Research Article
Effect of Acacia nilotica Fruit Extract on Serum Glucose and Lipid Concentrations in Alloxan-induced Diabetic Rats
Department of Biochemistry, King Saud University, P.O. Box 2455, Riyadh 11451, Saudi Arabia
Acacia nilotica Delile (Fabaceae) is a spiny tree widely distributed in tropical and subtropical regions of Africa. In folk medicine, A. nilotica has been used for treating haemorrhages, tuberculosis, leprosy, colds and diarrhoea (New, 1984; Al-Mustafa and Dafallah, 2000). The efficacy of A. nilotica in the treatment of diabetes has been previously reported (Akhtar and Khan, 1985). The methanolic extract of A. nilotica was observed to cause a decrease in the concentration of plasma glucose, cholesterol, triglycerides and low density lipoprotein in alloxan-induced diabetic rabbits (Ahmad et al., 2008). Several reports have indicated a strong antioxidant potential of A. nilotica extracts in metal ion chelation and free radicals scavenging (Sultana et al., 2007; Singh et al., 2009).
To our knowledge, no report study the effect of A. nilotica fruit on diabetic animal models, however, the present study was conducted to investigate the effects of A. nilotica fruit extract on serum concentrations of Total Cholesterol (TC), Low-density Lipoprotein Cholesterol (LDL-C), High-density Lipoprotein Cholesterol (HDL-C), Triglycerides (TG) and glucose in alloxan-induced diabetic rats.
Chemicals and reagents: Alloxan was obtained from CDH, Ltd. (New Delhi, India). Methanol and other chemicals were of analytical grade and were obtained from BDH (Poole, UK). Gallic acid, Folin-Ciocalteu reagent, quercetin,2,2-azinobis (3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS) and potassium persulfate were purchased from Sigma-Aldrich (St. Louis, MO,USA). Trolox (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid) was obtained from Fluka Chemie AG (Buchs, Switzerland). Kits for the assays of (TC), (LDL-C), (HDL-C), (TG) and glucose were purchased from Human GmbH (Wiesbaden, Germany).
Plant material and extraction: Acacia nilotica fruit was collected from the western province of Sudan in April 2011 and identified by Dr. A. Almeligy, Department of Botany, King Saud University, Riyadh, Saudi Arabia. The A. nilotica fruit was immersed in liquid nitrogen prior to blending and 100 g of powder of the A. nilotica fruit was prepared with an electric blender. The methanol extract of the A. nilotica powder was obtained by dissolving the 100 g of A. nilotica powder in 900 mL methanol-water (4:1v/v) at room temperature overnight using an orbital shaker. The filtrates were pooled and centrifuged at 8000 rpm for 10 min and the supernatant was concentrated under reduced pressure at 40°C using a rotary evaporator which yielded 22.3 g of A. nilotica methanol extract powder.
Determination of antioxidants in A. nilotica fruit: Using the aforementioned protocol, 5 g of powdered A. nilotica fruit was extracted with 50 mL methanol-water (4:1 v/v). The final extract was dissolved in distilled water, the final volume was recorded and the suspended extract was divided into aliquots and kept in the dark at room temperature until determination of antioxidant properties.
Total phenolics: The concentration of total phenolics was determined using the method of Kim et al. (2003). The total phenolic content of A. nilotica fruit was expressed as grams of gallic acid per 100 g of A. nilotica.
Total flavonoid: The concentration of total flavonoid was measured with the aluminum chloride colorimetric method described by Chang et al. (2002). The total flavonoid content of A. nilotica fruit was expressed as milligrams of quercetin per 100 g of A. nilotica.
Radical cation ABTS scavenging activity: The ABTS free radical decolorization assay was performed as described by Re et al. (1999), using Trolox as a standard. The free radical scavenging activity of A. nilotica fruit extract was expressed as the Trolox equivalent antioxidant capacity mg Trolox equivalents per 100 g of A. nilotica.
Animal groups and dosage: Male Wistar rats (180-205 g) were obtained from the animal house at the College of Pharmacy, King Saud University. All animals were housed in cages with a 12/12 h light/dark cycle at 22°C. The animals were given rat chow and water ad libitum. Diabetes was induced via an intraperitoneal injection of alloxan (150 mg kg-1 dissolved in 0.2 mL of physiological saline). The animals were kept under observation for 1 week prior to the start of treatment. All animal experiments were carried out in accordance with the King Saud University Ethical Committee Acts. Extract of A. nilotica was administered orally at a volume of 0.2 mL, equivalent to a concentration of 200 mg kg-1 b.wt., using oral gavage. Supplementation with extract continued regularly on a daily basis over a period of 35 days.
