ABSTRACT
The inhibition by sucrose of chromoplast reversion to chloroplast in citrus epicarp was studied by observing the effects of several sugars, sugar metabolites and 1-iodoacetate on chlorophyll reaccumulation in cultured Citrus paradisi Macf. pericarp segments. Pericarp segments of 1 cm in diameter were cut from yellow fruits and cultured on modified medium plus the indicated metabolites and kept under continuous fluorescent light. Accumulation of chlorophyll in the segments was measured with a spectrophotometer fitted with sphere reflectometer. Respiration was determined via., an infrared gas analyzer. Inhibition of regreening was not specific to a particular sugar. The organic acids malate, citrate, succinate, 2-oxoglutarate and especially malonate elicited effects similar to sucrose, but at much lower concentrations. However, malonate inhibition of chlorophyll accumulation was overcome by increased concentrations of glutamine. At concentrations that usually inhibited chlorophyll, malonate did not reduce CO2 production in the presence of glutamine or KNO3. Sucrose effects on regreening were reduced by 1-iodoacetate. These results indicate that sugar regulation of plastid reversion during regreening in citrus epicarp is not directly due to sugars, but is instead mediated through metabolism of sugars to organic acids, especially malonic acid.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2009.246.251
URL: https://scialert.net/abstract/?doi=pjbs.2009.246.251
INTRODUCTION
In the subtropics, citrus fruits degreen during the cool winter season and when left on the tree, certain varieties regreen during the ensuing spring and summer (Matile et al., 1999; Hortensteiner, 2006). Ultrastructural studies by Mayfield and Huff (1986) demonstrated the regreening of Valencia orange epicarp (C. sinensis L. Osbec, cv. Valencia) signals a partial reversion of chromoplasts to chloroplasts. Chloroplast-chromoplast transition, in tomato, is marked by the accumulation of carotenoids and the disappearance of chlorophyll, the degradation of the highly structured thylakoid membrane system and a reduction in the levels of proteins and mRNAs (Cheung et al., 1993). In citrus, the interconversion of plastids and consequential degreening and regreening has been related to the accumulation of sugars (mainly reducing sugars) in the epicarp during the winter and a reduction in sugar content in the spring (Matile et al., 1999; Hortensteiner, 2006). In addition, in vivo sucrose supplementation to Satsuma mandarin (Citrus unshiu Mark. cv. Okitsu) epicarp promoted sucrose accumulation and advanced colour break (Domingo et al., 2001).
Cultured epicarp segments will regreen, or remain green, when kept on media with low (15 mM) sucrose concentrations, whereas similar segments on media with high (150 mM) sucrose concentrations will either degreen or are inhibited from regreening (Domingo et al., 2001). Glucose and fructose affect regreening and degreening similarly to sucrose, whereas several nitrogenous substances counteract the effects of these sugars (Hortensteiner, 2006). Similarly, in Satsuma mandarin (Citrus unshiu Mark. cv. Okitsu) colour break correlated positively with sucrose content and negatively with nitrogen content (Domingo et al., 2001). Supply of nitrogen did not inhibit bleaching in Chlorella protothecoides (Hortensteiner et al., 2000). Although the foregoing studies have shown that sugars are factors regulating plastid interconversions in citrus epicarp, it is not known whether the effects are due specifically to sugars or sugar metabolites such as organic acids.
Various carbohydrates also influence chlorophyll accumulation in several other organisms and suggestions as to the primary inhibiting substances vary. Chlorophyll content in Citrus sinensis epicarp correlated best with total sugar concentrations, which were predominantly glucose and fructose (Domingo et al., 2001). Ethanol and acetate inhibition of chlorophyll accumulation in Euglena gracilis klebs appears to differ from sucrose inhibition and may be mediated through glyoxylate metabolism (Hortensteiner et al., 2000). The suppression of chlorophyll synthesis by sugars may be analogous to the glucose effect on heme biosynthesis. Chlorophyll and heme biosynthesis share hepatic delta-aminolevulinic acid (ALA) synthase as a commonfirst intermediate (Pruzinska et al., 2007). Puntel et al. (2005) observed that glucose metabolites were less effective than glucose in inhibiting the induction of ALA synthase in rat brain and concluded that glucose was the effective substance.
