ABSTRACT
The concentration of the fungicide Zineb required to inhibit 50 and 90% of mycelial growth for the melanotic fungi was relatively high. Sclerotium cepivorum appeared to be more sensitive to carboximide than Fusarium oxysporum. Alternaria solani was comparatively the least sensitive. The inhibition of fungal growth by Zineb was reversed by α-tocopherol and the effect appeared to be dose dependent. Fifty percent inhibition of growth led to an accumulation in their lipid peroxide contents in 3 tested fungi as compared to that in the absence of fungicide. This increase in lipid peroxide was reversed by addition of α-tocopherol to Zineb growth culture. The former being a lipophilic antioxidant could eliminate the oxidant free radicals initiated by Zineb. Resistance factors of the 3 melanotic increased by inclusion of dopamine, melanin precursor, in Zineb growth culture. On the other hand, the melanin inhibitors Na2 EDTA really increased the inhibitory effect of fungicides in the three tested pathogens. A clear correlation exists between the presence of some melanin inhibitors in Zineb growth cultures and the retardation in fungicide sensitivity values. The resistance factor as dropped by about 25 to 40%. The evidence for a relationship between melanization and resistance to oxidant thus seems very strong, leading the elimination of inhibitory effect of the fungicide. To improve the effectiveness of carboximide fungicides, in general, it must be associated with selective melanin inhibitors particularly in controlling melanotic pathogens either in plant pathology or in medicinal field, under relatively low oxidative stress.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ppj.2002.32.35
URL: https://scialert.net/abstract/?doi=ppj.2002.32.35
INTRODUCTION
Fungicides having dicarboximide group are effective against a number of fungi pathogenic to soft fruits, ornamentals, vegetables and vines (Christopher and Nair, 1993). It has been used effectively and safety on crops for 25 years.
The precise mode of action of dicarboximide fungicides reflects no inhibitory effect on cell organelles or cell function, at concentration capable of inhibiting growth (Pappas and Fisher, 1979), but improved the crop yields (Raj Kumar et al., 1989).
It is only very slightly soluble in water and is applied as a suspension of solid particles, these particles reside on crop or soil surfaces with little or no penetration. They are degraded by weathering processes especially sunlight. It is fortunate that its degradates are highly susceptible to photodegradation and to metabolism by plants, animals and soil microorganisms. For this reason, it is rarely found in detectable amounts (<0.01 ppm) in any food or feed. (Rohm and Haas Research Laboratories in Spring House, Pennsylvania, USA, 1999).
Resistance against dicarboximides has since been recorded in a number of fungal pathogens, particularly the melanotic pathogens as Botrytis cinerea (Christopher and Nair, 1993) Alternaria tenius (Razak et al., 1991), Sclerotium rolfsii (Ohazurike, 1996), Alternaria solani (Maheshwari and Singh, 1998), Fusarium spp. (Gaurilcikiene, 1997) and A. solani, F. oxysporum and S. cepivorum (Abo Ellil and Sharaf, 2000).
Despite the isolation of numerous resistant strains during the past few years, information on their mode of resistance to dicarboximide fungicides is scanty. Carboximides produce antimicrobial oxidants by introducing oxygen free radicals, however many microorganisms have evolved defenses against oxidants (Christman et al., 1985; Hassan and Fridovich, 1979).
Melanin may have an important role in resistance of the melanotic fungi to carboximides, the protective role of melanin against free radicals indicates that it is able to scavenge such toxic free radicals (Jacobson and Emery, 1991).
Efficient inhibitors of melanin synthesis are nowadays needed in the field of phytopathology (Abo Ellil, 1999). Such inhibitors are necessary to enhance drug efficiency of carboximide in the presence of suppressed melanin production. This is highly desirable for fungicides used against soil borne pathogens that survive as melanized propagules.
Few studies have been undertaken on the role of melanin and resistance to carboximide fungicides (Abo Ellil and Sharaf, 2000). In this study, melanization of the tested pathogens was suppressed in a trial to pave the way in front of dicarboximide to show its antifungal effect. Success of this aim will be a solid proof revealing the role of melanin as an antioxidant.
