in control (C) and PQ groups (around 11.5%) but less expressed in LA and LAPQ groups (2.45 and 5.07%, respectively). The band of 19.08 kDa was expressed in PQ-treated group only, while that of 13.8 kDa was expressed in LA group only and that of 84.76 kDa was expressed in LAPQ only. There were three missing bands in PQ-treated group versus the control (C) group. These were 50.26, 37.84 and 25.22 kDa. Five bands were missing in LA-treated group. These were 129.53, 84.76, 50.26, 37.84, 21.98 and 16.59 kDa. On the other hand, two bands were missing in LAPQ versus the control group. These were 199.32 and 32.75 kDa.
DISCUSSION
The oxidation of proteins plays an essential role in the pathogenesis of an important number of degenerative diseases and in aging, which is now widely recognized (Berlett and Stadtman, 1997; Stadtman and Berlett, 1998). Many different types of protein oxidative modification can be induced directly by Reactive Oxygen Species (ROS) or indirectly by reactions of secondary by-products of oxidative stress (Berlett and Stadtman, 1997). The toxic effects of oxidative stress on cells depend upon the intensity of the stress and/or the cell type. Low ROS concentrations have been shown to increase the antioxidant enzyme activity and to promote cell proliferation. Conversely, high concentrations of ROS induce apoptosis or necrosis (Dong-Yun et al., 2003; Kang and Zheng, 2004). The necrosis induced by severe oxidative stress is poorly understood at the molecular level while more is known about the role of antioxidants (Combs and Peterson, 1983; Cheng et al., 1999).
The present study aimed to investigate the effect of acute oxidative stress on the process of gene expression and protein activity in female rats as manifested by changes in serum isoenzyme expression and protein patterns induced by acute paraquat toxicity. Meanwhile, the protective role of α-lipoic acid administration has been challenged.
The present data revealed a significant rise in total serum activity of MDH, ACP and ASAT and a nonsignificant change in the total activity of G6PDH and EST in PQ group versus the control group. These findings might reflect the necrotic effect of acute oxidative stress induced by acute PQ toxicity on different body cells. α-LA administration exhibited a high protective effect against the changes in total serum activity of ACP and ASAT induced by PQ while a considerable protection in case of total serum MDH activity. The protective effect of α-LA can be explained by its greater ability to scavenge hydroxyl, singlet, peroxide and superoxide radicals as supported by the results of Packer et al. (1995). It can also be explained by the ability of lipoic acid to decrease lipid peroxidation and to regulate reduced glutathione and plasma total antioxidant power levels as cited by Guimarães et al. (2007).
In the present study, the fractional activities of serum isoenzymes exhibited the same trend as that demonstrated in serum total activity except in case of EST. Up to date, nothing has been recorded about the effect of acute oxidative stress on the hereby measured isoenzyme expression. The above findings are of special importance and could prove the possibility to use the isoenzyme expression as an additional marker to differentiate between the molecular effects of the oxidant and/or antioxidant in female animals. This conclusion can be supported by the recent study of El-Zayat and Amer (2003) and El-Zayat (2007) in male animals subjected to different types of oxidative stress.
Likewise, α-LA administration exhibited a high protective effect against the changes in serum isoenzyme activities of ACP, ASAT and EST while a considerable protection in case of MDH. However, LAPQ group exhibited a significant reduction in EST6 versus the control group. These findings provide additional evidence to the protective role of α-lipoic acid against the acute oxidative stress-induced changes in total activities of measured serum enzymes. In this context recent studies have proved that α-lipoate acts as a potent antioxidant by inhibiting lipid peroxidation and restoring reduced levels of other antioxidants in vivo (Arivazhagan et al., 2000, 2002, 2003; Moini et al., 2002; Sethumadhavan et al., 2005; Ganapathy et al., 2006). On the other hand, McCarty (2001) speculated that α-LA might exert its protective effects by catalyzing the formation of intramolecular disulfides in certain signaling proteins that function as detectors of oxidants and trigger heat-shock and phase II responses.
The mechanism by which α-LA revitalizes the antioxidant status can be explained by its potential to increase glucose uptake both in vitro and in vivo conditions (Packer et al., 1997). According to these authors, enhanced glucose uptake by cells serves as a fuel for both pentose phosphate pathway and oxidative phosphorylation thereby bringing up the cellular levels of NADPH/NADP+ which in turn enhances the activity of catalase. With increased levels of NADPH, glutathione reductase (GR) activity also improves, thereby raising the levels of glutathione (GSH), the substrate for glutathione peroxidase (GPx).