The rats were divided into four groups: Normal Control rats (NC), normal control rats administered A. nilotica (NC+AN), Diabetic Control rats (DC) and diabetic rats administered A. nilotica (DC+AN). Each group comprised 10 to 14 rats. Body weights and food intake of all groups were recorded daily. Aliquots of blood samples from all groups were collected weekly in plain tubes. Serum samples were separated from cells by centrifugation at 5000xg in a refrigerated centrifuge.
Biochemical assays: Concentrations of the following biochemical parameters were determined in serum samples by standard enzymatic methods using Human kits on a Humalyzer 3000 (Human GmbH, Wiesbaden, Germany): TC (Richmond, 1973), LDL-C (Okada et al., 1998), HDL-C (Izawa et al., 1997), TG (Nagele et al., 1984) and glucose (Barham and Trinder, 1972).
Data analysis: The data are presented as Means±SD. Comparisons between experimental and corresponding control rats were performed using Student’s t-test. The probability value p<0.05 was considered significant.
Body weights and food intake of control and alloxan diabetic rats administered A. nilotica fruit extract: Table 1 shows that gains in body weight and food intake in DC rats was significantly reduced (p<0.001) compared with those in NC rats. However, NC+AN and DC+AN did not show any significant changes with regard to these parameters.
Antioxidant activity of A. nilotica fruit extract: The percentage concentration of total phenolics, total flavonoid and Trolox equivalent of A. nilotica fruit extracts are shown in Table 2. A high concentration of total phenolics was observed (30.65±2.2 g per 100 g) in the A. nilotica fruit extract. Present results are in agreement with previous reports in which considerable amounts of total phenolics were reported in bark extracts of A. nilotica (Sultana et al., 2007; Singh et al., 2010).
| Table 1: | General characteristics of control and alloxan-induced diabetic rats supplemented with A. nilotica fruit extract |
 | |
| Data are expressed as Means±SD, with number of rats given in parentheses, As compared with matched groups, *p<0.001 (student t-test) | |
| Table 2: | Concentration of total phenolics, total flavonoid and Trolox equivalent scavenging activity in A. nilotica fruit extract |
| Data are Mean±SD (n = 3), All calculations made on dry mass basis | |
| Table 3: | Effect of the A. nilotica fruits extract on serum levels of glucose, total cholesterol (TC), triglyceride (TG), low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C) in normal control rats for 5 weeks |
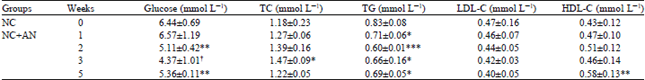 | |
| Data are expressed as Mean±SD, with number of rats given in parentheses. As compared with matched groups, *p<0.05, **p<0.02, †p<0.005 ***p<0.001 (student t test) | |
| Table 4: | Effect of the A. nilotica fruits extract on serum levels of glucose, total cholesterol (TC), triglyceride (TG), low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C) in alloxan-induced diabetic rats for 5 weeks |
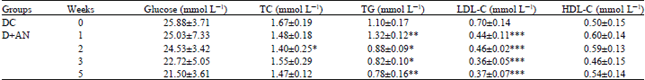 | |
| Data are expressed as Mean±SD, As compared with matched groups, *p<0.05, **p<0.02, ***p<0.001 (student t-test) | |
The total flavonoid (as quercetin equivalent) percentage concentration in A. nilotica fruit extract was 227.21±5.7 mg per 100 g in the present study; however, Sultana et al. (2007) previously reported that the percentage concentration of total flavonoid (as catechin equivalent) in bark extract of A. nilotica was 3.21±0.12 g per 100 g. The difference in the total flavonoid concentration was because Sultana et al. (2007) used the bark of the plant, whereas the present study used the fruit of the plant. Furthermore, Sultana et al. (2007) expressed total flavonoid as catechin equivalent; however, in the present study, total flavonoid were expressed as quercetin equivalent.
