ABSTRACT
Various types of microorganisms were isolated from fermented products of locust bean seeds (Parkia biglobosa), castor bean seeds (Ricinus communis), African oil bean seeds (Pentaclethra macrophylla) and mesquite seeds (Prosopis africana) and characterized. The fermented products, namely, iru, ogiri, ugba and okpei, respectively, are mainly used as condiments in soups, sauces and porridges among consuming populations in Nigeria. The results show that only bacteria were isolated from the fermented condiments. The organisms isolated included species of Micrococcus, Lactobacillus, Staphylococcus and Bacillus. From the results of morphological and biochemical tests carried out on the isolated species, Bacillus subtilis, Bacillus cereus, Lactobacillus brevis, Lactobacillus fermenti, Staphylococcus aureus, Staphylococcus saprophyticus, Micrococcus roseus and Micrococcus varians were found to be present. The isolates were used to ferment freshly prepared oilseed samples, with subsequent evaluation of the desirable quality characteristics of texture, color and aroma. B. subtilis was found to give the products with acceptable quality attributes.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjn.2009.279.283
URL: https://scialert.net/abstract/?doi=pjn.2009.279.283
INTRODUCTION
The prevailing population pressure in Nigeria, as in other less-developed countries, has resulted in an increasing demand for wild under-exploited nutritious plant products with aesthetic and organoleptic appeal in the daily diet (Enujiugha, 2005). The common edible portions of most under-utilized plants are the seeds, which in some cases are cooked or roasted and eaten directly as snack foods e.g. conophor nut and bambara groundnut, while some are cooked and fermented for use as soup and sauce ingredients e.g. African oil bean, locust bean, castor bean and melon. There are various plant seeds that are fermented and used as food in some rural parts of Nigeria, among which are, >iru’ from African locust bean (Parkia biglobosa), >ogiri’ from castor bean (Ricinus communis), >okpei’ from mesquite seed (Prosopis africana) and >ugba’ from African oil bean (Pentaclethra macrophylla). Diverse groups of bacteria comprising species of Bacillus, Micrococcus, Leuconostic, Staphylococcus and Enterobacteriaceae have been reported by various authors (Enujiugha and Badejo, 2002; Anosike and Egwuatu, 1981; Obeta and Ugwuanyi, 1996; Odunfa, 1981) as contributing to the individual fermentations. However, the indigenous fermentations are mostly achieved via natural inoculations. The need to standardize the processing techniques and to obtain hygienic and safe products, has led to the search for microbial starters for these local fermentations. The objective of the present study was to characterize the fermentative organisms with a view to identifying the major starters.
Materials and Methods
Materials: Traditionally fermented products of African oil bean (ugba), locust bean (iru), mesquite seed (okpei ) and castor bean (ogiri ) were bought from the King’s market at Akure, Nigeria. Locust bean seed, African oil bean seed, mesquite seed and castor bean seed were obtained from local farmers at Owena in Ondo State, Nigeria. Aluminum foil used for the work was bought from the King’s market. The syringes and cotton wool were purchased from Matador Pharmaceutical in Akure. Ethanol and simple sugars used in the study were bought from Wintech chemicals at Akure. All the chemicals and reagents used in the study were of analytical grade.
Isolation of microorganisms: Fermented samples (ugba, ogiri, iru, okpei) were taken aseptically from traditionally fermented beans. One gram of the sample was thoroughly mashed with laboratory pestle and mortar and mixed with 9 ml of normal saline water as a diluent in a McCartney bottle and the content was thoroughly shaken. Subsequent serial dilutions (10-2, 10-3, 10-4, 10-5, 10-6) were made from this solution by adding serially 1ml of solution from preceding concentration to 9ml of the diluent, using sterile syringe. The nutrient agar was prepared by adding 11ml of distilled water to 2.8 g of agar in a conical flask. Unto a petri-dish containing 0.1 ml of the inoculum, 10 to 15 ml of the sterile warm (45oC) nutrient agar was poured. The plates were labeled appropriately, inverted and incubated at 30oC for 24 hrs in a Gallenkamp incubator.