The present study has also revealed major changes in the electrophoretic mobility of some separated isoenzymes among the three treated groups (LA, PQ and LAPQ) especially in case of ACP2, ACP3, ASAT2 and EST3. These changes might reflect a considerable degree of oxidation to protein molecules, which might in turn results in tertiary structural alterations that promote protein aggregation and amyloid formation as supported by the study of Shinall et al. (2005). The mechanisms involved in the oxidation of proteins by ROS has also been described by the work of Stadtman (2004) in which amino acids, simple peptides and proteins were exposed to ionizing radiations under conditions where hydroxyl radicals or a mixture of hydroxyl/superoxide radicals are formed. The authors concluded that the side chains of all amino acid residues of proteins, in particular cysteine and methionine residues of proteins are susceptible to oxidation by the action of ROS/RNS. The oxidation of cysteine residues may lead to the reversible formation of mixed disulphides between protein thiol groups (-SH) and low molecular weight thiols, in particular GSH (S-glutathiolation).
Fractional activities of serum EST isoenzymes also exhibited some interesting trends under the present experimental conditions. A significant rise in EST1, EST4, EST5 and EST7 but a significant reduction in EST6 has been demonstrated in PQ-treated group despite the nonsignificant changes demonstrated in the same group in total serum activity as compared to the respective control values. The obtained data also revealed that EST2 is not expressed at all in both LA and PQ groups. This might be explained in the PQ group as oxidative damaging effect of PQ on DNA and/or protein. Moreover, EST3 expression seemed to be resistant to change under acute oxidative stress induced by PQ toxicity. On the other hand, a significant reduction has been detected in EST6 in LAPQ group versus the control value. This reduction might be attributed to the oxidant/antioxidant interaction. The process of gene expression is rather complicated and many factors might account for these changes.
The present data has also reflected a wide-range variation in serum protein patterns duo to PQ and/or α-lipoic acid treatment, which indirectly reflect a parallel variation the process of gene expression and/or proteolytic activity. One of the most important mechanisms underlying the effect of oxidative stress and free radicals are their effect on DNA. Oxygen free radicals have shown to induce DNA breaks (Balin and Allen, 1986; Bedwell et al., 1989; Nakahara et al., 1998). The appearance and/or disappearance of protein fractions in the present study may be attributed to this damage or may be explained by the report of Bedwell et al. (1989) who claimed that the free radicals promote sulfhydral-mediated cross-linking of the labile amino acids such as methionine, histidine, cysteine and lysine causing a fragmentation of polypeptide chains in the protein molecule.
It is worth to mention here that variations encountered in isoenzyme expression, mobility or protein patterns in LA group can be explained by a relative prooxidant effect of α-lipoic acid as recently suggested by Cakatay et al. (2005).
As a conclusion, the present investigation supports the use of isoenzyme expression and activity, rather than total enzyme activity, for better understanding of the molecular basis of oxidative stress in female animals as has been previously documented in male animals subjected to different types of oxidative stress (El-Zayat and Amer, 2002; El-Zayat, 2007).
REFERENCES
- Arivazhagan, P., S. Shila, S. Kumaran and C. Panneerselvam, 2002. Effect of DL-α-lipoic acid on the status of lipid peroxidation and antioxidant enzymes in various brain regions of aged rats. Exp. Gerontology, 37: 803-811.
CrossRefDirect Link - Arivazhagan, P., S.R. Panneerselvam and C. Panneerselvam, 2003. Effect of DL-α-lipoic acid on the status of lipid peroxidation and lipids in aged rats. J. Gerontol.: Ser. A, 58: B788-B791.
CrossRefPubMedDirect Link - Balin, A.K. and R.G. Allen, 1986. Mechanisms of biologic aging. Dermatol. Clin., 4: 347-358.
CrossRefDirect Link - Beck, D. and D. Feir, 1989. The isolation of acid phosphatase isoenzymes of Oncopeltus fasciatus, (Dallas) (Hemiptera: Lygaeidae). Comput. Biochem. Physiol. Part B: Comp. Biochem., 93: 799-802.
CrossRef - Bedwell, S., R.T. Dean and W. Jessup, 1989. The action of defined oxygen-centered free radicals on human low-density lipoprotein. Biochem. J., 262: 707-712.