Materials and Methods
Isolates and culture condition: Three phytopathogens were used in this study, being Alternaria solani, Fusarium oxysporum f.sp. Lycopercici and Sclerotium cepivorum. These isolates have the capability to produce melanin and melanin like pigments.
They have been previously isolated and identified (Abo Ellil and Sharaf, 2000), from the target hosts and maintained on Czapek’s Dox and potato dextrose agar media. For use, the cultures were transferred to potato dextrose agar (PDA) plates and grown at 25°C. Sensitivity to Zineb (a dicarboximide fungicide) was determined on a series of PDA plates supplemented with fungicide by using an agar disc (4 mm) from an axenic culture as an inoculum. Radial growth was measured when the fungus had colonized 50-75% of the control (fungicide-free) plates by measuring from the edge of the inoculum to growing edge of culture.
The mycelium used for determination of the effects of ∝-tocopherol on growth, lipid-peroxide content and activities of melanin precursors and inhibitors was produced on Czapek-Dox liquid medium with agitation at 25°C.
Fungicides: The fungicides used were Zineb [zinc ethylene bis (dithiocarbamate)] and benelate [methyl 1-(butyl-carbamoyl) benzimidazol-2-carbamate] (Ciba Geigy). The concentrations of the fungicides used were 10 ppm up to 3500 ppm.
Effect of ∝-tocopherol on Zineb treated cultures: ∝-tocopherol was added to flasks containing Czapek-Dox liquid medium (50 ml), in a 250 μl volume acetone to give was the concentration used, 0.25, 0.50 and 1.0 mg ml-1. Fungicide was diluted to give a range of concentration from 100 to 1500 ppm, when added to the medium. A second series of flasks (control) with the same fungicide dilutions contained 250 μl acetone free from α-tocopherol. The final concentration was 1% v/v. the flasks were inoculated by addition of 1 ml of an homogenized culture in the logarithmic phase of growth. This inoculum corresponded to 0.005 g dry wt of mycelium. Cultures were grown at 25oC with agitation for 96 h, harvested by centrifugation, washed with 3 volumes of phosphate buffer and the dry weight was determined by transferring the fungal pellets to preweighed filter paper, the concentration of fungicide required to inhibit growth by 50% (I50) was calculated by linear regression analysis.
Measurement of lipid peroxide
| a) | Extraction of total lipids: The samples were extracted on the day of harvest to minimize lipid peroxidation due to sample storage. Extraction of total lipids was done according to Folch et al. (1957). |
| b) | Total lipid peroxidation and total lipid estimation: Estimation of lipid peroxidation products was carried out with thiobarbituric acid (Quinian et al., 1988). All values (means of 3 determinations) of extracted total lipids from samples were referred to total phospholipids. The total phospholipids were indirectly estimated from the phosphorus in dry lipid residue. Phosphorus was measured as recommended by Bartlett (1959). |
Effect of melanin precursors and inhibitors on melanization and growth: The isolation of melanin from the mycelial mats grown on Czapek-Dox liquid medium with or without various amounts of melanin precursors and melanin inhibitors was carried out according to the method of Ellis and Griffith (1974). Dopamine hydrochloride (DOPA) (Jacobson and Tinnell, 1993) was used as melanin precursor. Na2 EDTA (disodium ethylene diamine tetracetic acid) (Chet and Henis, 1969), tricyclazole [5 methyl. 1,2,4–triazole (3,4,6,)–benzothiozole]. (Woloshuk et al., 1980) and chloben thiazone. (4 chloro–3–methyl benzothiazol–2(3 h)–one) (Inoue and Krato, 1983) were used as melanin inhibitors. Melanization was monitored at 400 nm as the incremental optical density (O.D) with reference to comparable culture without melanin precursors or inhibitors. Cultures with melanization beyond an O.D of 1 were diluted 10 folds and measured against a similarly dilute reference culture (Nicolaus et al., 1964).