Characterization and identification of isolates: Colonies obtained after incubation were sub-cultured on nutrient agar which was incubated for 24 hours at 30oC. The cultural characteristics of isolates on the agar plates were observed. The motility of the isolates was examined using hanging drop technique. Gram staining reactions and cell morphology from heat fixed smears were done. The identification procedures for the micro-organisms were carried out using Cowan and Steel (1966) methods. Pure cultures of the different organisms isolated were sub-cultured and preserved on agar slants at refrigeration temperature (4oC).
Laboratory fermentation of ugba, okpei, ogiri and iru with identified isolates: The fermentation was carried out as shown on Fig. 1. The seeds of Parkia biglobosa, Prosopis africana and Pentaclethra macrophylla were boiled to soften the hulls for easy removal and separation of the cotyledons. Ricinus communis seeds were mechanically dehulled without parboiling. African oil bean seeds were sliced using a kitchen knife into a thickness of about 0.2-0.4cm. The dehulled and sliced beans where washed several times with clean water and boiled for 2-3 hours. The boiling serves to further soften the seeds and remove some of the anti- nutritional substances. The dehulled and boiled seeds were washed again with clean water and then soaked for 12 hours in tap water. After the soaking period, the seeds were drained and washed again until they were thoroughly cleaned. Mashing was done for castor bean seed only. The seeds were packed in washed banana leaves, about 30g in each wrap. The wraps were packed inside a container and sterilized in the autoclave at 121oC for 15 mins. 10 ml of Nutrient both were poured into test tubes and sterilized at 121oC for 15 mins. They were allowed to cool down to 25oC, a loopful of the organisms from the agar slants where transferred into the broth and incubated at 37oC for 24 hrs. The cell suspensions obtained were shaken together and 1ml was taken using a sterile syringe for the different organisms obtained from the traditionally fermented seeds. The wraps were allowed to ferment for 4 days in an incubator. Each wrap fermented with a different organism was evaluated for fermentative ability in bringing about desirable changes in color, texture, aroma and overall acceptability of the fermented beans seeds.
Sensory analysis: A panel of 10 adults who were conversant with the quality parameters evaluated the products. The panel rated the products based on color, texture and aroma. The rating was on 7 point hedonic scale where: 7 was dislike extremely, 4 was intermediate and 1 was like extremely for aroma and color. For texture, 7 was extremely hard, 4 was intermediate and 1 was extremely soft. Data obtained for all parameters were reported as means of 10 judgments. Analysis of variance (ANOVA) was computed for each sensory attribute (Snedecor and Cochran, 1976). Differences among sample means were separated using least significant difference (LSD) test.
 | |
| Fig. 1: | Flow chart for the fermentation of Iru, Ogiri, Ugba and Okpei |
Results and Discussion
Microorganisms isolated from the fermented iru, ogiri, ugba and okpei: Since the major constituents of these seeds are proteins, fats and carbohydrates, the organisms responsible for fermenting them must be capable of utilizing these three constituents. Most of the organisms isolated from the fermented seeds are known to posses such characteristics. The organisms isolated from fermented iru were Bacillus, Micrococcus and Staphylococcus species. Bacillus species were the predominant microorganisms present. These are known to have proteolytic ability and are also able to break down oils (Forgarty et al., 1974). Bacillus subtillis has been associated with fermenting locust bean for iru production (Antai and Ibrahim, 1986) and for fermenting soy bean for natto production (Hesseltine, 1965).