PubMedDirect Link - Bergamini, C.M., S. Gambetti, A. Dondi and C. Cervellati, 2004. Oxygen, reactive oxygen species and tissue damage. Curr. Pharm. Des., 10: 1611-1626.
PubMedDirect Link - Berlett, B.S. and E.R. Stadtman, 1997. Protein oxidation in aging, disease and oxidative stress. J. Biol. Chem., 272: 20313-20316.
Direct Link - Biewenga, G.P., G.R.M.M. Haenen and A. Bast, 1997. The pharmacology of the antioxidant lipoic acid. Gen. Pharmacol.: Vasc. Syst., 29: 315-331.
CrossRefDirect Link - Bilska, A. and L. Wlodek, 2005. Lipoic acid-the drug of the future?. Pharmacol. Rep., 57: 570-577.
PubMed - Cadenas, E., 1989. Biochemistry of oxygen toxicity. Annu. Rev. Biochem., 58: 79-110.
CrossRefPubMedDirect Link - Cakatay, U., R. Kayali, A. Sivas and F. Tekeli, 2005. Prooxidant activities of α-lipoic acid on oxidative protein damage in the aging rat heart muscle. Arch. Gerontol. Geriatrics, 40: 231-240.
CrossRefPubMedDirect Link - Cheng, W.H., B.A. Valentine and X.G. Lei, 1999. High levels of dietary vitamin E do not replace cellular glutathione peroxidase in protecting mice from acute oxidative stress. J. Nutr., 129: 1951-1957.
PubMed - Combs, Jr., G.F. and F.J. Peterson, 1983. Protection against acute paraquat toxicity by dietary selenium in the chick. J. Nutr., 113: 538-545.
PubMedDirect Link - Dinis-Oliveira, R.J., F. Remiao, J.A. Duarte, A.S. Navarro, M.L. Bastos and F. Carvalho, 2006. Paraquat exposure as an etiological factor of Parkinson's disease. Neurotoxicol., 27: 1110-1122.
CrossRef - Dinis-Oliveira, R.J., C. Sousa, F. Remiao, J.A. Duarte, A.S. Navarro, M.L. Bastos and F. Carvalho, 2007. Full survival of paraquat-exposed rats after treatment with sodium salicylate. Free Radic. Biol. Med., 42: 1017-1028.
CrossRefPubMedDirect Link - Shi, D.Y., Y.R. Deng, S.L. Liu, Y.D. Zhang and L. Wei, 2003. Redox stress regulates cell proliferation and apoptosis of human hepatoma through Akt protein phosphorylation. FEBS. Lett., 542: 60-64.
CrossRef - El-Zayat, E.M. and S.A. Amer, 2003. Protective effects of antioxidants on age-related changes in the electrophoretic patterns of cardiac LDH, hepatic ALP and serum proteins in male golden hamster. Cell Biochem. Funct., 21: 137-145.
CrossRef - Amudha, G., A. Josephine and P. Varalakshmi, 2006. Role of lipoic acid in reducing the oxidative stress induced by cyclosporine A. Clin. Chem. Acta, 372: 134-139.
CrossRef - Guerin, J.P. and P. Kerambrun, 1982. Effects of diet on esterases, alkaline phosphatase, malate dehydrogenase and phosphoglucomutase activity observed by polyacrylamide gel electrophoresis in Tisbe holothuriae (harpacticoid copepod). Comput. Biochem. Physiol. Part B: Biochem. Mol. Biol., 73: 761-770.
CrossRef - Hermann, R., G. Niebch, H.O. Borbe, H. Fieger-Buschges and P. Ruus et al., 1996. Enantioselective pharmacokinetics and bioavailability of different racemic-lipoic acid formulations in healthy volunteers. Eur. J. Pharm. Sci., 4: 167-174.
CrossRef - Hwang, E.S. and G.H. Kim, 2007. Biomarkers for oxidative stress status of DNA, lipids and proteins in vitro and in vivo cancer research. Toxicology, 229: 1-10.
CrossRef - Jensen, S.J.K., 2003. Oxidative stress and free radicals J. Mol. Struct. Theochem., 666-667: 387-392.
CrossRefDirect Link - Jung, I.L. and I.G. Kim, 2003. Thiamine protects against paraquat-induced damage: Scavenging activity of reactive oxygen species environment. Environ. Toxicol. Pharmacol., 15: 19-26.