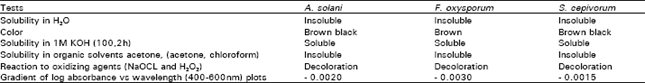
Isolation and characterization of melanin: Twenty air dried mycelial mats were used for melanin extraction. Because of rapid oxidation of pigment in air, extraction and analysis were carried out under nitrogen. Weights were determined whether the substance obtained was melanin and melanin-like pigments or not, the following points were examined according to Ellis and Griffiths (1974); (1) solubility in water, (2) colour, (3) solubility in 1M KOH (100°C, 2 h), (4) precipitation in HCL, (5) solubility in organic solvent, (6) reaction with oxidant (NaOCl and H2O2), (7) gradient of log absorbance in visible light (400-600 nm) and (8) IR- spectrum.
Results
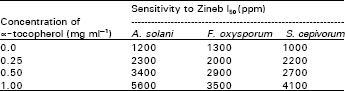
Sensitivity to fungicides: The I50 and I90 (the concentrations of the fungicide required to cause 50 and 90% inhibition of mycelial growth, respectively) for the three studied fungi are shown in Table 1. Sclerotuim cepivorum appeared to be more sensitive to Zineb than Fusarium oxysporum, Alternaria solani was comparatively the least sensitive. This inhibition of fungal growth by Zineb was reversed by ∝!tocopherol and the effect appeared to be dose dependent (Table 2). However, the growth inhibition of benelate on the 3 tested fungi was not affected inclusion of ∝!tocopherol with the fungicide (Table 3).
| Table 1: | Sensitivity of Sclerotium cepivorum, Fusarium oxysporum and Alternaria solani to Zineb |
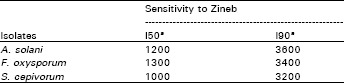 | |
| a= Concentration of Zineb (ppm) required to inhibit mycelial growth by 50 and 90% | |
| Table 2: | Effect of ∝!tocopherol on the sensitivity of the 3 studied fungi to Zineb |
 | |
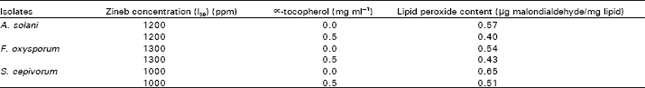
Lipid peroxide content: The lipid peroxide content of three tested fungi grown in presence and absence of sufficient Zineb to cause approximately 50% inhibition of growth (Table 4). On the basis of malondialdehyde content, culturing of 3 fungi in presence of Zineb led to an increase in their lipid peroxide contents as compared to that in the absence of the fungicide. This increase in lipid peroxide was also reversed by addition of ∝!tocopherol to culture medium (Table 5).
Characteristics of melanin: Melanin were all insoluble in water, acetone and chloroform but soluble in IM KOH at 100°C (Table 6). Their colour ranged from brown to black. Furthermore the patterns of IR-absorption spectra of extracted melanins were almost the same as the patterns of fungal melanins previously reported (Potgieter and Alexander, 1966; Chet and Henis, 1969; Ellis and Griffiths, 1974).
Melanization and resistance to Zineb: In comparison to cultures grown without dopamine (a melanin precursor) (Table 7), resistance to Zineb was demonstrable and peaked at 1.5-1.9 fold, matching closely with melanization. Such treated fungi produce extra melanin and acquire resistance from prolonged growth in dopamine medium.
| Table 3: | Comparative effect of ∝-tocopherol on the sensitivity of the 3 tested fungi to Benelate and Zineb |
 | |
| Table 4: | Lipid peroxide contents of the 3 tested fungi grown in presence and absence of Zineb |
 | |
| Table 5: | Effect of ∝-tocopherol on lipid peroxide content of the 3 tested fungi grown in the presence of Zineb at I50 |
 | |
| Table 6: | Diagnostic tests for melanin of the 3 Isolates under test |
 | |
| Table 7: | Melanization and resistance to Zineb in the 3 Isolates grown in the presence and absence of dopamine, as melanin precursor |
 | |
| Resistance factor = b/a, O.D. = Optical density | |
| Table 8: | Effect of the melanin inhibitor (Na2 EDTA) on melanization and resistance to Zineb by the 3 isolates under test |
 | |
| O.D. = Optical Density | |
| Table 9: | Effect of some other melanin inhibitors on the resistance to Zineb by the 3 isolates |
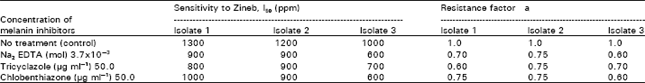 | |
| P<0.01, Isolate 1= A. solani, Isolate 2= F. oxysporum, Isolate 3= S. cepivorum | |
On the other hand resistance factors were decreased by 0.6–0.75 fold, directly proportional to the decrease of melanization in Na2EDTA-Zineb growth cultures of the 3 organisms (Table 8).