Domingo et al. (2001) suggested that sugar regulation of chloroplast to chromoplast conversion in citrus fruit epicarp appears to operate via., ethylene. Hortensteiner (2006) identified chlorophyllase among three chlorophyll catabolic enzymes and concluded that chlorophyll breakdown is a prerequisite to permit the remobilization of nitrogen from chlorophyll-binding proteins to proceed during senescence. The expression of chlorophyllase (chlase 1) in oranges is enhanced by exogenous application of ethylene (Jacob-Wilk et al., 1999). The regulatory role of ethylene in degreening citrus suggests that endogenous levels of C2H4 regulate ethylene production (Thomas et al., 2003). Alexander and Grierson (2002) elucidated the ethylene-signaling pathway from receptor through intermediate components and predicted a model to control the expression of ethylene-regulated genes for climacteric fruit ripening. Further studies on what controls these processes in non-climacteric fruits (like citrus) are required for the understanding of ripening in this group.
The objective of this study was to determine whether chromoplast reversion as signaled by the accumulation of chlorophyll in citrus epicarp is regulated by specific sugars or mediated through some metabolites such as organic acids. Results obtained when C. paradisi (Macf) peel segments were regreened on media with a variety of sugars, organic acids and 1-iodoacetate indicate that sugar inhibition of regreening is closely associated with organic acids especially malonic acid.
MATERIALS AND METHODS
Plant materials and pericarp culture: Yellow coloured grapefruit, (Citrus paradisi, (Macf) cv. Marsh), were obtained from the University of Arizona Experimental Farm at Tempe, Arizona from November, 1983 through June, 1986. Fruits were surface sterilized with 0.5% (v/v) sodium hypochlorite and segments of pericarp 1 cm in diameter were cut from strips submerged in sterile water using a cork borer. These segments were then cultured on a basal medium with indicated additives as previously described by Mayfield and Huff (1986) and kept under continuous fluorescent light (Sylvania Cool White) between 4 and 12 W m-2 at about 30 °C for 14 days.
Chlorophyll determination: Chlorophyll content of individual segments was determined in situ, initially and after 14 days, using differences in absorbance at 675 and 735 nm measured with a spectrophotometer fitted with an integrated sphere reflectometer. Absorbance data were converted to nmol chlorophyll per segment using a standard curve prepared by comparing mean A675-A735 with the concentration of extracted chlorophyll from groups of similar segments (Mayfield and Huff, 1986).
Respiration: Respiration of pericarp segments was periodically measured by sampling CO2 produced by groups of three segments cultured in 25 mL Erlenmeyer flasks over time. The flasks were temporarily sealed on days 1, 7 and 14 and a 3 mL air sample removed by a syringe initially and again after 10 min incubation in the dark at 24 °C. Carbon dioxide in the air samples was determined using an infrared gas analyzer. After the final sampling at 14 days, flask headspace was measured by displacement with water and the segments blotted dry and weighed.
RESULTS AND DISCUSSION
When partially degreened pericarp segments were cultured on media with several different sugars at 15 mM, degreening was reversed and the segments reaccumulated chlorophyll (Fig. 1). On similar media with the sugar content increased to 150 mm, the pentoses, ribose and xylose inhibited reaccumulation of chlorophyll, whereas sucrose along with the hexoses, mannose and galactose sustained a continued loss of chlorophyll.
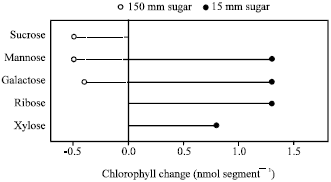 | |
| Fig. 1: | Chlorophyll content of pericarp segments kept 14 days under continuous fluorescent light on media containing several sugars. Each datum is the mean loss (--) or gain (–) of chlorophyll in 10 segments from an initial mean of 1.9 ± 0.3 nmol chlorophyll segment-1 |
All of the tricarboxylic acid cycle (TCA) intermediates tested, here, inhibited chlorophyll reaccumulation in fully degreened pericarp (Table 1). With the exception of succinate, inhibition by TCA intermediates was greater than by sucrose at the same concentration.
Both citrate and sucrose exhibited dual behavior by inhibiting chlorophyll accumulation at high concentrations and promoting chlorophyll accumulation at low concentrations (Table 2). Both the promotion of chlorophyll accumulation by low sucrose concentrations and inhibition by high sucrose concentrations were reduced by 1-iodoacetate (Table 3), which irreversibly inhibits glyceraldehyde 3-phosphate dehydrogenase, and phosphoenol-pyruvate (PEP) carboxylase.
Malonic acid is the most prevalent organic acid other than malic acid in citrus epicarp and was the most effective inhibitor of chlorophyll reaccumulation tested (Table 4). As with sucrose inhibition, malonate inhibition of chlorophyll accumulation was overcome by increased concentrations of glutamine (Table 5). In the present study, malonate, an inhibitor of succinate dehydrogenase and respiration (Kim, 2002), did not reduce CO2 production in the presence of KNO3 or glutamine at concentrations that strongly inhibited chlorophyll accumulation (Fig. 2).