| Table 1: | Colony Characteristics of the Isolates |
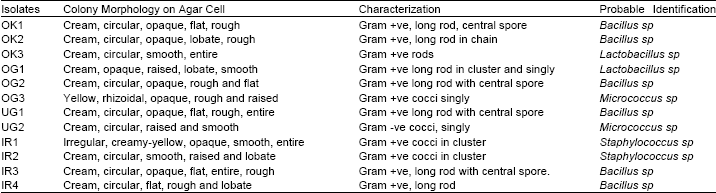 | |
Table 2: | Biochemical and morphological characteristics of isolated organisms from the condiments {okpei, ogiri, ugba, iru.} |
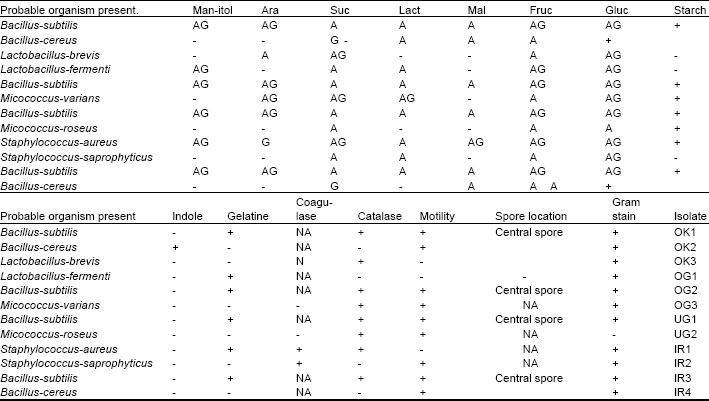 | |
| Foot Note: - = negative, AG = Acid and Gas, Glu=Glucose, Ara = Arabinose, OK = Okpei, IR = Iru, + = Positive,A = Acid, Mal = Maltose, Suc = Sucrose, UG = Ugba, NA = not applicable, G = Gas, Lact = Lactose, OG = Ogiri. | |
Although Staphylococcus sp. and Micrococcus sp. were isolated from the fermented bean, they were present in low numbers compared to Bacillus sp. They did not appear to be important in the fermentation process. The organisms isolated from fermented castor oil seed (ogiri) were species of Bacillus, Micrococcus and Lactobacillus. As in the case of iru, the predominant organisms observed were Bacillus sp. Lactobacillus was present probably because temperatures above 22oC favour its growth. Lactobacillus produces acid, which further inhibits the growth of non desirable organisms. The organisms isolated from ugba were species of Bacillus and Micrococcus. Bacillus species were the predominant microorganisms present. This agrees with the observation of Isu and Njoku (1997) that Bacillus species constitute over 95% of the total microbial population density in ugba fermentation. This may be because Bacillus cells exhibit very high protease activity compared with the other bacteria isolates. Organisms isolated from fermented mesquite seeds were Bacillus sp. and Lactobacillus sp. Like the other products discussed earlier, the Bacillus sp. were predominant with the other organisms adding little or no value to the product.
Biochemical and morphological characteristics of isolates: Bacillus sp. Are known to utilize the three major constituents of raw oil bean seeds, that is, protein, carbohydrate and oil very well because they are important sources of lipolytic enzymes as well as proteases and amylases (Pederson, 1979). Tables 1 and 2 show the biochemical and morphological characteristics of the isolated organisms.
| Table 3: | Mean scores of the sensory evaluation of the fermented products* |
| (a) “IRU” Mean Score | |
 | |
| IR1 | = | sample fermented with Staphylococcus aureus |
| IR2 | = | sample fermented with Staphylococcus saprophyticus |
| IR3 | = | sample fermented with Bacillus subtilis |
| IR4 | = | sample fermented with Bacillus cereus |
| (b) “OGIRI” Mean Score |
|
| OG1 | = | sample fermented with Lactobacillus fermenti |
| OG2 | = | sample fermented with Bacillus subtilis |
| OG3 | = | sample fermented with Micrococcus varians |
| (c) “OKPEI” Mean Score |
 |
| OK1 | = | sample fermented with Bacillus subtilis |
| OK2 | = | sample fermented with Bacillus cereus |
| OK3 | = | Sample fermented with Lactobacillus brevis |
| (d) “UGBA” Mean Score |
| Ug1 | = | sample fermented with Bacillus subtilis |
| Ug2 | = | sample fermented with Micrococcus roseus |
| *Means with the same letter(s) in a column are not significantly different. |
Bacillus subtilis and Bacillus cereus are similar morphologically but biochemically, Bacillus subtilis is arabinose positive while Bacillus cereus is negative to the test (Turchetti, 1982). B. subtilis is indole negative while B. cereus is positive to the test. B subtilis is positive to manitol while B. cereus is not. B subtilis hydrolyses gelatine while B. cereus does not hydrolyze it. Micrococus varians and Micrococcus roseus are similar morphologically and to a great extent biochemically. Pigmentation is the only character by which M. roseus can be distinguished from certain other micrococci (Cowan and Steel, 1966). M. roseus consists of strains with a common feature in producing a pink pigment, but may differ in biological character. The Micrococcus sp. are typically non motile and catalase positive and aerobic in nature. The Staphylococcus sp. are similar morphologically, but biochemically, S. aureus hydrolyses gelatine while S. saprophyticus is negative to the test. Also S. aureus hydrolyses starch while S. saprophyticus does not. Lactobacillus sp. also differ biochemically. L. brevis is catalase positive while L. fermenti is negative to the test. L. fermenti hydrolyses gelatine while L. brevis does not hydrolyze it. L. fermenti is positive to manitol test while L. brevis is negative to the test.