Effect of A. nilotica fruit extract on serum concentration of glucose and lipids of control and alloxan diabetic rats: Table 3 shows that the serum glucose concentration of NC+AN rats was significantly decreased compared with that of the NC rats. The decrease in serum glucose level was observed starting from the second week of supplementation (p<0.02) and continued until the third and fifth weeks (p<0.005 and p<0.02, respectively). On the other hand, no significant effect was noticed in the DC+AN rats throughout the study (Table 4), although the serum glucose concentration was decreased from 25.88 to 21.5 mmol L-1 during the fifth week of A. nilotica administration. However, this decrease was not significant. Asad et al. (2011) reported that the A. nilotica leaf extract at a dose of 300 mg kg-1 b.wt. produced hypoglycemic and antiplatelet aggregation activity in streptozotocin-induced diabetic rats and that the effects were comparable to those of glyburide. In another report by Ahmad et al. (2008) aqueous methanolic extract of A. nilotica pods at a dose of 400 mg kg-1 b.wt. caused a decrease in blood glucose levels in alloxan-induced diabetic rabbits.
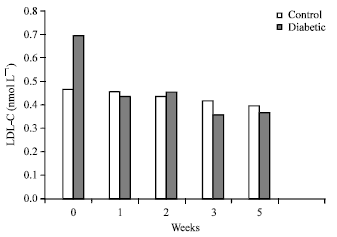 | |
| Fig. 1: | Effect of A. nilotica fruit extract on serum LDL-C levels of control and alloxan-induced diabetic rats |
Although no clear effect was noticed in serum TC concentrations of NC and DC rats, significant (p<0.001) decreases in the second (37%) and fourth (47%) weeks were observed in the serum LDL-C concentration of DC+AN (Fig. 1, Table 4). This beneficial effect of A. nilotica extract on the LDL-C could be due to the presence of a high concentration of polyphenol compounds, which are known to have antioxidant properties. This finding on the effect of A. nilotica extract on LDL-C is comparable to that in the report of Lerman et al. (2010) which suggested that individuals with a high risk of developing cardiovascular diseases may benefit from a medical food containing proanthocyanidins from A. nilotica because of the observed decreases in their LDL-C levels. A significant decrease in the serum TG level was also observed in NC+AN and DC+AN rats (Table 3 and 4). In NC+AN rats, the serum levels of TG decreased by 28% during the second week of supplementation (p<0.001).
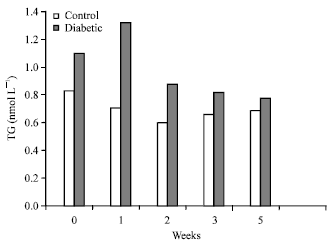 | |
| Fig. 2: | Effect of A. nilotica fruit extract on serum TG levels of control and alloxan-induced diabetic rats |
Hypertriglyceridemia reportedly occurs in alloxan-induced diabetic animal models, (Wojtowicz et al., 2004; Abuelgassim, 2010) and the significant increase observed in the present experiments was in agreement with these reports. Diabetes is characterized by an accumulation of fatty acyl-CoA and diacylglycerol in muscle or liver cells which interferes with the insulin signaling process. Serum TG levels were significantly reduced in DC+AN rats compared with DC rats (Fig. 2, Table 4). This decrease in serum TG level could be attributed to the high concentration of antioxidants present in A. nilotica fruit extract. However, serum HDL-C concentrations did not significantly change in both NC+AN and DC+AN rats compared to their matched control rats, except during the fifth week in which the serum HDL-C level of NC+AN increased from 0.43 to 0.58 mmol L-1 (p<0.02) (Table 3).
Although, the current study did not reveal any significant decreases in the serum glucose concentration of alloxan-diabetic rats, this investigation shows that oral administration A. nilotica fruit extract has a strong hypolipidemic effect in terms of significantly decreasing serum TG and LDL-C levels (p<0.02 and p<0.001, respectively).
To a certain extent, the results of the present study support the traditional use of A. nilotica as an ethnomedical plant. However, further detailed studies are needed to elucidate the mechanism of the hypolipidemic effects of the A. nilotica fruit and how this plant may be beneficial in atherosclerosis therapy. Our future work will focus on separation of the active ingredients of A. nilotica fruit extract and identification of their specific activities and roles in the treatment of atherogenic rats.
The findings of the present investigation suggest the possibility of utilizing A. nilotica fruit extract in a pharmaceutical preparation that could help in the treatment of diabetes related-complications and hyperlipidemia.
This study was financially supported by a grant from the Deanship of Scientific Research, College of Science Research Center, King Saud University.