Results presented here indicate that sugar regulation of the reversion of chromoplasts to chloroplasts in regreening citrus epicarp is not specific to any particular sugar, but is rather mediated by metabolism of sugars to organic acids, especially malonic acid via., oxaloacetic acid (OAA) and phosphoenol-pyruvate (PEP).
Sugar regulation of chloroplast development is not specific since several sugars, including both hexoses and pentoses, inhibited chlorophyll reaccumulation (Fig. 1).
| Table 1: | Inhibition of chlorophyll accumulation in Citrus paradisi pericarp segments by intermediates of the tricarboxylic acid cycle, pyruvate and sucroseA |
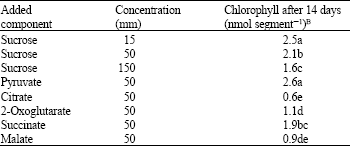 | |
| ASegments were kept on media containing 5 mm KNO3 and the indicated acid or sucrose. Mean ± SE initial chlorophyll was 0.18 ± 0.03 nmol segment-1. BValues are means of 10 segments and those followed by the same letter are not significantly different | |
 | |
| Fig. 2: | Malonate inhibition of chlorophyll accumulation is not accompanied by inhibition of respiration. Segments were kept 14 days under continuous fluorescent light on media containing 15 mM sucrose and 5 mM KNO3 (•) or glutamine (GLN) |
| Table 2: | Dual effects of citrate and sucrose on chlorophyll accumulation during chromoplast reversion in C. paradisi pericarp segmentsA |
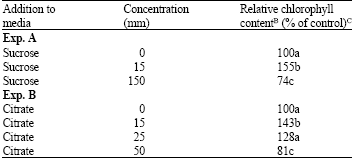 | |
| ASegments were kept on media containing 5 mm KNO3 in addition to the indicated concentrations of sucrose and citrate.BThe amount of chlorophyll in the untreated segments was 1.99 ± 0.06 and 2.76 ± 0.19 nmol segment-1 in experiments A and B, respectively. C Means are of 10 segments and those followed by the same letter are not significantly different | |
| Table 3: | Iodoacetate inhibition of sucrose effects on chlorophyll accumulation in C. paradisi (Macf) pericarp segmentsA |
 | |
| ASegments were kept on media containing 5 mM KNO3, 15 mM and 300 mm sucrose and 1 mM 1-iodoacetate. BEach value represents the Mean ± SE of 10 segments | |
| Table 4: | Malonate inhibition of chlorophyll accumulation in yellow C. paradisi epicarp segmentsA |
 | |
| Asegments were kept on media containing 5 mM KNO3, 15 mM sucrose and malonate. Bcontrol segments without malonate gained 2.16 ± 0.16 nmol segment-1 | |
| Table 5: | Glutamine counteracts malonate inhibition of chlorophyll reaccumulation in C. paradisi peel segmentsA |
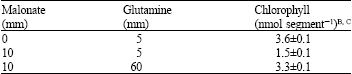 | |
| ASegments were kept on media containing 15 mM sucrose along with the indicated amounts of malonate and glutamine. BInitial chlorophyll was 0.43 ± 0.6 nmol segment-1. CEach value is the Mean ± SE of 10 segments | |
That sugar metabolism via., glycolysis is involved in the inhibition of regreening is indicated by the greater efficacy of organic acids than sugars in inhibiting chlorophyll accumulation (Table 1). It is also indicated by the ability of 1-iodoacetate, which inhibits glyceraldehyde 3-phosphate dehydrogenase, to suppress both the promotive effect of low sucrose concentrations and the inhibitive effect of high sucrose concentrations on regreening (Table 3). Although 1-iodoacetate inhibited PEP carboxylase, this does not, as will be discussed later, affect the overall conclusions drawn here. Sugars and organic acids might have inhibited chlorophyll reaccumulation by separate mechanisms. The antagonism between glutamine and both malonate and sucrose (Table 5), along with the similarity between citrate and sucrose in both promoting and inhibiting regreening (Table 2) suggest a common mechanism.
While it is apparent that metabolism via., glycolysis is a part of the mechanism by which sugars affect plastid differentiation in yellow epicarp tissue, metabolic reactions beyond PEP are speculative. Presumably, low concentrations of sucrose promote regreening by providing energy through oxidative respiration. However, it appears that there is a substantial diversion of carbon to pathways other than through pyruvate to the TCA cycle. It is likely that malate is also a product of glycolysis and that glycolytic intermediates enter the TCA cycle via., the malate pathway concurrently with the pyruvate pathway (Puntel et al., 2005, 2007).