CrossRef - Kang, J. and R. Zheng, 2004. Dose-dependent regulation of superoxide anion on the proliferation, differentiation, apoptosis and necrosis of human hepatoma cells: The role of intracellular Ca2+. Redox Rep., 9: 37-48.
PubMed - Laemmli, U.K., 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227: 680-685.
CrossRefDirect Link - Margolis, A.M., S. Porasuphatana and G.M. Rosen, 2000. Role of paraquat in the uncoupling of nitric oxide synthase. Biochim. Biophys. Acta, 1524: 253-257.
CrossRef - Marnett, L.J., 2000. Oxyradicals and DNA damage. Carcinogenesis, 21: 361-370.
CrossRefPubMedDirect Link - McCarty, M.F., 2001. Versatile cytoprotective activity of lipoic acid may reflect its ability to activate signaling intermediates that trigger the heat-shock and phase II responses. Med. Hypotheses, 57: 313-317.
CrossRefPubMedDirect Link - Moini, H., L. Packer and N.E.L. Saris, 2002. Antioxidant and prooxidant activities of α-lipoic acid and dihydrolipoic acid. Toxicol. Applied Pharmacol., 182: 84-90.
CrossRefDirect Link - Mollace, V., M. Iannone, C. Muscoli, E. Palma and T. Granato et al., 2003. The role of oxidative stress in paraquat-induced neurotoxicity in rats: Protection by non-peptidyl superoxide dismutase mimetic. Neurosci. Lett., 335: 163-166.
CrossRef - Nakahara, M., T. Kanoo, Y. Inai, K. Utsumi, M. Hiramatsu, A. Mori and L. Packer, 1998. Mitochondrial dysfunction in the senescence accelerated mouse (SAM). Free Radical Biol. Med., 24: 85-92.
CrossRef - Packer, L., E.H. Witt and H.J. Tritschler, 1995. Alpha-lipoic acid as a biological antioxidant. Free Radic. Biol. Med., 19: 227-250.
CrossRefPubMedDirect Link - Packer, L., H.J. Tritschler and K. Wessel, 1997. Neuroprotection by the metabolic antioxidant α-lipoic acid. Free Radic. Biol. Med., 22: 359-378.
CrossRefPubMedDirect Link - Savitha, S., J.Tamilselvan, M. Anusuyadevi and C. Panneerselvam, 2005. Oxidative stress on mitochondrial antioxidant defense system in the aging process: Role of DL- -lipoic acid and L-carnitine. Clin. Chem. Acta, 355: 173-180.
PubMed - Shinall, H., E.S. Song and L.B. Hersh, 2005. Susceptibility of amyloid beta peptide degrading enzymes to oxidative damage: A potential alzheimer's disease spiral. Biochemistry, 44: 15345-15350.
CrossRefPubMedDirect Link - Siems, W.G., T. Grune and H. Esterbauer, 1995. 4-Hydroxynonenal formation during ischemia and reperfusion of rat small-intestine. Life Sci., 57: 785-789.
CrossRef - Stadtman, E.R. and B.S. Berlett, 1997. Reactive oxygen-mediated protein oxidation in aging and disease. Chem. Res. Toxicol., 10: 485-494.
PubMedDirect Link - Toncheva, D., T. Evrev and M. Tzoneva, 1982. G6PD in immature and mature human brain. Electrophoretic and enzyme kinetic studies. Hum. Hered., 32: 193-196.
PubMedDirect Link - Valko, M., H. Morris and M.T.D. Cronin, 2005. Metals, toxicity and oxidative stress. Curr. Med. Chem., 12: 1161-1208.
CrossRefPubMedDirect Link - Valko, M., C.J. Rhodes, J. Moncol, M. Izakovic and M. Mazur, 2006. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem. Biol. Interact., 160: 1-40.
CrossRefPubMedDirect Link - Valko, M., D. Leibfritz, J. Moncol, M.T.D. Cronin, M. Mazur and J. Telser, 2007. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol., 39: 44-84.
CrossRefPubMedDirect Link - Van Der Laarse, A., L. Hollaar and C.H. Bloys Van Treslong, 1980. Evaluation and comparison of a column-chromatographic and an electrophoretic technique for separation of the two isoenzymes of aspartate aminotransferase (ast). Clin. Chim. Acta, 28: 257-260.
PubMed