Melanin inhibitors: A clear correlation exists between the presence of some melanin inhibitors (Na2EDTA, tricyclazole and chlobenthiazone) in Zineb growth cultures and the retardation in fungicide sensitivity values. The resistance factors were dropped by about 25 to 40% (Table 9).
DISCUSSION
In this experiment the concentration of Zineb fungicide required to cause 50 and 90% inhibition of mycelial growth was relatively high, ranging from 1300 to 3600 ppm in cultures of three melanotic pathogenic isolates. Being classified as dicarboximide fungicide, Zineb may interface with cytochrome C reductase and a number of other flavin enzymes leading to accumulation of peroxy radicals and the superoxide anion. These reactive radicals then cause the formation of lipid peroxides within the cell membrane which shall be the ultimate cause of cell death in dicarboximide-treated fungi (Christopher and Nair, 1993). These free reactive radicals are removed by the activity of non-enzymatic antioxidants. These results revealed that tocopherol, acting as a free radical scavenger, alleviated the inhibitory effect of Zineb. This alleviation was more established by elevated concentration of ∝-tocopherol. This action supports the hypothesis that the net fungitoxic effect of Zineb is free radical formation. A similar effect was also seen with some related dicarboximide fungicides (Christopher and Nair, 1993).
In contrast, the toxicity of benelate was not affected by the addition of this antioxidant in the medium of the tested pathogens. Benelate is a benzimidazole fungicide which binds to the B-subunit of the tubulin protein, involved in cell division (Davidse, 1986), lipid peroxidation is a complex process involving the formation and propagation of lipid radicals leading to the eventual destruction of membrane lipids. It has been recorded that dicarboximide fungicides induce lipid peroxidation by means of oxygen activation in fungi (Radice et al., 1998). Growth in the presence of sufficient Zineb to cause 50% inhibition of growth led to an accumulation of lipid peroxides which were about 30% excess than the corresponding value in absence of the fungicide. Such higher content of lipid peroxides in Zineb-grown cultures, when assessed on the basis of malondialdehyde level, ensures and puts a further evidence that Zineb toxicity to fungi is through the formation of free oxidant radicals. A further evidence for this mode of action arises when ∝-tocopherol was included in Zineb growth cultures, the former being a lipophilic antioxidant (Beijersbergen Van Henegouwen, 1998) could eliminate the oxidant free radicals initiated by Zineb.
In this study, increased melanization caused by inclusion of dopamine, melanin precursor, in Zineb-growth cultures led to a decrease in the sensitivity of the melonatic pathogens towards the fungicide. An important use of melanin may be its ability to concentrate and immobilize a quantity of reducing equivalents just outside, the surface of the cell, while the oxygen free radical species appear to be harmlessly absorbed by melanin (Kwon-Chunge et al., 1982). Moreover electrically charged oxidants are excluded from the cytoplasm and react primarily with melanin (Korytowski and Sarna, 1990). Indeed melanin has been shown to neutralize potentially microbicidal strong oxidants similar to these produced by leukocystes in response to infection (Jocobson and Tinnell, 1993; Wang and Casadevall, 1994).
On the other hand, the present results showed that the melanin inhibitor, Na2 EDTA, really inhibited melanin formation by the three tested pathogens. Thus the fungal cell lost partly the capability of melanin to scavenge the free radicals generated by carboximide and it is likely that the free radicals freely cross the cell membrane to attack intercellular targets. The evidence for a relationship between melanization and resistance to oxidant thus seems very strong.