Sensory evaluation of the products fermented by different identified organisms: Significant differences exist among the various organisms in their ability to ferment locust bean seeds, castor oil seeds, mesquite seeds and African oil bean seeds (Table 3). Among the iru samples, the color of IR3 (fermented by Bacillus subtilis) was the most preferred by the judges followed by sample IR4. The color may have been developed by the microbial activity during the fermentation process. Sample IR3 gave the preferred iru aroma at (P = 0.05).
Samples IR1 and IR2 were not significantly different and were rated poorer than others. There was a slight difference between samples IR3 and IR4. Sample IR3 was significantly different from samples IR1 and IR2 and was rated better than others in terms of product texture. The results show that Staphylococcus species were not involved in the fermentation. Among the ogiri samples, OG2 was significantly different from samples OG1 and OG3. The color of OG2 (fermented by Bacillus subtilis) was most preferred, followed by that of sample OG3. There was no significant difference between samples OG1 and OG3. The aroma of Sample OG2 was most preferred and this could be attributed to the action of the fermenting organisms. The okpei sample OK1 (fermented by Bacillus subtilis) was preferred by the judges to the other samples in terms of colour, aroma and texture. There was no significant difference between samples OK2 and OK3. The results show that Lactobacillus brevis and Bacillus cereus did not contribute much to the fermentation. Among ugba samples, UG1 was preferred by the judges. It gave the desired colour, aroma and texture of ugba at P = 0.05.
Conclusion: Results of the present study show that the following organisms were isolated from the respective fermented oil seed samples. Iru-Staphylococcus aureus, Staphylococcus saprophyticus, Bacillus subtilis and Bacillus cereus. Ogiri-Lactobacillus fermenti, Bacillus subtilis, and Micrococcus varians. Okpei-Bacillus subtilis, Bacillus cereus and Lactobacillus brevis. Ugba-Bacillus subtilis and Micrococcus roseus. Bacillus subtilis was found to be common to all of them. It fermented the seeds very well and gave the desired colour, texture and aroma to the respective seeds fermented. Overall, all the judges preferred the samples that were fermented with Bacillus subtilis to those fermented with the other isolated organisms. Therefore, it could be concluded that Bacillus subtilis is the predominant organism responsible for the fermentation of protein-rich oil seeds to give the desired fermented products (ogiri, iru, ugba and okpei)
REFERENCES
- Antai, S.P. and H.P. Ibrahim, 1986. Micro-organisms associated with African locust bean (Parkia filicoidea Welw) fermentation for dawadawa production. J. Applied Bacteriol., 61: 145-148.
CrossRefDirect Link - Hesseltine, C.W., 1965. A millennium of fungi, food and fermentations. Mycologia, 57: 149-197.
PubMedDirect Link - Obeta, J.A.N. and M.A.A. Ugwuanyi, 1996. Microbiology of the production of okpeye, a condiment from seeds of Prosopis africana Taub. Trop. Sci., 36: 206-210.
Direct Link - Odunfa, S.A., 1981. Microorganisms associated with fermentation of African locust bean (Parkia filicoidea) during iru preparation. J. Plant Foods, 3: 245-250.
CrossRefDirect Link