During the maturation of juice vesicles in Hamlin oranges and other citrus fruits, the ratio of NADH to NAD increased (Domingo et al., 2001). Also, levels of ethanol, malate and the enzymes pyruvate decarboxylase, PEP carboxylase and malic enzyme, increased, while levels of pyruvate and OAA declined (Fedotcheva et al., 2006). The increased NADH to NAD ratio presumably inhibited the oxidation of malate and pyruvate by malate and pyruvate dehydrogenases and by malic enzymes thereby reducing the entry of glycolytic intermediates into the TCA oxidative pathway. This combined with the increased activities of pyruvate decarboxylase and PEP carboxylase accounted for the increases in ethanol and malate.
Since, malate and malonate and aspartate are the principle organic acids and free amino acids in citrus epicarp, it appears by analogy with the juice vesicles that a significant amount of PEP in the epicarp is diverted through OAA via the action of PEP carboxylase. This conclusion is reinforced by labeling studies in Valencia oranges epicarp that showed substantial dark 14CO2 fixation into aspartate, malate and serine, but not citrate (Matile et al., 1999). Oxaloacetate can be irreversibly metabolized to malonate (Kim, 2002; Puntel et al., 2007), while exogenously supplied citrate and malate can be reversibly oxidized to OAA. If pyruvate was diverted to ethanol or was otherwise poorly incorporated into citrate as in the vesicles this would explain why pyruvate had little effect on chlorophyll reaccumulation.
All organic acids might have directly influenced plastid differentiation (Thomas et al., 2003). However, their conversion to malonate seems more likely to be the mechanism by which they inhibited regreening. All of the acids that inhibited regreening could act through their common dicarboxylic acid functional grouping by mechanisms such as metal chelation (Puntel et al., 2005, 2007) or by the inhibition of membrane transport (Puntel et al., 2007). However, metabolism of the exogenous sugars and acids to malonate is favored because of the greater efficacy exhibited by malonate in inhibiting chlorophyll accumulation at very low concentrations (Table 4). Malonate can be formed via., OAA from any of the organic acids that inhibited regreening (Kim, 2002). The ability of 1-iodoacetate to counteract sucrose effects on chlorophyll reaccumulation (Table 3) would be reinforced by its ability to inhibit PEP carboxylase.
Malonate inhibition of plastid differentiation in yellow citrus epicarp does not appear to be through the inhibition of respiration by its action on succinate dehydrogenase in spite of the fact that several TCA intermediates inhibited chlorophyll reaccumulation. The ability of malonate to inhibit succinate dehydrogenase and respiration and to cause accumulation of succinate is dependent on both pH and its concentration within the tissue (Kim, 2002). At concentrations and pH, that malonate strongly inhibited chlorophyll in citrus epicarp; malonate had little or no apparent effect on pericarp respiration (Fig. 2). Under similar conditions, malonate also had no effect on respiration and succinate concentrations in carrot root tissues (Kim, 2002). The ability of malonate to affect plastid differentiation in citrus epicarp is, therefore, considerably stronger than and independent of, its ability to affect the TCA cycle and respiration.
Malonate concentrations in citrus epicarp are low compared with values of 5 to 25 nmol kg-1 fr. wt. in green leaves of various species (Matile et al., 1999). In addition, malonate concentrations in citrus epicarp may be declining during the period of chlorophyll loss (Thomas et al., 2003). These observations suggest that further metabolism of free malonate is more important to plastid differentiation in citrus epicarp than malonate itself. Unfortunately, there is little information on the metabolism of free malonate in plant tissues. Malonic dialdehyde and malonyl coenzyme A may be important precursors of malonate (Davydov, 1993). About half of the labeled carbon from 2-14C-malonate provided to leaves of citrus, was found in citrate and malate (Kim, 2002), probably by formation of malonyl CoA followed by loss of CO2 to give acetyl CoA (An and Kim, 1998). An and Kim (1998) confirmed the formation of acetyl-CoA and malonyl-CoA from malonate by thin-layer chromatographic analysis and suggested the gene cluster encodes proteins involved in the malonate-metabolizing system in Rhizobium trifolii.
CONCLUSION
Certain citrus varieties revert their colour and regreen during the hot summer (or spring) if left on the tree. This is a big problem in Sudan and subtropical areas. Although this does not affect the internal quality of the fruits, consumers are often reluctant to purchase such fruits. The regreening is due to increased nitrogen assimilation or reduction in carbohydrate metabolism within the tissues. Reduction in nitrogen fertilization towards the end of the season minimized the problem of regreening.