From the results, the antioxidant when associated with the carboximide in growth media of the tested fungi, may play its role on two axes. The first is the capability of the antioxidant to scavenge the toxic free radicals, generated by Zineb in aqueous medium, thus leading the elimination of the inhibitory effect of the fungicide. The second is the capacity of antioxidant to lower the oxygen tension in melanotic fungi. The generated antioxidants radicals are probably sufficient to bring the melanin from the completely oxidized state to partly reduced state, thereby regenerating the redox buffer. However melanin is a strong redox buffer, so it becomes greedy to get oxygen species radicals to maintain its stability by oxidation.
In conclusion, to improve the effectiveness of carboximide fungicides, in general, it must be associated with selected melanin inhibitors particularly in controlling melanotic pathogens either in plant pathology or in medicinal field, under relatively low oxidative stress.
REFERENCES
- Ellil, A.H.A., 1999. Oxidative stress in relation to lipid peroxidation sclerotial development and melanin production by Sclerotium rolfsii. Phytopathology, 147: 561-566.
Direct Link - Davidse, L.C., 1986. Benzimidazole fungicides mechanism of action and biological impact. Ann. Rev. Phytopathol., 24: 43-65.
CrossRefDirect Link - Ellis, D.H. and D.A. Griffiths, 1974. The location and analysis of melanins in the cell walls of some soil fungi. Can. J. Microbiol., 20: 1379-1386.
CrossRefDirect Link - Folch, J., M. Lees and G.H.S. Stanley, 1957. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem., 226: 497-509.
CrossRefPubMedDirect Link - Hassan, H.M. and I. Fridovich, 1979. Superoxide, hydrogen peroxide and oxygen tolerance of oxygen sensitive mutants of Escherichia coli. Rev. Infect. Dis., 1: 357-369.
PubMedDirect Link - Inoue, S. and T. Kato, 1983. Mode of rice blast control by chlobenthiazone. J. Pesticide Sci., 8: 333-338.
Direct Link - Jacobson, E.S. and H.S. Emery, 1991. Catecholamine uptake, melanization and oxygen toxicity in Cryptococcus neoformans. J. Bacteriol., 173: 401-403.
PubMedDirect Link - Jacobson, E.S. and S.B. Tinnell, 1993. Antioxidative function of fungal melanin. J. Bacteriol., 175: 7102-7104.
Direct Link - Korytowski, W. and T. Sarna, 1990. Bleaching of melanin pigments: Role of copper ions and hydrogen peroxide in autooxidation and photooxidation of synthetic DOPA-melanin. J. Biol. Chem., 265: 12410-12416.
Direct Link - Kwon-Chung, K.J., I. Polacheck and T.J. Popkin, 1982. Melanin-lacking mutants of Cryptococcus neoformans and their virulence for mice. J. Bacteriol., 150: 1414-1421.
PubMedDirect Link - Nicolaus, R.A., M. Piattelli and E. Fattorusso, 1964. On some natural melanins. Tetrahedron, 20: 1163-1172.
PubMedDirect Link - Ohazurike, N.C., 1996. Effect of some fungicides on extracellular enzymes of Sclerotium rolfsii sacc. Nahrung, 40: 150-153.
Direct Link - Potgieter, H.J. and M. Alexander, 1966. Susceptibility and resistance of several fungi to microbial lysis. J. Bacteriol., 91: 1526-1532.
PubMedDirect Link - Quinian, J.G., B. Halliwell, C.P. Moorhouse and C.M.J. Cutteridge, 1988. Action of lead (II) and aluminum (III) ions in iron-stimulated lipid peroxidation in liposomes, erythrocytes and rat liver microsomal fractions. Biochim. Biophys. Acta, 962: 196-200.
PubMedDirect Link - Razak, A.A., H. El-Tantawy, H.H. El-Sheikh and M.M. Gharieb, 1991. Influence of selenium on the efficiency of fungicide action against certain fungi. Biol. Trace Element Res., 28: 47-56.
CrossRef - Wang, Y. and A. Casadevall, 1994. Susceptibility of melanized and nonmelanized Cryptococcus neoformans to nitrogen and oxygen-derived oxidants. Infect. Immunity, 62: 3004-3007.
PubMedDirect Link