The inhibition of regreening of Citrus paradise Macf. was studied when peel segments were cultured on media containing several sugars, organic acids and 1-iodoacetate. It was found that colour reversion is not due to sugars, but was rather due to organic acids, especially malonic acid. Malonic acid prevented regreening at very low concentrations. Further studies along this line should focus on the metabolism of free malonate in plant tissues.
ACKNOWLEDGMENT
Published with the approval of the Director of the University of Arizona Agricultural Experimental Station.
REFERENCES
- Alexander, L. and D. Grierson, 2002. Ethylene biosynthesis and action in tomato: A model for climacteric fruit ripening. J. Exp. Bot., 53: 2039-2055.
CrossRefPubMedDirect Link - An, J.H. and Y.S. Kim, 1998. A gene cluster encoding malonyl-CoA decarboxylase (MatA), malonyl-CoA synthetase (MatB) and a putative dicarboxylate carrier protein (MatC) in Rhizobium trifolii. Eur. J. Biochem., 257: 395-402.
CrossRefDirect Link - Cheung, A.Y., T. McNellis and B. Piekos, 1993. Maintenance of chloroplast components during chromoplast differentiation in the tomato mutant green flesh. Plant Physiol., 101: 1223-1229.
CrossRefDirect Link - Davydov, V.V., 1993. Pathways of endogenous malonate formation in the rat liver. Ukr. Biokhim. Zh., 65: 85-88.
Direct Link - Domingo, J.I., R.T. Francisco, L. Francisco, P.M. Edwardo and T. Manuel, 2001. In vivo sucrose stimulation of colour change in citrus fruit epicarps; Interaction between nutritional and hormonal signals. Physiol. Plantarum, 112: 244-250.
CrossRef - Fedotcheva, N.I., A.P. Sokolov and M.N. Kondrashova, 2006. Nonezymatic formation of succinate in mitochondria under oxidative stress. Free Radic. Biol. Med., 41: 41-45.
CrossRef - Hortensteiner, S., J. Chinner, P. Matile, H. Thomas and I.S. Donnison, 2000. Chlorophyll breakdown in Chlorella protothecoides: Characterization of degreening and cloning of degreening-related genes. Plant Mol. Biol., 42: 439-450.
PubMed - Hortensteiner, S., 2006. Chlorophyll degradation during senescence. Ann. Rev. Plant Biol., 57: 55-77.
CrossRef - Jacob-Wilk, D., D. Holland, E.E. Goldschmidt, J. Riov and Y. Eyal, 1999. Chlorophyll breakdown by chlorophyllase: Isolation and functional expression of the Chlase1 gene from ethylene-treated Citrus fruit and its regulation during development. Plant J., 20: 653-661.
CrossRefDirect Link - Kim, Y.S., 2002. Malonate metabolism: Biochemistry, molecular biology, physiology and industrial application. J. Biochem. Mol. Biol., 35: 443-451.
PubMed - Matile, P., S. Hortensteiner and H. Thomas, 1999. Chlorophyll degradation. Annu. Rev. Plant Physiol. Plant Mol. Biol., 50: 67-95.
CrossRefDirect Link - Mayfield, S.P. and A. Huff, 1986. Accumulation of chlorophyll, chloroplastic proteins and thylakoid membranes during reversion of chromoplasts to chloroplasts in Citrus sinensis Epicarp. Plant Physiol., 81: 30-35.
CrossRefDirect Link - Pruzinska, A., I. Anders, S Aubry, N. Schenk, E. Tapernoux-Luthi, T. Muller, B. Krautler and S. Hortensteiner, 2007. In vivo participation of red chlorophyll catabolite reductase in chlorophyll breakdown. Plant Cell, 19: 369-387.
PubMed - Puntel, R.L., C.W. Nogueira and J.B.T. Rocha, 2005. Krebs cycle intermediates modulate Thiobarbituric Acid Reactive Species (TBARS) production in rat brain in vitro. Neurochem. Res., 30: 225-235.
CrossRefPubMedDirect Link - Puntel, R.L., D.H. Roos, D. Grotto, S.C. Garcia, C.W. Nogueira and J.B.T. Rocha, 2007. Antioxidant properties of Krebs cycle intermediates against malonate pro-oxidant activity in vitro: A comparative study using the colorimetric method and HPLC analysis to determine malondialdehyde in rat brain homogenates. Life Sci., 81: 51-62.
CrossRefPubMedDirect Link - Thomas, H., H.J. Ougham, C. Wagstaff and A.D. Stead, 2003. Defining senescence and death. J. Exp. Bot., 54: 1127-1132.
PubMed








